Abstract
Objective
The methionine synthase reductase (MTRR) gene encodes the MTRR enzyme involved in the metabolic pathway of homocysteine. Several studies investigated the effect of the MTRR rs1532268 gene polymorphism on the risk of gastric cancer (GC), but the results have been inconsistent.
Methods
We performed a comprehensive and systematic search of PubMed, Google Scholar, MEDLINE, Science Direct, Scopus, CNKI, and Web of Science. Five studies were included in this meta-analysis to determine whether MTRR rs1532268 polymorphism contributes to the risk of GC.
Results
Pooled data indicated that the MTRR rs1532268 polymorphism significantly increased GC risk under the allele comparison model (odds ratio [OR] = 1.14, 95% confidence interval [CI] = 1.01–1.29) and dominant model (OR = 1.14, 95% CI = 1.00–1.30). In the analysis stratified by ethnicity, no relationship was found in Whites or Asians.
Conclusion
Our meta-analysis suggests a positive correlation between MTRR rs1532268 polymorphism and GC development.
Introduction
Gastric cancer (GC) is one of the most aggressive malignancies, ranking third among cancers leading to death worldwide. In 2021, there were an estimated 26,560 new GC cases and 11,180 related deaths in the United States.1–2 Environmental factors, such as smoking, diet, and Helicobacter pylori infection, are contributors to the development of GC, and up to 10% of GC cases are estimated to be due to an underlying hereditary susceptibility caused by germline pathogenic variants. 3 Polymorphisms in various genes, changes in the expression of micro-RNAs and long noncoding RNAs, and alterations in microbiome profiles may result in GC. In addition, some of these alterations may be used for screening, treatment, and prognostic predictions. 4 Polymorphisms in several genes were found to be associated with GC, such as DAL-1, Mucin, TLR2, MTR, TP73, and TP53,5–6 and a prognostic model of GC was built according to the analysis of gene polymorphisms. 7
The methionine synthase reductase (MTRR) gene is positioned on the short arm of chromosome 5 (region 5p15.2 to 15.3) and encodes the MTRR enzyme involved in the metabolic pathway of homocysteine.8 Aberrant MTRR functions may contribute to carcinogenesis through altered DNA methylation (e.g., DNA hypomethylation) and impaired thymidylate synthesis, resulting in nucleotide imbalances, increased uracil misincorporation into DNA, DNA strand breaks, and impaired excision repair. As a result, the susceptibility of DNA to mutations and damage may be increased. 8 Seven polymorphisms have been reported in the MTRR gene, including Ex2-64A>G (rs1801394), Ex5 + 123C>T (rs1532268), Ex15 + 572C>T(rs9282787), Ex15-405A>T (rs8659), Ex9-85C>T (rs2287780), Ex15-526G>A (rs9332), and Ex14 + 14C>T (rs10380). 9 Among these polymorphisms, CT/TT or TT genotypes of the single nucleotide polymorphism rs1532268 in the MTRR gene coding region are significantly associated with a poorer overall survival compared with the CC genotype. 10 Several studies have evaluated the relationship between the risk of GC and the rs1532268 polyporphism,8,9,11–13 but the results have been inconsistent. Thus, we conducted this meta-analysis to assess the effect of the rs1532268 polymorphism on GC susceptibility.
Materials and methods
Publication search strategy
A comprehensive and systematic search of PubMed, Google Scholar, MEDLINE, Science Direct, Scopus, CNKI, and Web of Science was performed according to 2020 PRISMA guidelines using the following terms: “MTRR” or “methyltetrahydrofolate-homocysteine methyltransferase reductase”, “polymorphism” or “gene mutation” or “gene variation”, “stomach” or “gastric”, and “tumor” or “cancer” or “neoplasm” (the last search was updated 24 March 2022). All relevant publications were reviewed. There were no language or sample size limitations in the included studies. Articles in reference lists were also searched for potentially relevant publications. Because this study is a meta-analysis, the need for ethical approval and informed consent was waived. This investigation uses published or publicly available summary data. No original data were collected for this manuscript. Ethical approval for each of the studies included in the present analysis can be found in the original publications.
Inclusion and exclusion criteria
Studies included met the following criteria: (1) case-control study or cohort study, (2) evaluation of MTRR rs1532268 polymorphism and GC risk, and (3) sufficient data to examine the odds ratio (OR) with the 95% confidence interval (95% CI). The major criteria for exclusion were as follows: (1) not related to GC, (2) insufficient data for analysis, or (3) the distribution of genotypes among controls not in Hardy–Weinberg equilibrium.
