Abstract
Objective
To investigate the effects of piperlongumine (PL) and vitamin C (VC) on signal transducer and activator of transcription 3 (STAT3) signalling in gastric cancer cell lines.
Methods
In vivo tumour xenograft anticancer assays were undertaken to confirm the anticancer activity of PL. Cell viability, flow cytometry and Western blot assays were undertaken to evaluate the anticancer effects of PL, VC and combinations of PL and VC in AGS and KATO III cells.
Results
Both PL and VC induced apoptosis and inhibited cell proliferation in AGS and KATO III cells. These effects were dependent on reactive oxygen species (ROS). PL effectively suppressed STAT3 activation while VC caused abnormal activation of STAT3. The combination of PL and VC exhibited a stronger apoptotic effect compared with either agent alone. PL reversed the abnormal activation of STAT3 by VC, which could be a key to their synergistic effect.
Conclusions
PL combined with VC exhibited a stronger anticancer effect by regulating the ROS–STAT3 pathway, suggesting that this combination might be a potential adjuvant therapy for gastric cancer.
Keywords
Introduction
Gastric cancer (GC) has become a serious cancer with high rates of morbidity and mortality. 1 Surgical resection is a common option for early GC, while advanced GC requires radiotherapy, chemotherapy and targeted therapy. 2 Increasing numbers of patients are diagnosed with advanced GC, but because targeted drugs are expensive, chemotherapy remains a routine treatment for GC. Unfortunately, nonselective killing and drug resistance have become the major problems associated with current chemotherapy, leading to severe side-effects and adverse reactions. 3 Hence, improving the current chemotherapy status through combined application of several drugs has become a promising strategy. Fluorouracil (5-FU), oxaliplatin, irinotecan and paclitaxel have been used as first-line chemotherapy drugs against GC.4,5 Although the combined application of these drugs is a routine treatment strategy, multidrug resistance has become a serious obstacle. 6
Vitamin C (VC) has gradually shown its anticancer potential over the past few decades.7,8 Recent research has demonstrated that VC targets three vulnerabilities of cancer cells, including the redox imbalance, epigenetic reprogramming and oxygen-sensing regulation. 8 Current clinical studies showed that intravenous injection of high-dose VC significantly enhanced the efficacy of several GC chemotherapy drugs, such as 5-FU, oxaliplatin, irinotecan and paclitaxel.9,10 These exciting findings indicate that VC has the potential to be used as an adjuvant agent in future chemotherapy regimens. Although VC cannot fulfil the anticancer requirements when used alone, this unique vitamin exhibits good adjunctive effects when combined with other anticancer agents.11,12 Considering the safety and economic advantages of VC, finding a solution to improve its application in cancer therapy could be a valuable approach.
Piperlongumine (PL), a natural alkaloid, was originally isolated from Piper longum L. 13 Previous research has demonstrated that PL is a potent anticancer compound with reliable selectivity. 14 PL promotes reactive oxygen species (ROS) production and induces apoptosis of different types of cancer cells. 15 Its mechanism of action for killing cells involves diverse signalling pathways, including mitogen-activated protein kinase, phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin, nuclear factor kappa B, glutathione S-transferase P1 and thioredoxin reductase-1.15,16 However, poor water solubility and organ toxicity limit the prospects of PL as a single anticancer agent.13,17 Recent studies demonstrated that PL enhanced the sensitivity of a variety of chemotherapeutic drugs, including cisplatin, paclitaxel and doxorubicin (DOX) in different types of cancer cells.18,19 Meanwhile, PL has been shown to be a natural inhibitor of STAT3, 20 indicating that PL may improve drug resistance when combined with other chemotherapeutic drugs. Today, adjuvant chemotherapy has become a critical strategy for clinical cancer therapy. 5 Given the good anti-cancer prospects of PL and the good safety of VC, it is important to study whether the combination of PL and VC could produce a stronger anticancer effect and even improve drug resistance.
This present study aimed to confirm the anticancer activity of PL in vivo and then to evaluate the effects of using PL and VC alone or in combination on cell proliferation and apoptosis in two GC cell lines in order to determine if the combination of VC and PL has the potential to be useful in future clinical chemotherapy.
Materials and methods
Reagent preparation
Piperlongumine was purchased from ApexBio (Houston, TX, USA) and was dissolved in dimethyl sulfoxide (DMSO). VC and N-acetyl-L-cysteine (NAC) were purchased from Sigma-Aldrich (St Louis, MO, USA) and were dissolved in deionized H2O.
Cell lines and culture
Human gastric cancer cell lines AGS and KATO III were purchased from the American Type Culture Collection (Manassas, VA, USA). Both cell lines were cultured in Dulbecco’s Modified Eagle’s Medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Thermo Fisher Scientific) and 100 U/ml penicillin and streptomycin (Thermo Fisher Scientific). Both cell lines were cultured in a 37 °C incubator with 5% CO2.
