Abstract
Objective
To investigate the anticancer effect of nelfinavir (NFV) on human A549 cells.
Methods
The inhibitory effects of NFV on the proliferation of human A549 cells were assessed using a MTT assay. Apoptotic cells were observed by fluorescence microscopy following Hoechst 33342 staining. Apoptosis of A549 cells was assessed using Annexin-V/propidium iodide staining and flow cytometry. Expression levels of signal transducer and activator of transcription 3 (STAT3) and p-STAT3 were measured by western blotting. STAT3 RNA silencing was used to investigate the pro-apoptotic mechanism of NFV in A549 cells.
Results
NFV dose-dependently suppressed proliferation of human A549 cells and induced significant apoptosis. Western blotting showed that the antitumor function of NFV might be mediated by STAT3 inhibition. A549 cell apoptosis in response to 20 µM NFV was significantly increased following STAT3 silencing. NFV significantly impeded the expression of the anti-apoptotic proteins Bcl-xL and Bcl-2, by increased the expression of the pro-apoptotic protein Cle-PARP.
Conclusions
Our findings highlight STAT3 as a promising therapeutic target. NFV is a novel anti-cancer drug for the treatment of non-small-cell lung cancer.
Keywords
Introduction
In recent years, lung cancer has become one of the most common malignant tumors. Non-small-cell lung cancer (NSCLC) carries high morbidity and mortality. 1 Most patients with lung cancer have already reached advanced stages of disease when they are diagnosed, and thus are not good candidates for surgery. 2 Currently, the three most common treatment methods for advanced NSCLC are surgical resection, chemotherapy, and radiotherapy. 3 Although chemotherapy has some efficacy, traditional chemotherapy drugs such as platinum complexes are toxic to normal tissues, limiting their wide application in clinical practice. 4 Radiotherapy is typically only effective for small-cell lung cancer. 5 A variety of NSCLC-driving genes have been discovered, and targeted drugs and individualized treatments for these specific mutations have gradually entered clinical practice. 6 Some studies have shown that epidermal growth factor receptor tyrosine kinase inhibitors, echinoderm microtubule-associated protein-like 4-anaplastic lymphoma kinase, and vascular endothelial growth factor antagonists are effective in the treatment of NSCLC; these include crizotinib, alectinib, gefitinib, afatinib, and bevacizumab .7–9 However, acquired drug resistance is still a major challenge in patients receiving molecular targeted therapy. Therefore, the development of novel drugs to treat NSCLC is critically important.
The signal transducer and activator of transcription (STAT) protein family is a class of transcription factors. STAT3, an important member of the STAT family, has been recognized as an oncogenic protein. STAT3 does not directly cause cancer but induces the expression of certain key gene products by activating target genes, thus affecting the occurrence, deterioration, metastasis, or progression of tumors. 10 STAT3 is constitutively activated in 22% to 65% of NSCLCs. Several studies have shown that high expression of p-STAT3 is a predictor of poor prognosis in patients with NSCLC. 11
Nelfinavir (NFV) is a protease inhibitor used in patients with human immunodeficiency virus type-1 infection and acquired immunodeficiency syndrome. Its antitumor properties have been confirmed in various cancers, including ovarian, prostate, breast and pancreatic cancer cells. 12 Concurrent treatment with NFV, radiotherapy, and chemotherapy resulted in 5-year survival rate of 37.1% of patients with NSCLC. 13 However, the antitumor mechanisms of NFV remain unclear.
In this study, we explored the impact of NFV on the proliferation and apoptosis of human A549 lung cancer cells and the biological mechanism underlying NFV inhibition of STAT3 signaling. Our data suggest that NFV represents a new therapeutic drug for NSCLC.
Materials and methods
Reagents
NFV was obtained from Shanghai Beilang Biotechnology Co., Ltd. (Shanghai, China). NFV was dissolved in dimethylsulfoxide (DMSO). Antibodies against Bcl-xL, Bcl-2, Cle-poly (ADP-ribose) polymerase (PARP), as well as horseradish peroxidase-conjugated goat anti-rabbit IgG and goat anti-mouse IgG, were purchased from ythxbio (Beijing, China). Antibodies against p-STAT3 and STAT3 were purchased from Cell Signaling Technology (Danvers, MA, USA). DMSO and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Aladdin (Shanghai, China). The Annexin V-fluorescein isothiocyanate/propidium iodide (PI) double staining apoptosis detection kit was purchased from Shenzhen Kuyuan Biotechnology Co., Ltd. (Shenzhen, China). Hoechst 33342 was purchased from Shanghai Wanlei Biology Co., Ltd. (Shanghai, China) Anti-fluorescence quenching agent was purchased from Beyotime Institute of Biotechnology (Beijing, China). Small-interfering RNA (siRNA) targeting STAT3 and the scrambled control were purchased from RIBOBIO (Suzhou, China). Lipofectamine® RNAIMAX reagent was purchased from Thermo Fisher Scientific (Waltham, MA, USA).