Data extraction
The eligible data in studies were extracted by two investigators, and a consensus was reached by discussion. The following key information was extracted from each study: first author’s name, year of publication, ethnicity, numbers of cases and controls with CC, CT, and TT genotypes, and sample size of cases and controls. For subgroup analyses, the ethnic populations were classified into Asian and White.
Statistical analysis
The OR with 95% CI was used to measure the strength of association between the MTRR rs1532268 polymorphism and GC risk. Hardy–Weinberg equilibrium in the control group was tested using the Pearson chi-square test for the goodness of fit, and P < 0.05 was considered significant. The statistical significance of the pooled OR was assessed using a Z test, with a two-tailed P < 0.05 considered statistically significant. We evaluated the risk using the allele comparison model (T vs. C), dominant model [(TT + CT) vs. CC], recessive model [TT vs. (CT + CC)], heterozygote model (TC vs. CC), and homozygote model (TT vs. CC). Stratified analyses by ethnicity were also carried out. Statistical heterogeneity among studies was evaluated using I2 statistics (ranges from 0% to 100%), λ2 tests, and P values. 14 The fixed effects model method was used, but when a significant Q test (P < 0.05) or I2 > 50% indicated the existence of heterogeneity among studies, the random effects model was applied. 15 Heterogeneity was also explored in the subgroup analysis with ethnic groups (Asian and White). Beggar’s funnel plots were drawn to estimate the potential publication bias, in which the standard error of log (OR) for each study was plotted against its log (OR). Sensitivity analysis was performed to assess the stability of the results. Whether the funnel plot was symmetrical or not was assessed using Egger’s test, 16 and publication bias was considered to exist if P < 0.05. Trial sequential analysis (TSA) was performed using TSA Version 0.9.5.10 Beta (www.ctu.dk/tsa, Centre for Clinical Intervention Research, Copenhagen, Denmark). We applied a two-sided test, type I error of 5%, and power of 80% and assumed a 10% relative risk reduction for MTRR rs1532268 polymorphism. All statistical tests for this meta-analysis were performed with Review Manager 5.4 (Cochrane Collaboration, London, UK) and STATA 12.0 (StataCorp LLC, College Station, TX, USA).
Results
Characteristics of included studies
Sixty-two potentially relevant papers were identified based on the search strategy. Five studies with 2800 cases and 2679 controls were finally included in the meta-analysis (Figure S1). The characteristics of the included studies were listed in Table 1. These five studies included four focused on Asian populations8,11–13 and one in a White population. 9 In one study, the numbers of patients with CT and TT genotypes were not given individually. Patients with GC were confirmed pathologically.
Main characteristics of the studies included in this meta-analysis.

Quantitative analysis
The association between MTRR rs1532268 polymorphism and GC risk was investigated in the five included studies. The pooled data indicated a significant association between MTRR rs1532268 polymorphism and the risk of GC. Several models were applied, including the allele comparison model (OR = 1.14, 95% CI = 1.01–1.29, Pheterogeneity = 0.56, P = 0.04, Figure 1), dominant model (OR = 1.14, 95% CI =1.00–1.30, Pheterogeneity = 0.64, P =0.04, Figure 2), recessive model (OR = 1.14, 95% CI = 0.81–1.60, Pheterogeneity = 0.68, Figure 3), heterozygote model (OR = 1.16, 95% CI = 1.00–1.35, Pheterogeneity = 0.56, P = 0.06, Figure 4), and homozygous model (OR = 1.24, 95% CI = 0.88–1.76, Pheterogeneity = 0.73, Figure 5). Subgroup analyses were carried out according to ethnicity; only one study was in the White population, and the data were pooled for Asian populations. Increased risks were not found in Asians (allele model, OR = 1.13, 95% CI = 0.97–1.31, Pheterogeneity = 0.37, Figure 1; dominant model, OR = 1.11, 95% CI = 0.96–1.28, Pheterogeneity = 0.64, Figure 2; recessive model, OR = 1.28, 95% CI = 0.79–2.07, Pheterogeneity = 0.59, Figure 3; heterozygote model, OR = 1.11, 95% CI = 0.94–1.32, Pheterogeneity = 0.59, Figure 4; and homozygous model, OR = 1.31, 95% CI = 0.81–2.12, Pheterogeneity = 0.55, Figure 5).