Cell viability assay
Both cell lines were inoculated into 96-well plates at a density of 3 × 103 cells/well. After overnight incubation, cells were exposed to a concentration of the agent, either alone or in combination, for 24 h, 48 h and 72 h. Then the cells were treated with a working solution of 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich) for 4 h. The absorbance of each well was measured at 570 nm using an Epoch microplate reader (BioTek, Winooski, VT, USA).
Cell apoptosis assay
An annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) double staining kit (BD Biosciences, San Jose, CA, USA) was used to identify apoptotic and necrotic cells. Both cell lines were exposed to different concentrations of PL and VC, either alone or in combination, for 48 h. After incubation, cells were collected and resuspended in 500 μl binding buffer and then stained with 5 μl of annexin V-FITC and 5 μl of PI in the dark for 15 min. The proportions of apoptotic and necrotic cells were detected using an Accuri C6 flow cytometer (BD Biosciences). Cells showing up as annexin V–/PI– were identified as healthy cells, annexin V–/PI+ were identified as necrotic cells, annexin V+/PI– were identified as early apoptotic cells and annexin V+/PI+ were identified as late apoptotic cells.
Western blot analysis
Human gastric cancer cell lines AGS and KATO III (3 × 106 cells) were homogenized under the action of protein lysate buffer and then centrifuged at 13 800
In vivo antitumour study
Five-week-old male nude mice (weight range: 16–18 g; HuaFuKang Bioscience, Beijing, China) were housed in an animal barrier facility at Xi’an Medical University. All animals were housed in individually ventilated cages under a 12-h light/12-h dark cycle with free access to food and water. AGS cells (5 × 106) were injected subcutaneously into the right side of the nude mice. When the tumours reached a volume of 50 mm3, the nude mice were randomly divided into two groups and injected intraperitoneally (i.p.) with the vehicle (10 mM PBS pH 7.4) or 4 mg/kg PL dissolved in 10 mM PBS (pH 7.4) once every day for 9 days. Tumours were measured daily with a calliper and the volume was determined using the formula of (length ×width 2 )/2. At the end of the therapy, all nude mice were executed and each tumour was excised and weighed. All animal study procedures were performed in accordance with protocols approved by Xi’an Medical University Medical Ethics Review Committee. All animal experiments complied with the ARRIVE guidelines and were carried out in accordance with the UK Animals (Scientific Procedures) Act, 1986, and associated guidelines from the EU Directive 2010/63/EU for animal experiments.
Statistical analyses
All statistical analyses were performed using GraphPad Prism 5.0 (Graphpad Software Inc., San Diego, CA, USA). Data are expressed as the mean ± SD. One-way analysis of variance was used to analyse the significance of any differences between multiple treatment groups. A P-value <0.05 was considered statistically significant.
Results