Culture of A549 and H1224 cells
Human A549 and H1224 cells were purchased from the Cell Biology Institute of the Chinese Academy of Sciences (Shanghai, China). Cells were cultured in Dulbecco’s modified Eagle medium containing 10% fetal bovine serum and 1% penicillin/streptomycin under a humidified atmosphere containing 5% CO2 at 37°C.
Transfection
The following siRNA sequences were used: STAT3-siRNA sense: 5′-GGGACCUGGUGUGAAUUAUTT-3′, STAT3-siRNA antisense: 5′-AUAAUUCACACCAGGUCCCTT-3′, scrambled control siRNA sense: 5′-UUCUCCGAACGUGUCACGUTT-3′, and scrambled control siRNA antisense: 5′-ACGUGACACGUUCGGAGAATT-3′. A549 cells were transfected with STAT3-siRNA or Ctrl-siRNA using Lipofectamine® RNAiMAX according to the manufacture's protocol. After 48 hours, cells were collected and knockdown was evaluated using western blotting.
Cell viability assay
We determined the effects of NFV on A549 cell growth using an MTT assay. Human A549 cells were plated in 96-well plates at a density of 8000 cells per well and cultured overnight at 37°C. The next days, cells were treated with different concentrations of NFV (1, 2.5, 5, 10, 20, 40 and 60 µM) for 24 and 48 hours. MTT reagent was added to each well and incubated for 4 hours at 37°C. The formazan crystals formed by the reduction of MTT by mitochondrial succinic dehydrogenase secreted by viable cells were dissolved using 150 µL of DMSO. Absorbance was measured at 490 nm. The experiment was repeated using H1224 cells.
Colony formation assays
To assess colony formation by human A549 cells, cells were treated with NFV at various concentrations (5, 7.5 and 10 µM) for 20 hours. Subsequently, the NFV-containing medium was removed and the cells were incubated for an additional 6 to 10 days in NFV-free medium. Colonies were stained using crystal violet dye. A colony was defined as a cluster of at least 50 cells that could be observed microscopically.
Hoechst 33342 staining
Human A549 cells were cultured on glass coverslips and treated with different concentrations of NFV (10, 20 and 30 µM) for 24 hours. Subsequently, cells were fixed with 4% paraformaldehyde for 10 minutes at room temperature, then stained with 1 mg/mL Hoechst 33342 at room temperature for 5 minutes. After washing three times with phosphate-buffered saline (PBS), the slides were mounted with anti-fluorescence quenching agent and cover slips were added. The cells were immediately imaged using a fluorescent microscope (Olympus BX43, Tokyo, Japan) at ×400 magnification.
Western blotting
Cell lysates were prepared by extracting proteins with RIPA Lysis Buffer (Thermo Fisher Scientific) containing protease inhibitors (AbMole, Houston, TX, USA). Protein levels were quantitated using a protein quantification kit (Thermo Fisher Scientific). Proteins were separated by SDS-PAGE and transferred to a polyvinylidene difluoride membrane. The membrane was blocked with 5% skim milk powder for 2 hours, then incubated with a specific primary antibody at 4°C overnight. After incubation for 45 minutes with horseradish peroxidase-conjugated secondary antibody, cells were washed three times with Tris-buffered saline containing 0.01% Tween-20 on a shaker. Finally, blots were developed using enhanced chemiluminescence. Band density was measured using ImageJ software (NIH, Bethesda, MD, USA).
Annexin-V/PI staining
Cells were collected and washed twice with PBS and then resuspended in 200 µL of 1× binding buffer. The cells were stained with 5 µL of Annexin V and 10 µL of PI, mixed well, and incubated protected from light for 30 minutes at room temperature. After adding 400 µL of PBS, cells were analyzed by flow cytometry.
Statistical analysis
All results were expressed as means ± standard deviations. Differences among mean values were assessed using one-way analysis of variance. Values of P<0.05 were considered statistically significant.
Results
NFV effectively decreases viability of human A549 cells
MTT assays were used to investigate whether NFV influenced the viability of human A549 cells. A549 cells were treated with NFV at different concentrations (1, 2.5, 5, 10, 20, 40 and 60 µM) for 24 or 48 hours. As the dose of NFV increased from 1 to 60 µM, the viability of A549 cells was decreased in a dose-dependent manner (Figure 1a and 1b). The same effect was observed in H1224 cells (Figure S1). We also assessed the effect of NFV on A549 cell colony formation. After treatment with NFV (5, 7.5, and 10 µM), colony formation by A549 cells was remarkably reduced (Figure 1c and 1d).