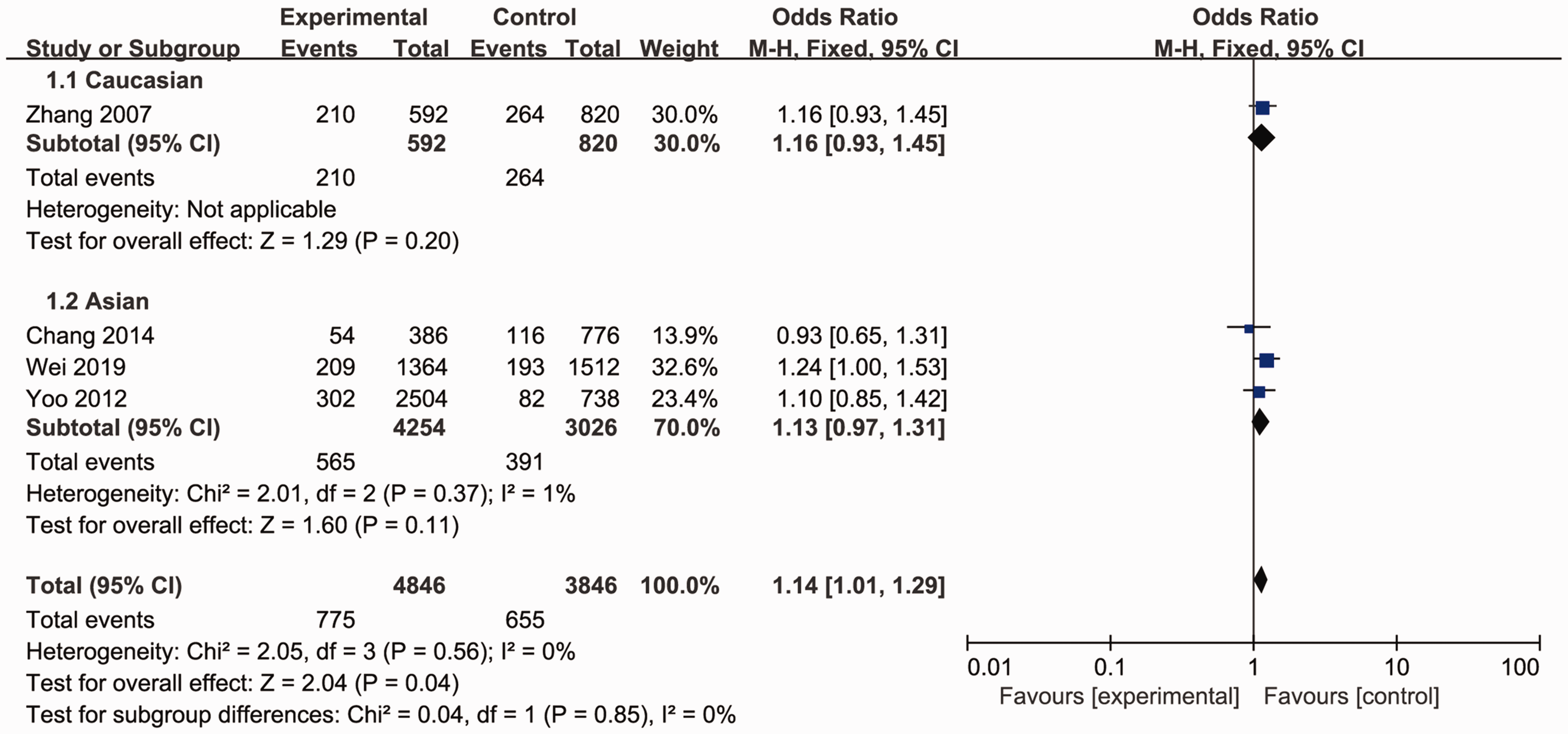
Forest plot for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the allele comparison model.
