Abstract
Objective
This study investigated the peripheral Th17/Treg balance and its potential controlling factor indoleamine 2,3 dioxygenase (IDO) in patients with periodontitis and atherosclerosis (AS), as well as its correlation with Porphyromonas gingivalis infection.
Methods
In this retrospective study, P. gingivalis-infected atherosclerotic patients (Pg-AS), atherosclerotic patients (AS), P. gingivalis-infected periodontitis patients (Pg), and healthy controls (HCs) were selected after clinical examination, subgingival plaque examination, and plasma anti-P. gingivalis antibody analysis. Treg and Th17 cell percentages, related transcription factors, and functional cytokines in peripheral blood were analysed. Plasma tryptophan (Trp) and kynurenine (Kyn) were measured to determine IDO activity.
Results
Atherosclerotic patients (Pg-AS and AS groups) had significantly lower IDO activity and higher Th17/Treg ratio than those in the Pg and HC groups. The Th17/Treg ratio was higher and IDO activity was lower in the Pg-AS group compared with the AS group. Transcription factors and cytokines exhibited the same trend as the Th17 and Treg cells. Additionally, IDO activity was negatively correlated with the plasma anti-P. gingivalis antibody titre and the Th17/Treg ratio in the atherosclerotic group.
Conclusions
P. gingivalis may reduce IDO activity and further promote Th17/Treg imbalance to facilitate AS development. IDO may be a novel molecular marker to predict periodontitis-associated AS.
Keywords
Introduction
Periodontitis is a chronic inflammatory disease caused by dental plaque bacteria. Bacterial accumulation on the tooth surface may induce local inflammation in the periodontal tissue characterized by gingival bleeding, alveolar bone resorption, tooth mobility, or tooth loss.1,2 Currently, an increasing amount of evidence suggests that chronic periodontitis can be a risk factor for the development of atherosclerosis (AS). Porphyromonas gingivalis, which is the major pathogen in periodontitis, plays a key role in the relationship between periodontitis and AS. 3
AS is a chronic inflammatory disease. The imbalance between T helper 17 (Th17) cells and regulatory T cells (Tregs), which is associated with Th17 cell upregulation and Treg downregulation, is an important cause of AS.4–6 Indoleamine 2,3-dioxgenase (IDO) may be a key protein that regulates the Th17/Treg balance. 7 IDO is mainly expressed in antigen-presenting cells and is the rate-limiting enzyme for tryptophan (Trp) catabolism through the kynurenine (Kyn) pathway. IDO activity can be assessed using the Kyn/Trp ratio. An IDO metabolite may inhibit Th17 cell differentiation and induce naïve CD4+ T cells to differentiate into Tregs, which may ultimately affect the Th17/Treg balance. 8 Therefore, IDO plays an important role in regulating the Th17/Treg balance.
As the central protein that regulates Th17/Treg balance, IDO may play a protective role during AS development. 9 Niinisalo et al. demonstrated that the Kyn/Trp ratio gradually decreased with increased intima-media thickness in patients with coronary heart disease, indicating that IDO activity correlates with carotid artery intima–medial thickness. 10 A previous study showed that P. gingivalis suppressed IDO mRNA expression through its virulence factors. 11 Although many studies have indicated a potential role of P. gingivalis in promoting AS, the role of IDO in this process remains unclear. Additionally, it is still unknown whether the Th17/Treg balance is associated with P. gingivalis infection in AS patients.
The aim of the present study was to investigate the peripheral Th17/Treg balance and IDO in patients with periodontitis and AS, and to establish their potential correlation with P. gingivalis. We hypothesized that IDO activity is a potential predicative factor for P. gingivalis-associated AS.
Patients and methods
This retrospective research was approved by the Institutional Review Board of Nanjing Stomatological Hospital, Medical School of Nanjing University (2017NL-016(KS) on 23 February 2017). This research was conducted in accordance with the World Medical Association Declaration of Helsinki. All participants provided written informed consent before inclusion into the study. We de-identified all patient information. The reporting of this study conforms to the STROBE guidelines. 12
Patients
To determine the role of P. gingivalis in patients with periodontitis and AS, patients were enrolled and stratified into the following four groups: patients with periodontitis and AS (Pg-AS); patients with AS only (AS); patients with periodontitis only (Pg); and healthy controls (HC). The inclusion criterion was age 18 to 70 years. Periodontitis was defined in accordance with the new periodontitis classification that was proposed in 2018. 13 Patients with Stage II–IV periodontitis were enrolled as periodontal cases in this study. P. gingivalis 16S rRNA was examined using reverse transcription polymerase chain reaction (RT-PCR) to identify P. gingivalis in subgingival plaques. To obtain subgingival plaque samples, supragingival plaques were removed, and the selected sites were dried with sterile cotton rolls. Then, subgingival plaques were obtained from the deepest periodontal pocket using sterile curettes.