Nelfinavir (NFV) dose-dependently inhibited proliferation of human A549 cells. a–b Cell viability was analyzed using an MTT assay. The proliferation of A549 cells treated with NFV (1, 2.5, 5, 10, 20, 40, and 60 µM) was decreased in a dose-dependent manner after treatment for 24 or 48 hours. c–d Effect of different NFV concentrations (5, 7.5, and 10 µM) on human A549 cell colony formation. A549 cells were incubated with NFV for 20 hours and allowed to grow for 6–10 days. Colonies were stained with crystal violet dye. The colony formation ability in each treatment group is shown. The results represent means ± standard deviations of three experiments. Dimethylsulfoxide (DMSO) alone was used a negative control. *P < 0.05, ** P < 0.01, *** P < 0.001.
NFV inhibits STAT3 phosphorylation in human A549 cells
We measured levels of p-STAT3 and STAT3 in human A549 cells by western blotting. Compared with DMSO alone, NFV treatment significantly attenuated STAT3 phosphorylation in a dose-dependent manner (Figure 2a and b). Studies have shown that STAT3 is the main factor that mediates the function of interleukin (IL)-6. 14 IL-6 binds to its receptor and activates Janus kinases to phosphorylate STAT3. Using the same NFV treatment conditions, p-STAT3 expression was increased in human A549 cells stimulated with IL-6 compared with NFV-treated cells without IL-6 stimulation. However, NFV still dose-dependently inhibited STAT3 phosphorylation (Figure 2c and d). As shown in Figure 2e and f, apoptosis of A549 cells treated with 20 µM NFV was significantly increased after STAT3 RNA silencing. These results indicated that NFV could be used as a STAT3 inhibitor to suppress STAT3 phosphorylation in human A549 cells.

Nelfinavir (NFV) impedes signal transducer and activator of transcription 3 (STAT3) phosphorylation in human A549 cells. (a) Levels of p-STAT3 and STAT3 were assessed by western blotting in human A549 cells. STAT3 was used as an internal control. (b) Quantification of data presented in panel (c) NFV inhibits interleukin 6 (IL6)-induced activation of STAT3 in human A549 cells. Cells were treated with NFV for 15 hours, followed by stimulation with IL6 (20 ng/mL) for 30 minutes. Cell lysate was subjected to immunoblotting to assess the expression of p-STAT3 and STAT3. (d) Quantification of data presented in panel (e) STAT3 expression was silenced to assess the mechanism underlying the effect of NFV on A549 cell apoptosis. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control. (f) Quantification of data presented in panel (e) Experiments were performed in triplicate and were independently repeated three times. **P<0.01, ***P<0.001 compared with dimethylsulfoxide (DMSO) alone; #P<0.05, ##P<0.01 compared with the negative control (NC) scrambled siRNA.
NFV induces human A549 cell apoptosis in a dose-dependent manner
Apoptotic cells were observed under a fluorescent microscope following Hoechst 33342 staining. The nuclei of DMSO-treated cells were intact and staining was not obvious. However, A549 cells treated with different concentrations of NFV (10, 20 and 30 µM) showed nuclei that were heavily condensed with chromatin as well as apoptotic bodies. The nuclei of NFV-treated cells were brighter than those of DMSO-treated cells (Figure 3a). These findings demonstrated that NFV induced apoptosis of A549 cells. We also used Annexin-V/PI staining and flow cytometry to measure the pro-apoptotic effect of NFV against A549 cells (Figure 3b). We further measured the expression of apoptosis-related proteins and found that NFV treatment significantly decreased the expression of Bcl-xL and Bcl-2 but promoted Cle-PARP expression (Figure 3c and d). Together, these data suggested that NFV impeded proliferation and promoted apoptosis in human A549 cells.
