Abstract
Methicillin-resistant Staphylococcus epidermidis (MRS) predominantly colonizes the skin and mucous membranes of humans and other animals. We describe the case of a male newborn of gestational age 39 weeks whose primary and repeated blood cultures and cerebrospinal fluid samples isolated MRS. The choice and duration of antibiotic therapy were determined by the clinical presentation, infection parameters, and results of bacteriological analyses of blood and cerebrospinal fluid samples obtained from the newborn on the day 5 of life. After 28 days of antibiotic therapy for sepsis accompanied by meningitis, the newborn was discharged home without sequelae.
Introduction
Staphylococcus spp. colonize the skin and mucous membranes of humans and other animals and can be found on various objects and surfaces. A coagulase test is used to differentiate coagulase-positive and coagulase-negative staphylococci (CNS). 1 S. aureus, as the most common coagulase-positive staphylococcus with a large number of virulence factors identified by sequencing its entire genome, represents one of the most common causes of infections that have different clinical manifestations. 2
CNS species form part of the normal physiological flora of animal skin and mucous membranes. The frequency of nosocomial infections in these species has led to a serious consideration of their virulence factors. Repeated CNS isolates with the same or similar phenotypic characteristics of antibiotic susceptibility, along with clinical parameters and specific biomarkers of infection, may confirm infection, although difficulties in interpreting findings can persist. 3
Case Report
A male newborn from a first controlled pregnancy was born naturally at 39 weeks’ of gestation, weighing 3800 g, with a body length of 53 cm, head circumference of 35 cm, and Apgar score of 9. His mother had a premature rupture of membranes lasting 40 hours. Cervical and vaginal swabs and a urine culture 2 days before delivery were normal, and blood test results and C-reactive protein (CRP) levels were within pre-delivery reference values. She received 2 g of ceftriaxone i.v. prior to delivery. Lochia and swabs from the vagina and cervix, and throat and nose showed no negative parameters of infection. The newborn was breastfed immediately after birth.
On day 5 of life, the newborn became febrile with a body temperature of 39.4°C, and venous bilirubin levels of 277 mmol/L. The white blood cell (WBC) count was within normal ranges (16.0 × 109/L), with hemoglobin levels of 178 g/L, a red blood cell count of 6.73 × 109/L, KG A Rh positivity, presepsin levels of 868 pg/mL, procalcitonin of 0.5 ng/mL, and CRP of 22.0 mg/L. The cerebrospinal fluid (CSF) was clear, without cellular elements. Antibiotic therapy was started with ampicillin 100 mg/kg/12 hours and amikacin 15 mg/kg/24 hours. Intravenous rehydration using 10% glucose and 0.9% NaCl and phototherapy were also initiated to venous bilirubin levels of 277 mmol/L. A urine sample cultured after 24 hours showed negative findings.
After 24 hours of incubation in microaerophilic conditions at 36°C, the primarily inoculated CSF was sieved with standard isolation media. After 18 hours of incubation, an increase in white non-hemolytic colonies was observed. A Staphylococcus slide culture test and in vitro plasma test were negative. The isolated culture was identified as methicillin-resistant S. epidermidis (MRS) using a VITEK 2 Compact device (bioMerieux, Marcy-l’Etoile, France).
On day 7 of life, the newborn was afebrile with a WBC count of 36.0 × 109/L, CRP levels of 23.0 mg/L, presepsin of 1868 pg/mL, and procalcitonin of 2.5 ng/mL. The liquor was yellow and cloudy. No bacterial or fungal elements were seen in the direct microscopic preparation from the CSF, although pleocytosis (up to 1500 WBC/mm3) was found as an indicator of meninges inflammation. Serum bilirubin levels were 378 mmol/L. Treatment with meronem 40 mg/kg/8 hours and vancomycin 20 mg/kg/12 hours was initiated. Vancomycin-sensitive S. epidermidis was isolated from hemoculture and CSF culture l, and therapy was continued according to the vancomycin 20 mg/kg/12 hours antibiogram (Table 1).
Antibiogram for Staphyloccocus epidermidis treatment.

After 72 hours, other CSF and blood samples taken at the same time were sent to the microbiological laboratory. MRS was isolated from both samples, and identified to the species level and antibiotic sensitivity using a VITEK 2 Compact device. No bacterial, fungal, or other elements indicative of an inflammatory reaction were seen in the direct microscopic preparation of CSF.
On day 10 of life, the WBC count was 12 × 109/L, CRP levels were 20 mg/L, presepsin was 668 pg/mL, and procalcitonin was 0.5 ng/mL. The CSF was clear with 368 WBC/mm3. The urine culture, CSF culture, and blood culture were all sterile. Vancomycin 20 mg/kg/12 hours therapy was continued.
On day 14 of life, blood test results were within reference values, with a WBC count of 10 × 109/L, CRP <5 mg/L, presepsin of 20 pg/mL, and procacitonin of 0.25 ng/mL. The liquor was clear without cellular elements. Liquor and blood cultures were sterile (Table 2).
Laboratory results of blood and cerebrospinal fluid.