AS was defined on the basis of coronary angiography examination results that indicated more than 50% stenosis of the coronary artery. Patients with a carotid artery intima–media thickness of less than 9 mm on carotid artery sonography were excluded from the AS risk group.
The exclusion criteria were patients with immunodeficiency disease or diabetes; or patients who took immunosuppressive drugs or antibiotics or who had an infection in the last 3 months.
The sample size was estimated on the basis of the results from six AS patients and six Pg-AS patients in a pilot experiment. Twenty-nine participants in each group were needed to achieve 80% power with a significance level of 5%. The patients were distributed into four groups according to the inclusion and exclusion criteria. In the Pg-AS group, patients were diagnosed with both periodontitis and AS, which was accompanied by P. gingivalis infection in the subgingival plaque. In the AS group, patients were diagnosed with AS with no clinical sign of periodontitis or P. gingivalis in subgingival plaques. In the Pg group, patients were diagnosed with periodontitis, and P. gingivalis was detected in subgingival plaques, but there were no clinical signs of AS and the carotid artery intima–media thickness was less than 9 mm on carotid artery sonography. In the HC group, subjects had no AS symptoms after carotid artery sonography and electrocardiography examination. No evidence of periodontal inflammation or P. gingivalis in subgingival plaques was found.
The average age was matched and sex distribution in each group was considered when selecting participants to ensure a normal distribution. The plaque index (PI), gingival index (GI), probing pocket depth (PPD), and clinical attachment level (CAL) were recorded. Smoking history was also recorded. Anti-P. gingivalis immunoglobulin (Ig)G antibodies in human plasma were examined as described by Ishikawa et al. 14
Flow cytometry analysis of Th17 and Treg cells
Fresh peripheral blood was collected in heparinized tubes. Plasma was obtained by centrifugation at 1000 × g for 10 minutes. Peripheral blood mononuclear cells (PBMCs) were isolated using Ficoll–Hypaque density gradient centrifugation (TBD Sciences, Tianjin, China). For Treg analysis, PBMCs were stained using anti-human CD4 fluorescent monoclonal antibodies labelled with fluorescein isothiocyanate (FITC) and anti-human CD25 labelled with allophycocyanin (APC) as a surface marker. After fixation and permeabilization, the cells were labelled with phycoerythrin (PE) conjugated anti-human FoxP3 antibodies (eBioscience, San Diego, CA, USA).
For Th17 cell subset analysis, PBMCs were stimulated for 6 hours with 50 ng/mL phorbol myristate acetate (PMA), 1 μg/mL ionomycin, and 1.7 μg/mL monesin (Sigma-Aldrich, St. Louis, MO, USA). FITC-conjugated anti-human CD4 antibodies were used for cell surface staining before fixation and permeabilization. Cells were incubated with Cytofix/Cytoperm reagent (BD Biosciences, San Diego, CA, USA) for 1 hour. Cells were then incubated with PE-conjugated anti-human interleukin (IL)-17A antibodies (eBioscience) for intracellular staining. CD4+CD25+FoxP3+Treg cells and CD4+IL17+Th17 cells were analysed by flow cytometry using a flow cytometer (FACSCalibur flow cytometer, BD Biosciences). The Treg and Th17 cell frequencies were analysed with FlowJo v.7.6.1 software (Tree Star, Ashland, OR, USA).