In vivo antitumour studies were undertaken in nude mice to determine the effectiveness of PL. Twelve male nude mice with AGS tumour xenografts were treated with i.p. injections of vehicle or 4 mg/kg PL. After 9 days, all nude mice were sacrificed and each tumour was removed and weighed. PL significantly decreased the tumour growth rates as demonstrated by tumour volume compared with the vehicle control group (P < 0.001) (Figures 1a and 1b). The tumour weight on day 9 of the vehicle control group was significantly higher than that of the PL group (P < 0.001) (Figure 1c). There were no significant differences in the changes in body weight between the two groups during treatment (Figure 1d), which indicated that PL had no significant toxicity.
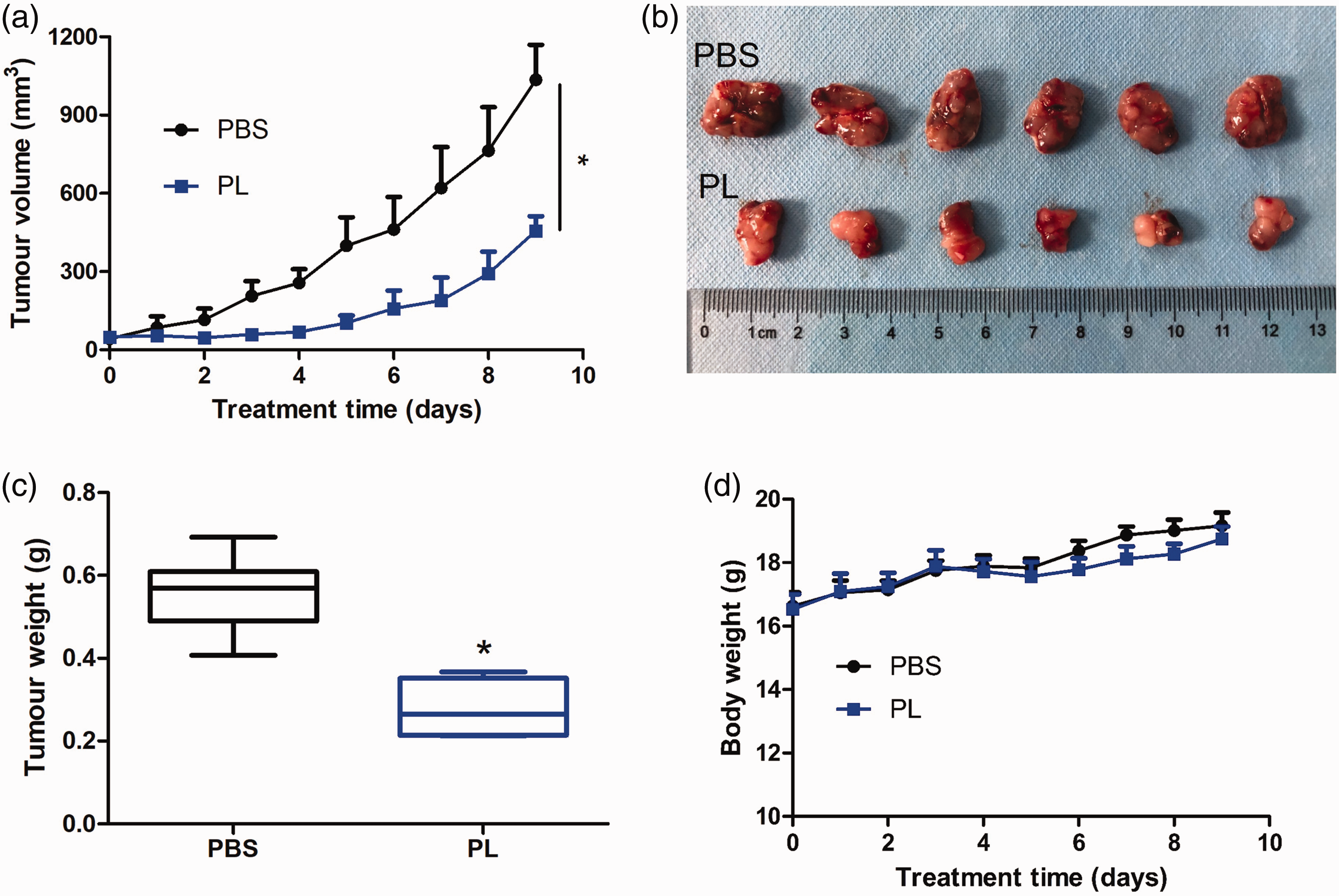
Evidence that piperlongumine (PL) suppressed AGS xenograft tumour growth in vivo. A total of 5 × 106 AGS cells were injected in the right flank of male nude mice and when the tumour volume reached 50 mm3, the nude mice were randomly divided into two groups (n = 6 per group). The nude mice were treated with 10 mM phosphate-buffered saline (PBS; pH 7.4) or PL dissolved in 10 mM PBS (pH 7.4) via intraperitoneal injections once every day for 9 days. (a) Tumours were measured using callipers each day and the volumes were calculated using the formula of (length × width 2 )/2. Data presented as mean ± SD. (b) On day 9 after treatment, all nude mice were sacrificed and the tumours excised. (c) Tumour weights on day 9. The central black horizontal line for each group is the median. The extremities of the boxes are the 25th and 75th percentiles and the error bars represent the minimum and maximum outliers. (d) Body weight of the treated nude mice over time. Data presented as mean ± SD. *P < 0.001 compared with the control group; analysis of variance. The colour version of this figure is available at: http://imr.sagepub.com.
The inhibitory effects of PL and VC were measured in two human GC cell lines, AGS and KATO III, over three treatment durations and a range of concentrations. Both cancer cell lines were inhibited by PL in a dose- and time-dependent manner, with IC50 values of 6.49 μM (24 h), 4.44 μM (48 h) and 4.09 μM (72 h) in AGS cells; and IC50 values of 8.58 μM (24 h), 6.25 μM (48 h) and 6.29 μM (72 h) in KATO III cells (Figure 2a). VC inhibited the growth of both cancer cell lines in a dose-dependent manner, with IC50 values of 1.75 mM (24 h), 1.91 mM (48 h) and 1.83 mM (72 h) in AGS cells; and IC50 values of 1.41 mM (24 h), 1.40 mM (48 h) and 1.30 mM (72 h) in KATO III cells (Figure 2b).

Piperlongumine (PL) and vitamin C (VC) inhibited the proliferation of two gastric cancer cell lines in vitro: (a) AGS and KATO III cells were treated with a range of concentrations of PL for 24 h, 48 h and 72 h. The cell viability was determined using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay; (b) AGS and KATO III cells were treated with a range of concentrations of VC for 24 h, 48 h and 72 h. The cell viability was determined using an MTT assay. A control group (0) was treated with the vehicle dimethyl sulfoxide. Data presented as the mean of triplicate experiments. The colour version of this figure is available at: http://imr.sagepub.com.