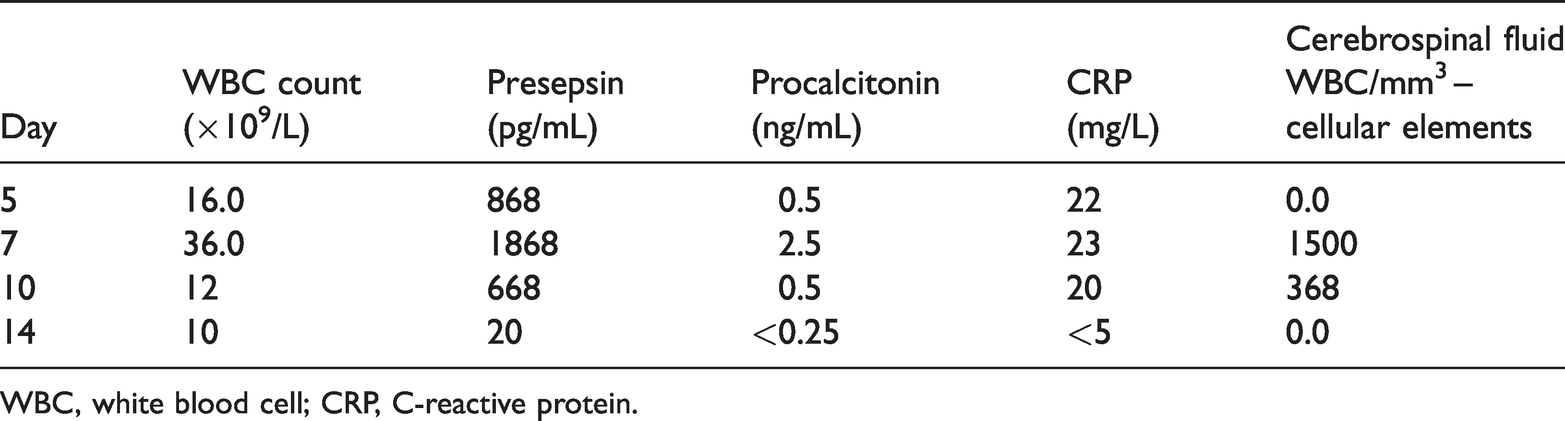
WBC, white blood cell; CRP, C-reactive protein.
Antibiotic therapy with vancomycin was continued for 28 days because of sepsis accompanied by meningitis with MRS CNS.
An ultrasound of the brain and abdomen, and electroencephalography findings were normal. Examinations by an ophthalmologist and audiologist in the first, third, and twelfth months of life were normal. Neurological development in the first, third, sixth, and twelfth months of life were normal.
Discussion
CNS isolated from clinical samples are commonly interpreted as commensal pathogens. The frequency of CNS species isolation, especially from nosocomial infections, has led clinicians to consider CNS virulent factor genes to be responsible for the production of toxins, biofilms, adhesins, hemolysins, exoenzymes, and superantigens. An agr quorum sensing system has been identified in CNS species, which responds to and is activated by self-generated, autoinducing peptides, similar to S. aureus.4,5
The most important step in the transition of CNS from a commensal organism into a pathogen with clinical manifestations is penetration through the skin barrier. In neonatal sepsis, the ST2 clone is most commonly isolated, but it can be difficult to determine whether it is the cause or a commensal organism of the anatomical region from which the sample was obtained for microbiological examination. 6 This clone has the ability to produce biofilm, but it is unclear whether this enables its invasiveness. 7
Molecular genomic studies have shown that CNS species S. lugdunensis, S. haemolyticus, S. saprophyticus, and S. capitis are also common causes of clinical infections.8–10 S. caprae forms part of the physiological flora of animal skin, including that of humans. It mainly causes mastitis in animals and osteoauricular infections in humans.11–13
The most common CNS species associated with human infections are S. epidermidis, S. lugdunensis, S. saprophyticus, S. haemolyticus, S. caprae, and S. capitis, with S. epidermidis the leading cause of CNS-associated nosocomial infections. The production of biofilm is the main way in which medical and prosthetic instruments and aids used in treatment are colonized. Moreover, the presence of mobile elements and virulence factors in the genome and CNS cells enables their horizontal transfer between species, which underlies their pathogenicity. 14 S. epidermidis has emerged as the predominant pathogen of sepsis in preterm infants, with the prevalence of S. epidermidis sepsis, defined as one or more positive blood cultures with corresponding clinical signs, ranging from 1.9% to 4.8% in the general neonatal population; it may be as high as 9.4% in very low birth weight infants. 15
Particularities of neonatal immunity may contribute to the emergence of S. epidermidis as an important pathogen. While S. epidermidis is the causative agent of severe hospital-acquired infections in some clinical cases, it can be difficult to distinguish between infection and contamination of the clinical specimen, 5 as evidenced in the present case. The reporting of this study conforms to CARE guidelines. 16
Conclusion
In neonatology, it can be difficult to interpret a single finding of CNS as a cause of sepsis or meningitis. A correlation between microbiological findings, clinical presentation, and biomarkers of infection is necessary to do so.
Footnotes
Ethics
Ethical permission was not applied for as this is a presentation of laboratory analyses of a case, which does not include photographs.
Declaration of interest
The authors report no conflict of interest. The authors are solely responsible for the content and writing of the article.
Consent
Informed written consent was obtained from the patient’s mother for analysis and treatment. Consent for publication was also obtained from the mother.
Funding
The authors received no financial support for the research, authorship, or publication of this article.