Real-time polymerase chain reaction analysis of transcription factors
Total RNA was isolated from PBMCs using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). RNA was reverse transcribed into cDNA using a reverse transcription kit (TaKaRa Biotechnology, Dalian, China). RT-PCR was performed in accordance with the manufacturer’s instructions using iTaq Universal SYBR green Supermix (Bio-Rad, Hercules, CA, USA). β-actin was a reference gene. The primers 15 are shown in Supplementary Table 1. All reactions were performed using a Step-one RT-PCR system (Applied Biosystems, Foster City, CA, USA). The expression levels of each mRNA were calculated using the comparative quantification algorithms 2ΔΔCT method. The relative expression of each gene was normalized to that of β-actin.
Enzyme-linked immunosorbent assay analysis of plasma cytokines
Plasma was obtained from peripheral blood samples and stored at −80°C. The levels of human IL-10 (eBioscience), transforming growth factor-beta 1 (TGFβ1; Dakewe, Shanghai, China), and IL-17A (eBioscience) in plasma were measured using enzyme-linked immunosorbent assay (ELISA) kits in accordance with the manufacturer’s instructions. Briefly, standard samples were added to a 96-well plate to obtain a standard curve. Plasma samples were added and incubated with antibody-coated plates. After the wells were washed with PBS, detection antibodies were added to the samples, and antibodies labelled with streptavidin conjugated with horseradish peroxidase were subsequently added. Substrate was added for coloration and stopped with sulfuric acid. Cytokine concentrations were measured using a spectrophotometer.
Indoleamine 2,3-dioxgenase activity determination
Kyn and Trp concentrations were determined by ELISA (LDN, Nordhorn, Germany). IDO activity is shown as the ratio of the Kyn concentration (μmol/L) to the Trp concentration (mmol/L).
Measurement of blood biochemistry
Level of blood lipids, including serum low-density lipoprotein cholesterol (LDL) and high-density lipoprotein cholesterol (HDL), were determined using enzymatic methods.
Statistical analysis
A power analysis was performed using PASS v.11 software (NCSS Corporation, Kaysville, UT, USA). The data were analysed using statistical software (SPSS, v.24.0, IBM Corp., Armonk, NY, USA). The Shapiro–Wilk test was performed to determine whether the data were normally distributed. Human anti-P. gingivalis IgG ELISA unit (EU) values were abnormally distributed. The results are presented as the median (range). The Kruskal–Wallis H test was used to analyse data among multiple groups, and the Mann–Whitney U test was used to analyse two groups after the Bonferroni correction. The other data were normally distributed, and the experimental results are presented as the mean ± standard deviation (SD). A one-way analysis of variance test was used to examine multiple groups. The correlation between IDO activity and the Th17/Treg ratio was analysed using the Pearson correlation test. The Spearman correlation test was used to analyse correlations between P. gingivalis antibody titre, Th17/Treg ratio, and IDO activity because the P. gingivalis antibody titre was not normally distributed. Differences of P < 0.05 were considered to be statistically significant.
Results
Patient characteristics
There were 1251 patients who were screened for this study and among them, 128 patients were enrolled. The average age of patients in the Pg-AS group (n = 32) was 55.40 ± 8.50 years and 17 were women while 15 were men. The average age of patients in the AS group (n = 32) was 54.25 ± 6.77, and 15 were women and 17 were men. The average age of patients in the Pg group (n = 32) was 53.00 ± 6.05 years, and 18 were women while 14 were men. The average age of participants in the HC group (n = 32) was 52.03 ± 6.54, and 17 were women and 15 were men. There was no significant difference in age or sex. No difference was observed in HDL levels among the groups. In the Pg-AS groups, LDL levels were higher than those in the Pg and HC groups (P < 0.05 for both). Basic demographic features of patients in the Pg-AS, AS, Pg, and HC groups are shown in Table 1.
Participants’ demographic characteristics and clinical parameters.

Normally distributed data are expressed as the mean ± standard deviation, and nonnormally distributed data are expressed as the median (minimum–maximum).
aP < 0.05 vs. control, bP < 0.05 vs. Pg, cP < 0.05 vs. AS.
Groups: HC, healthy controls; Pg-AS, P. gingivalis-infected atherosclerotic patients; AS, atherosclerotic patients; Pg, P. gingivalis-infected periodontitis patients.
AS, atherosclerosis; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; PI, plaque index; GI, gingival index; PPD, probing pocket depth; CAL, clinical attachment level; IgG, immune globulin G; Ab, antibody; EU, ELISA unit; ELISA, enzyme-linked immunosorbent assay.