As shown in Figure 3, both PL and VC induced the apoptosis of AGS and KATO III cells in a dose-dependent manner. AGS cells appeared to be more sensitive than KATO III cells to PL, while the sensitivity of both cell lines to VC was similar. The data demonstrated that PL induced apoptosis more strongly than VC, which was consistent with the MTT results.

Piperlongumine (PL) and vitamin C (VC) induced apoptosis in two gastric cancer cell lines in vitro. (a) AGS and KATO III cells were treated with a range of concentrations of PL for 48 h and then stained with annexin V/propidium iodide (PI) for the flow cytometric analysis of the levels of apoptosis; (b) the proportion of cells that were apoptotic after treatment with PL. Data presented as mean ± SD; (c) AGS and KATO III cells were treated with a range of concentrations of VC for 48 h and then stained with annexin V/PI for the flow cytometric analysis of the levels of apoptosis; (d) the proportion of cells that were apoptotic after treatment with VC. Data presented as mean ± SD; *P < 0.05, **P < 0.01, ***P < 0.001 compared with the control group (treated with the vehicle dimethyl sulfoxide); analysis of variance. The colour version of this figure is available at: http://imr.sagepub.com.
In order to determine the mechanism by which PL and VC induced apoptosis, Western blot analyses were undertaken to measure the influence of PL and VC on the STAT3 pathway. As shown in Figure 4, PL decreased STAT3 phosphorylation at different doses in both GC cell lines, while VC showed the opposite effect. PL inhibited the levels of survivin in KATO III cells, but upregulated the levels of survivin in AGS cells. VC increased survivin levels in AGS cells, but decreased survivin levels in KATO III cells.

Piperlongumine (PL) and vitamin C (VC) regulated signal transducer and activator of transcription 3 (STAT3) signalling in opposite ways in two gastric cancer cell lines in vitro: (a) AGS and KATO III cells were treated with a range of concentrations of PL for 24 h and then they were subjected to Western blot analysis to measure the protein levels of p-STAT3, STAT3 and survivin. β-actin served as an internal control; (b) the relative levels of protein compared with β-actin after treatment with PL. Data presented as mean ± SD; (c) AGS and KATO III cells were treated with a range of