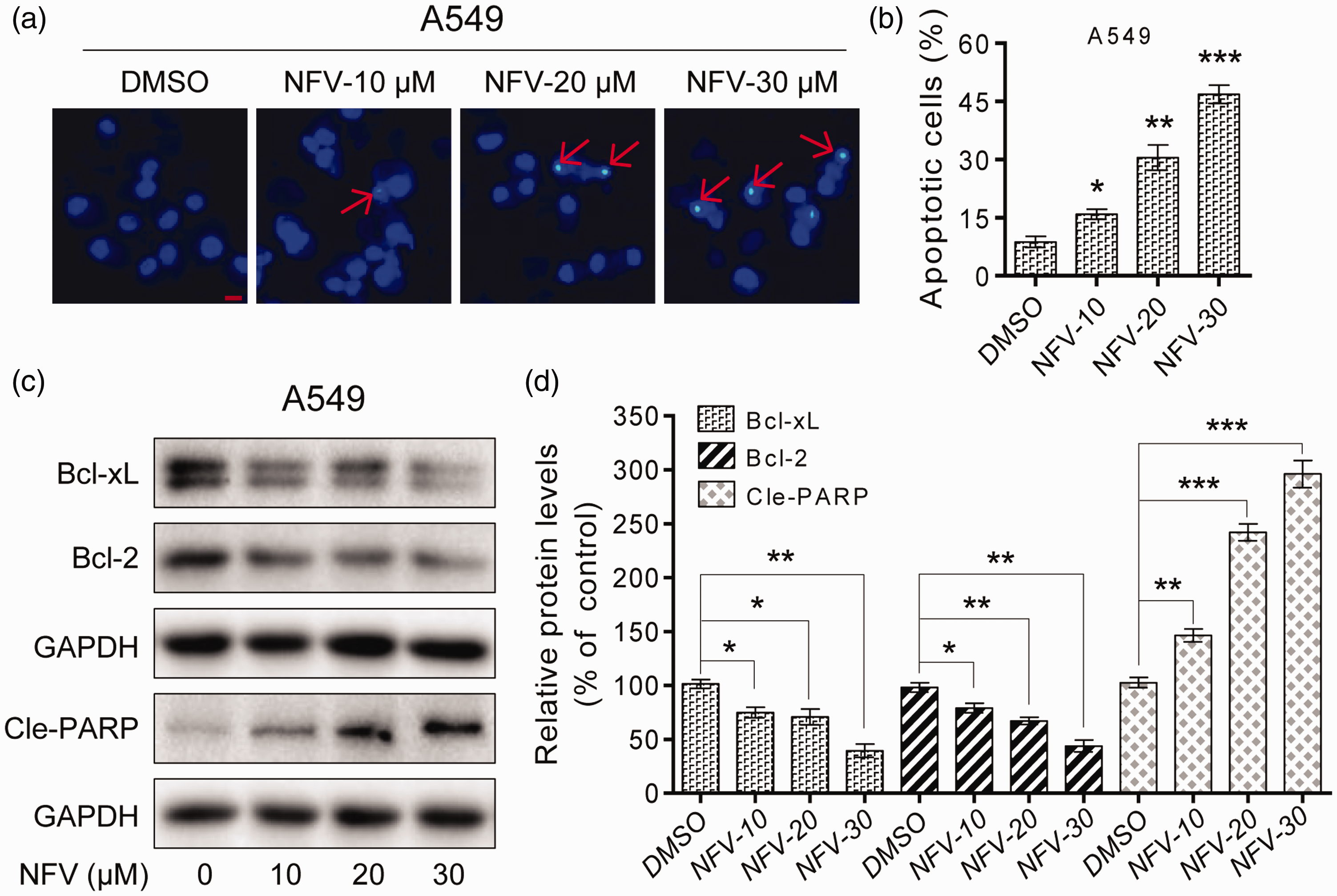
Nelfinavir (NFV) promotes apoptosis dose-dependently in human A549 cells. (a) Hoechst 33342 staining of the apoptotic cell population after A549 cells were exposed to NFV (10, 20, and 30 µM) for 24 hours. Apoptotic cells were observed under a fluorescence microscope. (b) A549 cells were treated with NFV (10, 20, and 30 µM) for 24 hours, then flow cytometry was used to quantify the percentage of apoptotic cells following Annexin-V/propidium iodide staining. (c) Expression levels of the apoptosis-related proteins Bcl-xL, Bcl-2 and Cle-PARP were determined by western blotting after treatment of A549 cells with NFV (10, 20, or 30 µM) for 24 hours. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control. (d) Quantification of data presented in panel (c) All images are representative of three independent experiments with similar results. The results represent means ± standard deviations of three experiments. *P<0.05, **P<0.01 and ***P<0.001 compared with dimethylsulfoxide (DMSO) alone.