To evaluate the periodontal infection level, periodontal parameters including PI, GI, mean PPD, and mean CAL were recorded. Individuals in the P. gingivalis-infected groups (Pg-AS and Pg groups) showed worse periodontal inflammation because the value PI, GI, mean PPD, and mean CAL values were higher compared with those in the non-P. gingivalis-infected groups (HC and AS group; P < 0.05 for all comparisons). Anti-P. gingivalis IgG antibody titres were also examined for all participants. Anti-P. gingivalis IgG antibody levels were 3.6-fold higher in the Pg-AS and Pg groups compared with those in the AS and HC groups (P < 0.05). No statistically significant differences in anti-P. gingivalis IgG antibody titres were found between the Pg-AS and Pg groups.
Circulating frequency of Treg and Th17 cells
The frequency of CD4+CD25+FoxP3+Treg cells among CD4+T cells in the Pg-AS, AS, PG, and HC groups are presented in (Figure 1). The frequency of the CD4+CD25+ FoxP3+Treg cells among CD4+T cells in the AS and Pg-AS groups was 2.64 ± 0.72% and 2.00 ± 0.51%, respectively. These levels were significantly decreased compared with those in the Pg group (4.14 ± 0.84%) and the HC group (4.16 ± 1.01%) (P < 0.05 for both; Figure 1c).
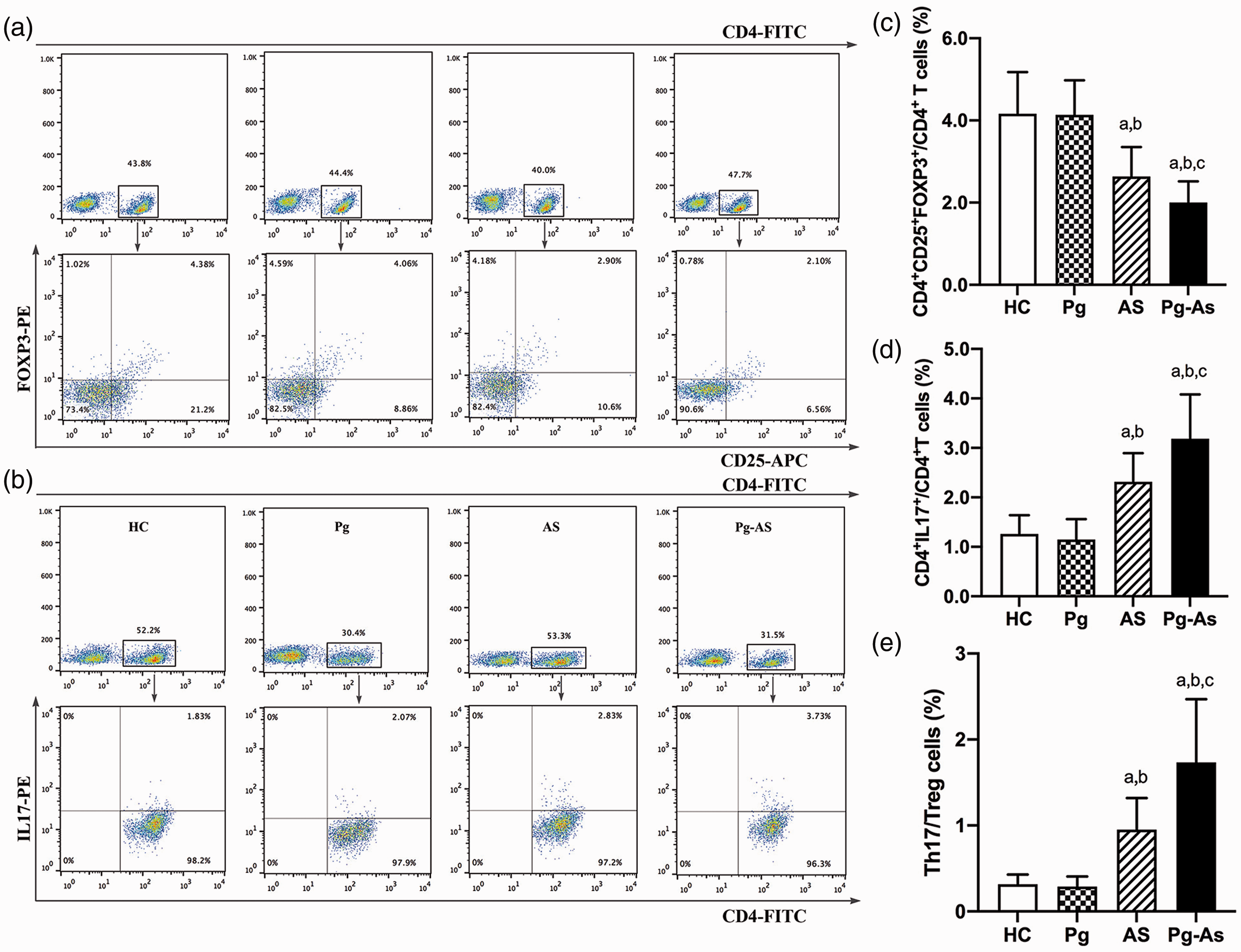
Immune balance between Th17 cells and Treg cells in peripheral blood of all participants. a and c. Comparison of the frequencies of CD4+CD25+FoxP3+T cells in the Pg-AS, AS, Pg, and HC groups. b and d. Comparison of the frequencies of CD4+IL17+T cells in CD4+T cells in the Pg-AS, AS, Pg, and HC groups. e. Comparison of Th17/Treg ratio in the Pg-AS, AS, Pg, and HC groups. aP < 0.05 vs. control, bP < 0.05 vs. Pg, cP < 0.05 vs. AS.
The percentage of Th17 cells in the Pg-AS group was 3.18 ± 0.90%, which was significantly higher than that in the AS group (2.31 ± 0.57%) (P < 0.05). Compared with those in the Pg-AS and AS groups, the percentages of Th17 cells in Pg (1.15 ± 0.41%) and HC groups (1.26 ± 0.38%) were significantly lower (P < 0.05; Figure 1d). Moreover, the Pg-AS group showed a significantly higher Th17/Treg ratio compared with the HC, Pg, and AS groups (P < 0.05 for all; Figure 1e). However, there was no significant difference in Th17 percentage, Treg percentage, or Th17/Treg ratio between Pg and HC groups.