concentrations of VC for 24 h and then they were subjected to Western blot analysis to measure the protein levels of p-STAT3, STAT3 and survivin. β-actin served as an internal control; (d) the relative levels of protein compared with β-actin after treatment with VC. Data presented as mean ± SD; *P < 0.05, **P < 0.01, ***P < 0.001 compared with the control group (treated with the vehicle dimethyl sulfoxide); analysis of variance.
The role of ROS in the apoptosis-inducing effects of PL and VC was determined using preincubation of AGS and KATO III cells with 5 mM NAC. As shown in Figure 5, high doses of PL and VC induced apoptosis, but preincubation with NAC inhibited this apoptosis-inducing effect.
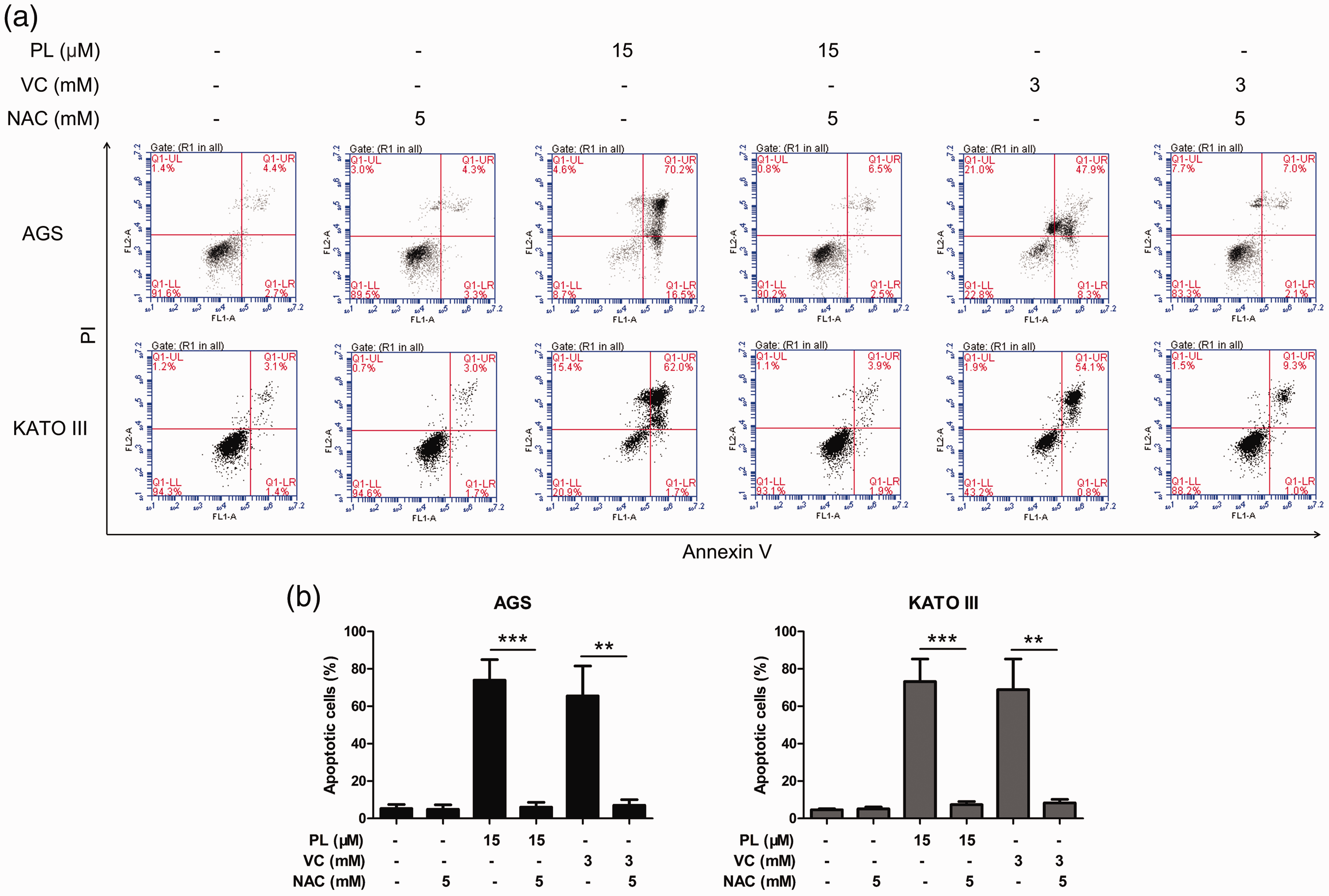
Piperlongumine (PL) and vitamin C (VC) induced reactive oxygen species-dependent apoptosis in two gastric cancer cell lines in vitro: (a) AGS and KATO III cells were treated with 15 µM PL or 3 mM VC with/without 5 mM N-acetyl-L-cysteine (NAC) preincubation for 48 h and then stained with annexin V/propidium iodide (PI) for the flow cytometric analysis of the levels of apoptosis; (b) the proportion of cells that were apoptotic after treatment with PL, VC and NAC. Data presented as mean ± SD. *P < 0.05, **P < 0.01, ***P < 0.001; analysis of variance. The colour version of this figure is available at: http://imr.sagepub.com.
To get a better understanding of the regulatory role of ROS in STAT3 signalling, protein levels of p-STAT3 and STAT3 were measured using Western blot analyses. As shown in Figure 6, a high dose of PL significantly inhibited STAT3 phosphorylation compared with the control group (P < 0.001). However, this inhibitory effect disappeared after NAC preincubation. The presence or absence of NAC did not affect the regulatory effect of VC on STAT3 phosphorylation.
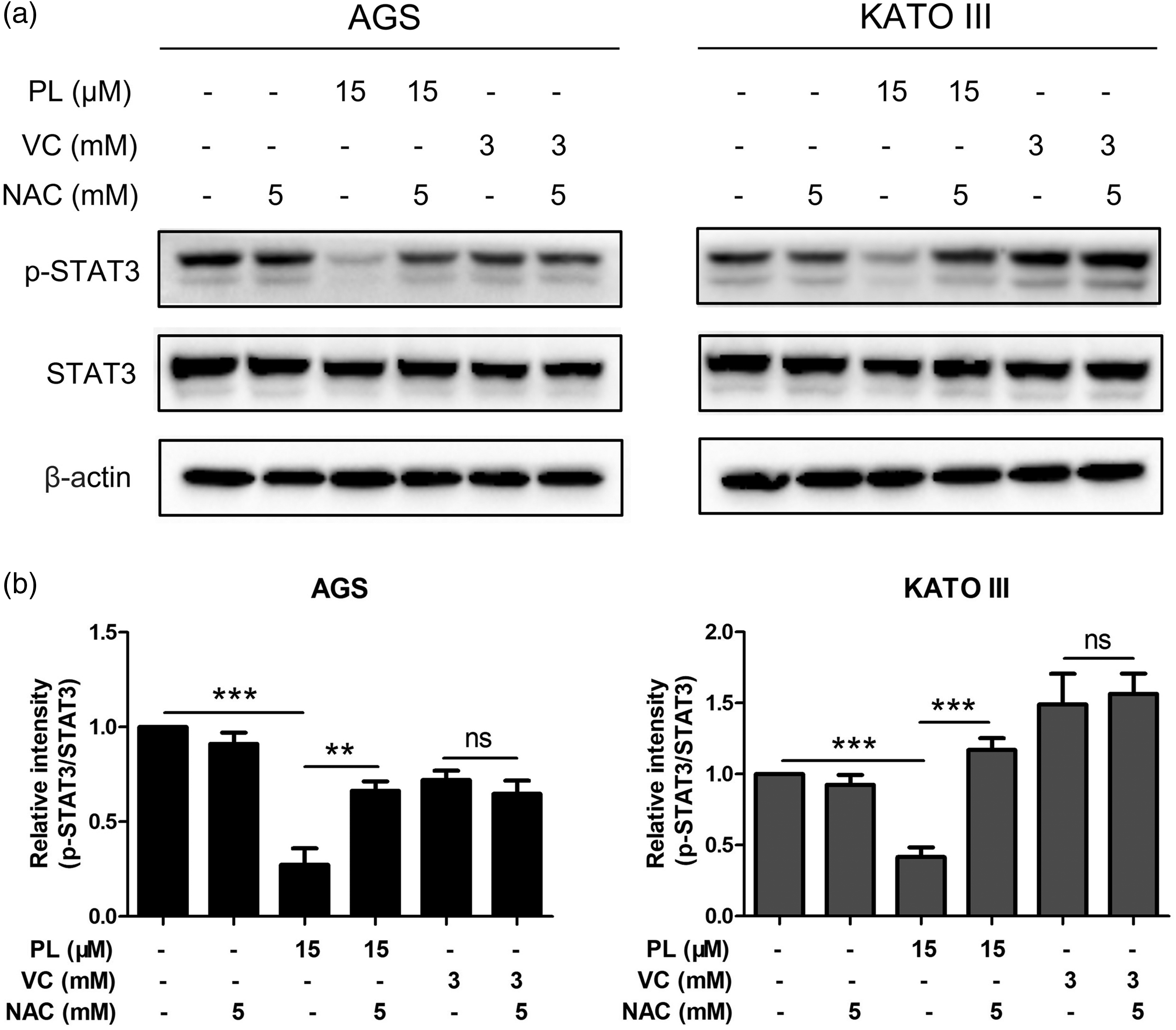
The regulation of signal transducer and activator of transcription 3 (STAT3) signalling by piperlongumine (PL) was dependent upon reactive oxygen species but that of vitamin C (VC) was not in two gastric cancer cell lines in vitro: (a) AGS and KATO III cells were treated with 15 µM PL or 3 mM VC with/without 5 mM N-acetyl-L-cysteine (NAC) preincubation for 48 h and then they were subjected to Western blot analysis to measure the protein levels of p-STAT3 and STAT3. β-actin served as an internal control; (b) the relative levels of protein compared with β-actin after treatment. Data presented as mean ± SD; *P < 0.05, **P < 0.01, ***P < 0.001; ns, not significant; analysis of variance.