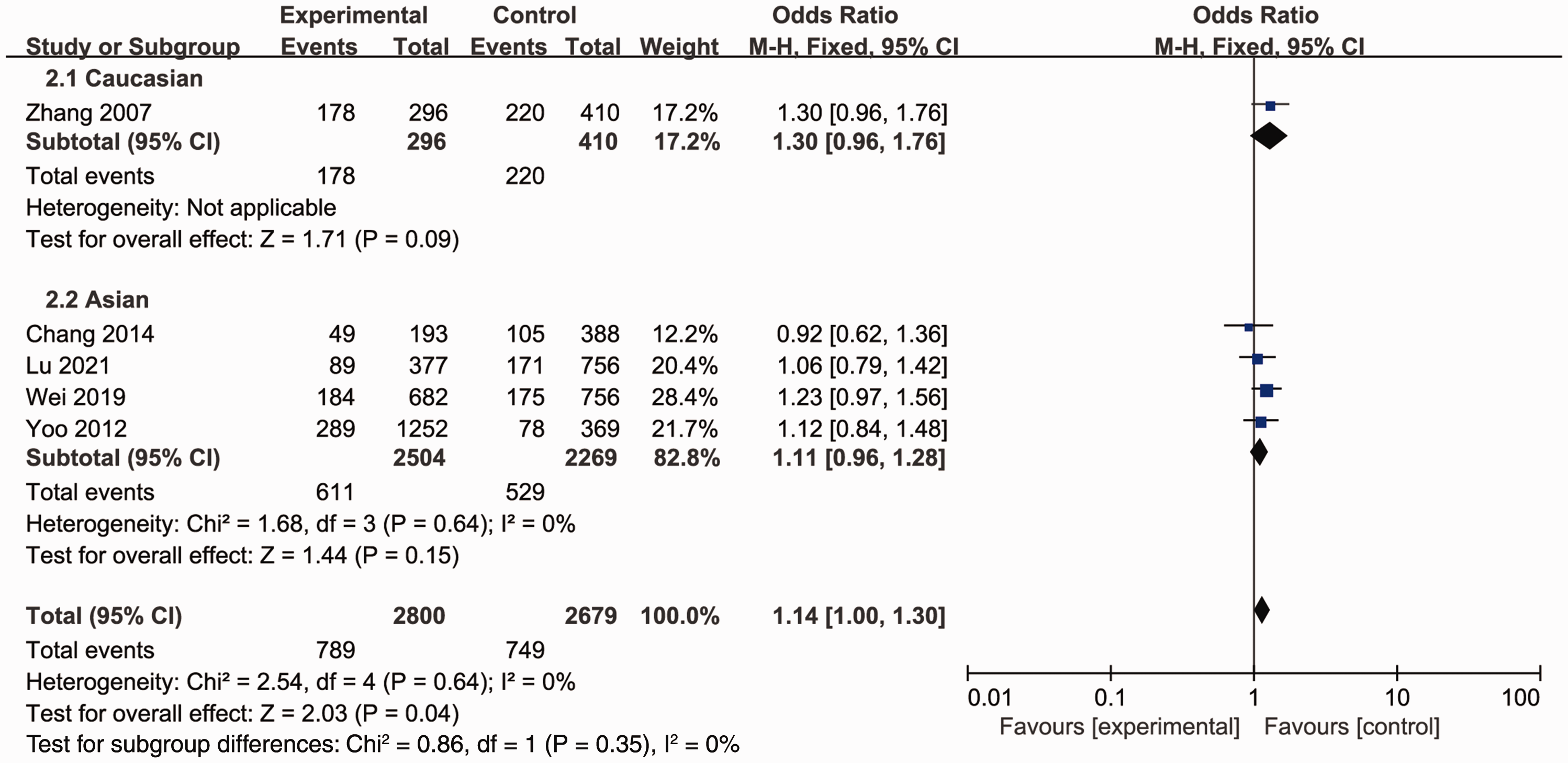
Forest plot for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the dominant comparison model.

Forest plot for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the recessive comparison model.

Forest plot for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the heterozygote comparison model.

Forest plot for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the homozygous comparison model.
Sensitivity analysis
One-way sensitivity analyses were performed to assess the stability of the pooled results. Each study included in the meta-analysis was removed one at a time to observe the influence of the individual data on the pooled ORs. No single study affected the pooled OR value, suggesting that the results of this meta-analysis were stable (Figure 6).

One-way sensitivity analysis of the pooled ORs and 95% CIs for the dominant comparison model, with the listed study omitted.
Publication bias
The relative symmetrical shape of funnel plots indicated minimal publication bias (Figure 7; dominant model). Beggar’s funnel plot and Egger’s test were performed to assess the publication bias. The statistical results also showed there was no significant publication bias (P = 0.806 for the dominant comparison model). Egger’s test was also performed to further assess the publication bias (P = 0.633 for the dominant comparison model).

Funnel plot for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the dominant comparison model.
TSA
Because of the low number of studies included in this meta-analysis, TSA was performed to provide accurate results for the study. The results were confirmed after adjusting for type 1 and 2 errors in TSA. The results indicated that the outcomes of this meta-analysis were reliable (Figure 8), as the Z-curve (blue line) crossed the conventional threshold (horizontal dark red line) and the trial sequential boundary (red inclined line).