Quantitative analysis of transcription factors
FoxP3, TGFβ1, and IL-10 mRNA expression in PBMCs was significantly lower in the Pg-AS group (0.46-fold, 0.54-fold, and 0.56-fold, respectively) and the AS group (0.73-fold, 0.70-fold, and 0.70-fold, respectively) than in the Pg and HC groups (P < 0.05). Additionally, FoxP3, TGFβ1, and IL-10 expression was lower in the Pg-AS group compared with the AS group (P < 0.05). Furthermore, the expression of RORγt, a transcription factor involved in Th17 cell differentiation, was 2.2-fold higher in the Pg-AS group and 1.6-fold higher in the AS group than in the Pg and HC groups, which was a significant difference (P < 0.05). There were no obvious differences in transcription factor and cytokine mRNA expression between the Pg and HC groups (Figure 2).

Gene expression of transcription factors in PBMCs. RNA was extracted from PBMCs. The mRNA expression of (a) FoxP3, (b) RORγt, (c) TGFβ, and (d) IL-10 were determined by RT-PCR. aP < 0.05 vs. control, bP < 0.05 vs. Pg, cP < 0.05 vs. AS.
Plasma cytokines
To determine the function of Treg cells and Th17 cells, the plasma levels of related cytokines were determined using ELISA. The TGFβ1 concentration was significantly lower in the Pg-AS (11.81 ± 1.38 ng/mL) and AS (16.27 ± 1.65 ng/mL) groups compared with that in the Pg (20.72 ± 1.52 ng/mL) and HC (19.77 ± 1.30 ng/mL) groups (P < 0.05). Similarly, the IL-10 concentration was significantly lower in the Pg-AS group (19.33 ± 1.97 pg/mL) and the AS group (24.82 ± 2.83 ng/mL) compared with that in the Pg group (30.12 ± 2.63 pg/mL) and HC group (30.34 ± 2.04 pg/mL) (P < 0.05 for all). Furthermore, TGFβ1 and IL-10 concentrations were significantly decreased in the Pg-AS group compared with those in the AS group.