To investigate whether PL and VC exhibited a synergistic effect on AGS and KATO III cell apoptosis, the cells were treated with a combination of doses of PL (5 µM, 10 µM) and VC (1 mM) and then subjected to flow cytometry to measure the rate of apoptosis. As shown in Figure 7, the combined use of 5 µM PL and 1 mM VC in both cell lines significantly increased the rate of apoptosis compared with either agent used alone (P < 0.05). In KATO III cells, the combination of 10 µM PL and 1 mM VC significantly increased the rate of apoptosis compared with 10 µM PL alone (P < 0.01). However, no such effect was observed in AGS cells.

Piperlongumine (PL) and vitamin C (VC) synergistically induced apoptosis in two gastric cancer cell lines in vitro: (a) AGS and KATO III cells were treated with PL (5, 10 µM) and VC (1 mM) alone or in combination for 48 h and then stained with annexin V/propidium iodide (PI) for the flow cytometric analysis of the levels of apoptosis; (b) the proportion of cells that were apoptotic after treatment. Data presented as mean ± SD. *P < 0.05, **P < 0.01, ***P < 0.001; ns, not significant; analysis of variance. The colour version of this figure is available at: http://imr.sagepub.com.
Western blot analysis was used to measure the levels of STAT3 pathway-related proteins, p-STAT3, STAT3 and survivin, in AGS and KATO III cells treated with PL (5 µM, 10 µM) and VC (1 mM) alone or in combination for 24 h. As shown in Figure 8, the combination of the two agents had a synergistic inhibitory effect on the levels of p-STAT3 in both cancer cell lines compared with either agent used alone. Similar to the previous apoptosis results, the combination of 10 µM PL and 1 mM VC in AGS cells did not show a greater inhibitory effect than the combination of 5 µM PL and 1 mM VC. The regulation of survivin by PL and VC was not exactly the same as that of p-STAT3. In AGS cells, only 10 µM PL and 1 mM VC synergistically inhibited the levels of survivin. In KATO III cells, the two combination groups showed a stronger inhibitory effect than PL alone, but no significant effect was observed compared with VC alone.