Trial sequential analysis for the meta-analysis of the association between MTRR rs1532268 polymorphism and gastric cancer risk under the dominant comparison model. The Z-curve (blue line) crosses the conventional threshold (horizontal dark red line) and the trial sequential boundary (red inclined line).
Discussion
MTRR is a key enzyme in homocysteine/methionine metabolism, which are essential amino acids required for protein synthesis and one-carbon metabolism.11,13 Variations in the MTRR gene have been extensively analyzed in several tumors, including meningioma, breast cancer, and GC.8,17,18 In 2003, MTRR rs1801394 polymorphism was first reported to influence the risk of GC. 19 However, a lack of an association between MTRR rs1801394 polymorphism and GC risk was also reported,9,20 which was validated by pooled data from a meta-analysis.9,20 In 2007, a borderline increased GC risk was found to be associated with the MTRR rs1532268 polymorphism.9,20 The researchers also indicated that the polymorphism contributed to GC,9,20 whereas other studies reported contrasting results.8,12,13 We performed this meta-analysis to combine the eligible studies to date and obtain a more precise estimate of the relationship between MTRR rs1532268 polymorphism and GC risk. The present meta-analysis found that MTRR rs1532268 polymorphism was significantly associated with GC risk. In the allele comparison model, MTRR-524C>T (rs1532268 polymorphism) increased GC morbidity by approximately 14%, and a similar result was obtained for the dominant model but not the recessive, heterozygote, and homozygous models. Subgroup analysis did not identify an association between MTRR-524C>T and GC risk in Asian or White populations. More data are needed to determine the relationship between rs1532268 polymorphism and GC risk in Whites.
The MTRR rs1532268 polymorphism may be a biomarker for GC risk. Because this enzyme is involved in folate metabolism, MTRR mutation disrupts folate metabolism. Researchers have indicated that high riboflavin consumption significantly decreases the risk of GC in TC and TT carriers compared with that in CC carriers,8,21 and it was reported that TT or CT/TT genotypes were significantly associated with poorer overall survival. 10 This provided a clue that riboflavin consumption may improve patient survival.
Although this was the first meta-analysis to evaluate the association of GC risk with MTRR rs1532268 polymorphism, there were some limitations. First, five studies with 2800 cases and 2679 controls were included in this meta-analysis, and the population was relatively small. More studies investigating the relationship between rs1532268 polymorphism and the risk of GC are needed to confirm the results. Second, subgroup analyses were only performed for Asian and White populations, and other ethnicities could not be stratified because of a limited number of studies. In addition, age, sex, diet, Helicobacter pylori infection, alcohol, smoking habits, and other potential related factors were not evaluated in this study, and other well-known prognostic factors, such as staging and surgical and histologic findings, were not reported because of a lack of data. Third, the number of CT and TT genotypes was not described in one study, 8 and only four studies with 2423 patients with GC and 1923 controls were included in the allele comparison model, recessive model, heterozygote model, and homozygous model. Forth, studies with positive association results are often easier to publish, and although no publication bias was observed in this meta-analyses, it may exist in the studies included. Finally, different polymorphisms of the MTRR gene may alter the OR values. Given these limitations, our conclusions should be interpreted cautiously.
Conclusion
Our meta-analysis provides evidence that MTRR rs1532268 polymorphism increases the risk of GC. Because of the limitations to this meta-analysis, well-designed and larger trials should be conducted to re-evaluate the results.
Supplemental Material
sj-jpg-1-imr-10.1177_03000605221097486 - Supplemental material for MTRR rs1532268 polymorphism and gastric cancer risk: evidence from a meta-analysis
Supplemental material, sj-jpg-1-imr-10.1177_03000605221097486 for MTRR rs1532268 polymorphism and gastric cancer risk: evidence from a meta-analysis by Guping Zhong, Xiaojin Luo, Ji Li, Yuanhang Liao, Guan Gui and Jianwen Sheng in Journal of International Medical Research
Footnotes
Author contributions
Project development: Jianwen Sheng; Data collection or management: Guping Zhong, Xiaojin Luo, Ji Li, Yuanhang Liao, and Guan Gui; Data analysis and interpretation: Guping Zhong and Xiaojin Luo; Manuscript writing: Guping Zhong; Manuscript editing: Jianwen Sheng; Study supervision: Jianwen Sheng. All authors have read and approved the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