Discussion
At present, surgery is the first choice for patients with NSCLC when there are no contraindications. 15 Depending on the tumor pathological type and presence or absence of lymph node metastasis, radiotherapy, chemotherapy, combination chemoradiotherapy, or molecular targeted therapy may be administered after surgery. 16 The aim of the current study was to elucidate the molecular mechanism of the anticancer effects of NFV against human A549 cells and to explore the effects of NFV on the growth of A594 cells. NFV is an anti-retroviral drug, but its anti-cancer effects and modulation of multiple signal transduction pathways have been demonstrated in several recent studies. Jensen et al. 17 suggested that NFV has an inhibitory effect on thyroid cancer through modulation of the PI3K/Akt and MAPK/ERK signaling pathways. An immunohistochemical staining analysis conducted by Gupta et al. 18 showed that NFV inhibits the growth and clonogenic death of adenoid cystic carcinoma cells by inhibiting the Akt pathway. In this study, we found that NFV significantly reduced the proliferation of human A549 cells at low concentrations (5 µM). A recent study showed that A549 cell proliferation could be inhibited by blocking the expression or activity of members of the STAT3 signaling pathway. 19 STAT proteins play a role in cancer development and deterioration. 20 Inhibition of STAT3 activation has been an important target for cancer therapy. 21 In NSCLC, STAT3 plays an important role by regulating the proliferation, survival, metastasis, and chemoresistance of cancer cells. 22 Bono et al. 23 showed that NFV-associated myeloma cell death was mediated by reduced STAT3 phosphorylation. Proliferation of prostate cancer cells could also be inhibited by NFV via blockade of STAT3 signaling. 24 Consistently, our results showed that apoptosis of A549 cells was significantly increased following treatment with 20 µM NFV when STAT3 expression was silenced. Thus, we demonstrated that NFV blocked STAT3 signaling in a dose-dependent manner, thus promoting the apoptosis of human A549 cells.
We also found that that apoptosis of human A549 cells was increased after NFV treatment. The expression of the pro-apoptotic protein Cle-PARP increased, while the expression of the anti-apoptotic proteins Bcl-2 and Bcl-xl decreased following NFV treatment. Xu et al. 25 showed that apoptosis of A549 lung cancer cells could be induced by reactive oxygen species-triggered STAT3 signaling, thereby promoting the expression of CLE-PARP. Activation of STAT3 can sustain tumor growth and oncogene manifestations, enhance the expression of the apoptosis inhibitors Bcl-2 and Bcl-xl, and antagonize chemotherapeutic drugs acting on apoptotic pathways. 26 Overall these events have anti-apoptotic effects, consistent with our study.
Recent studies have found that chronic inflammation is closely related to tumorigenesis. Various inflammatory cells in the tumor microenvironment can promote the occurrence and development of tumors by secreting cytokines or generating oxygen free radicals. 27 Many cancers, including NSCLC, are characterized by constitutive STAT3 activation. Thus, drugs capable of preventing STAT3 activation may have potential to treat cancer. 28 Yang et al. 29 reported that tumor-associated macrophages can activate STAT3 in tumor cells through paracrine pathways and subsequently upregulate SOX2 expression. Santiago et al. 30 found that extracellular NF-κB-dependent IL-6 production promoted STAT3 activation in colorectal cancer cells. IL-6, an important inflammatory factor, has been reported to be highly expressed in patients with lung cancer who have a poor prognosis. 31 After IL-6 binds to its receptors, JAK2/STAT3 signaling is activated, promoting JAK2 phosphorylation and STAT3 phosphorylation and formation of STAT3 homodimers, which are transported to nuclear junction DNA. There, they activate the transcription of STAT3 target genes that regulate cell growth, apoptosis, invasion, migration, and angiogenesis.32,33 In this study, we found that IL-6 stimulation could increase the expression of p-STAT3 and STAT3. However, these increases were inhibited following NFV treatment, indicating that NFV could inhibit STAT3 signaling in human A549 cells as well as IL-6-induced STAT3 activation. The overall effects of NFV treatment were inhibition of growth of human A549 cells, inhibition of colony formation, and promotion of apoptosis. Our findings agree with those of a previous study. 34 The detailed mechanisms underlying the anticancer effects of NFV need to be further studied.
Conclusion
This study investigated the anticancer effects of NFV on A549 lung cancer cells. We found that NFV induced apoptosis of human A549 cells by inhibiting STAT3 signaling. In conclusion, NFV has potential as a novel drug for the treatment of NSCLC. In addition, disruption of STAT3 signaling may represent a target for the development of new anticancer drugs.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211014989 - Supplemental material for Nelfinavir restricts A549 cell growth by inhibiting STAT3 signaling
Supplemental material, sj-pdf-1-imr-10.1177_03000605211014989 for Nelfinavir restricts A549 cell growth by inhibiting STAT3 signaling by Fangli He, Gang Wang, ZhengQing Xu and Zhihui Huang in Journal of International Medical Research
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