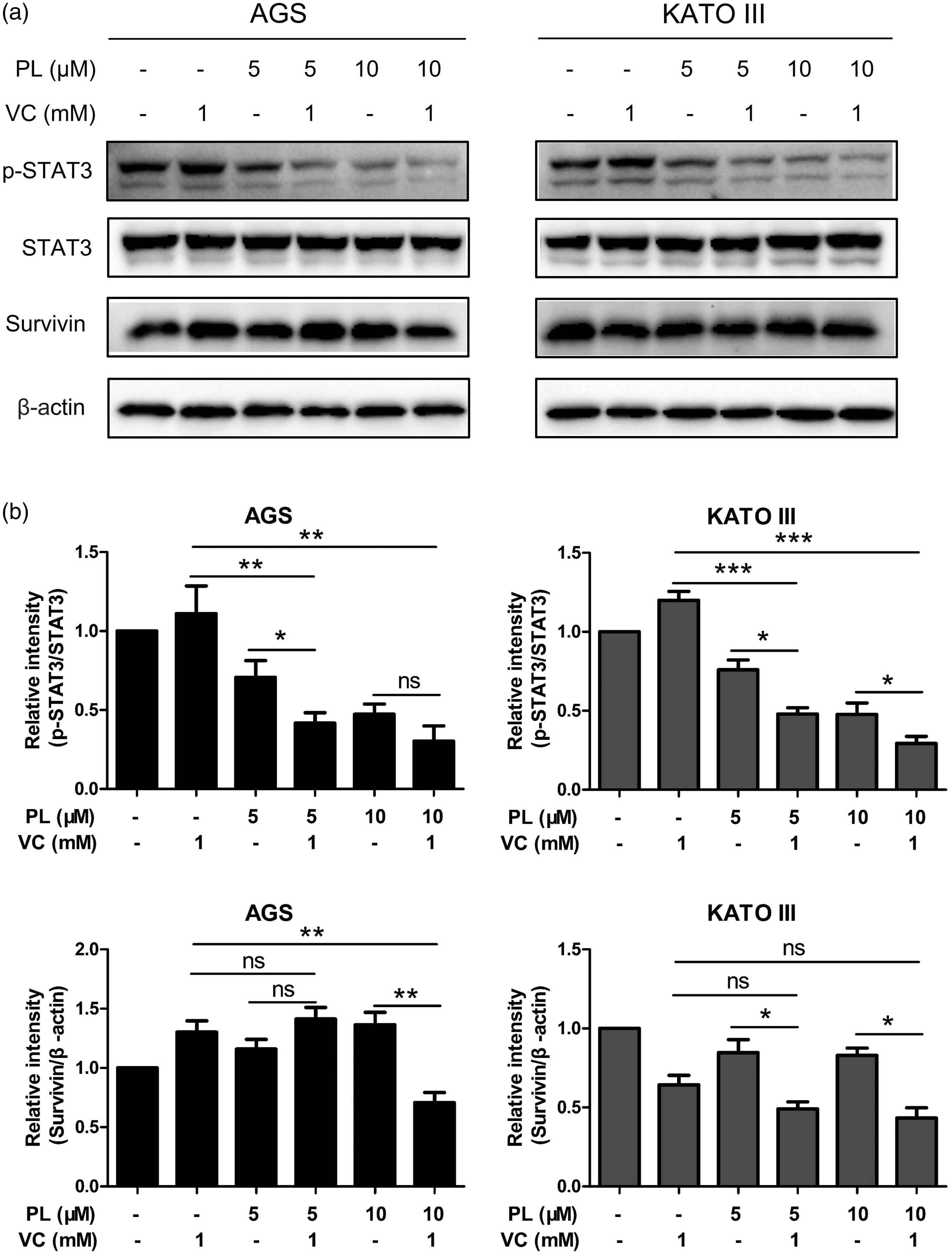
Piperlongumine (PL) and vitamin C (VC) synergistically inhibited signal transducer and activator of transcription 3 (STAT3) signalling in two gastric cancer cell lines in vitro: (a) AGS and KATO III cells were treated with PL (5, 10 µM) and VC (1 mM) alone or in combination for 24 h and then they were subjected to Western blot analysis to measure the protein levels of p-STAT3, STAT3 and survivin. β-actin served as an internal control; (b) the relative levels of protein compared with β-actin after treatment. Data presented as mean ± SD; *P < 0.05, **P < 0.01, ***P < 0.001; ns, not significant; analysis of variance.
Discussion
The current status of chemotherapy for GC is not optimistic. Drug resistance leads to recurrence and a low 5-year survival rate, 21 which needs to be improved urgently. Although recent clinical research has found that VC cooperates with a variety of first-line drugs against cancer, it is hard to avoid drug resistance. 22 Therefore, a specific agent, such as PL, might be needed to overcome this problem and help VC become a more effective anticancer treatment. This current study focused on the anticancer effects and mechanisms of action of PL combined with VC in AGS and KATO III cells. The current data suggest that these two agents synergistically inhibited GC cell proliferation. The regulation of the ROS–STAT3 pathway by PL could be the key mechanism of action of this combination.
Reactive oxygen species in cancer cells play a central role in regulating and inducing apoptosis, thereby modulating cancer cell proliferation, survival and drug resistance. 23 Recent studies reported that both PL and VC generate large amounts of ROS that cancer cells cannot tolerate, thereby inducing cancer cell apoptosis.24,25 In the present study, PL and VC effectively induced apoptosis of GC cells through oxidative stress. Both 15 µM PL and 3 mM VC caused more than 60% apoptosis in two GC cell lines. However, these apoptosis-inducing effects were completely inhibited after being preincubated with 5 mM NAC, which is an effective antioxidant. These current findings suggest that the apoptotic effect induced by PL and VC both depended on ROS, which was consistent with previous reports.25,26 In addition, Western blot analyses showed that PL and VC exhibited different mechanisms of action to induce apoptosis of GC cells. As shown in Figure 4, 15 µM PL effectively inhibited the phosphorylation of STAT3 in both GC cell lines. This effect was abrogated by preincubation with 5 mM NAC (Figure 6). Preincubation with NAC did not change the effects of VC on the phosphorylation of STAT3. Therefore, these current results demonstrated that ROS induced by PL could further inhibit the activation of STAT3, while VC showed no such effects.
It was reported that activated ROS production played a key role in the suppression of JAK2/STAT3 signalling. 27 In this current study, the effect of PL in inhibiting the activation of STAT3 was dependent on ROS, which was consistent with previous studies. 20 However, the abnormal activation of STAT3 by VC did not depend on ROS, which suggested that PL and VC regulated the STAT3 signalling pathway in very different ways. Taken together, both PL and VC induced apoptosis of GC cells by generating ROS, however, only the PL-induced inhibition of STAT3 was beneficial for the anticancer effects.
The JAK/STAT signalling pathway plays an important role in the proliferation, survival and drug resistance of cancer cells. 28 STAT3 is a key member of the STAT family and has become an important target due to its important role in cancer regulation. 29 STAT3 is abnormally activated in a variety of solid tumours and non-solid tumours thereby leading to cancer deterioration.30,31 STAT3 is constitutively activated in many cancer types and such hyperactivation is associated with a poor clinical prognosis. 32 Previous research has reported that constitutive activation of STAT3 leads to cisplatin resistance in GC cells and disruption of STAT3 could re-sensitize GC cells to chemotherapy drugs. 33 Since activation of STAT3 is an essential feature of chemotherapeutic drug resistance, blocking the STAT3 pathway to improve the efficacy of chemotherapy has become a promising strategy. 34
Among the many small molecules that can inhibit the activation of STAT3, PL has attracted the attention of researchers due to its good effectiveness and safety. 16 PL has broad-spectrum anticancer activity while showing less toxicity to normal cells. 35 In addition, the effective inhibition of STAT3 activation by PL suggests that it has the potential to improve chemotherapy resistance. 36 In the present study, PL and VC showed opposite regulatory effects on p-STAT3. PL effectively inhibited the activation of STAT3 in both GC cells, which was consistent with previous studies.37,38
However, VC causing activation of STAT3 indicates that it is likely to cause drug resistance when combined with other chemotherapy drugs. 35 Although having the opposite regulatory effects on p-STAT3, the combination of the two agents showed a synergistic inhibitory effect. Flow cytometric analysis demonstrated that the combination of the two agents induced a stronger apoptotic effect compared with either of the single-agent groups. Western blot analysis demonstrated that PL reversed the adverse effect of VC and together the two agents showed a stronger inhibitory effect on p-STAT3 in both GC cell lines.
Previous research reported that PL could reverse the abnormal activation of STAT3 by DOX, 19 which was consistent with these current findings. One theory is that the combined application of PL and VC creates some synergistic effects in which PL inactivates STAT3 through direct binding while VC enhances the bioavailability of PL. 39 Then these two agents work together to inhibit the STAT3 pathway, which in turn causes more ROS production thereby achieving a stronger anticancer effect. These current data indicated that PL and VC may be used together as a promising adjuvant to enhance the anticancer effects of chemotherapeutics, and more importantly, to overcome their drug resistance.
Unexpectedly, the regulation of survivin by the two agents was not similar to that of p-STAT3. PL increased the levels of survivin in AGS cells, while VC reduced the levels of survivin in KATO III cells. Nevertheless, 10 µM PL and 1 mM VC showed a synergistic inhibitory effect on survivin in both cell lines. These data indicated that although survivin can be regulated by other pathways, high-dose PL combined with VC could effectively reduce survivin levels.
In conclusion, PL and VC showed antiproliferative properties against GC cells and this effect was significantly enhanced when the two agents were combined. PL promoted ROS as well as reversing the abnormal activation of STAT3 caused by VC, which could be the key mechanism of action of this combination. This would be expected to overcome chemotherapy resistance. Therefore, the combination of PL and VC as an adjuvant provides a new strategy in future GC chemotherapy.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the China-Nepal Friendship Medical Research Laboratory of Professor Rajiv Kumar Jha at Xi’an Medical University (no. 18LJM07) and a Doctoral Research Start-up Fund Project of Xi'an Medical University (no. 2021DOC10).