The IL-17 concentration was higher in the Pg-AS group (41.12 ± 3.10 pg/mL) and in the AS group (28.17 ± 3.19 ng/mL) compared with that in the Pg (14.63 ± 2.56 pg/mL) and HC (14.98 ± 1.87 pg/mL) groups (P < 0.05). Additionally, the IL-17 concentration was significantly higher in the Pg-AS group than in the AS group. However, there were no significant differences between the Pg group and HC group (P < 0.05; Figure 3).

Plasma TGFβ1, IL-10, and IL-17 concentration in participants. (a) Plasma TGFβ1, (b) IL-10, and (c) IL-17 concentrations in the four groups were detected using ELISA. The concentrations of these cytokines are shown as the mean ± SD. aP < 0.05 vs. control, bP < 0.05 vs. Pg, cP < 0.05 vs. AS.
IDO activity and its correlation with Th17/Treg in Pg-AS
The Trp and Kyn plasma concentration and the Kyn/Trp ratio results are shown in Table 2. Patients with Pg-AS had lower plasma Kyn and higher Trp levels compared with those of AS patients, Pg patients, and HCs (P < 0.05 for all). The IDO activity, as determined by Kyn/Trp, was significantly lower in Pg-AS patients than in non-P. gingivalis-infected AS patients (P < 0.05). In Pg-AS patients, IDO activity was negatively correlated with the Th17 cell percentage (r = −0.424) and with the Th17/Treg ratio (r = −0.484). A positive correlation was shown between IDO activity and the Treg percentage (r = 0.373; Table 3, Figure 4).
Blood plasma concentrations of Trp, Kyn, and Kyn/Trp.
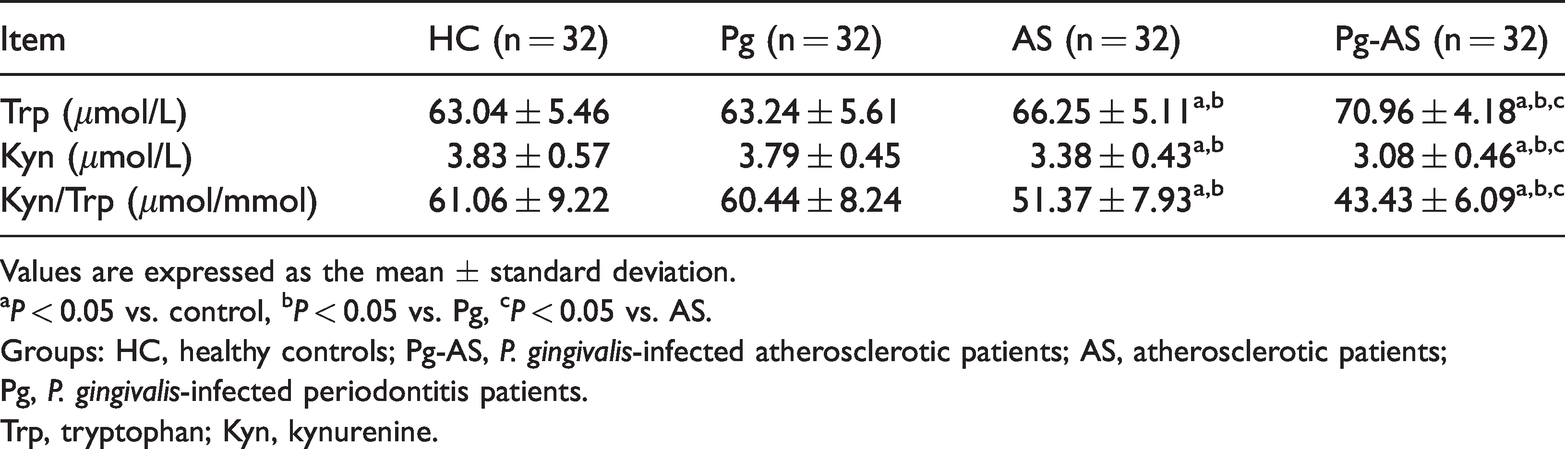
Values are expressed as the mean ± standard deviation.
aP < 0.05 vs. control, bP < 0.05 vs. Pg, cP < 0.05 vs. AS.
Groups: HC, healthy controls; Pg-AS, P. gingivalis-infected atherosclerotic patients; AS, atherosclerotic patients; Pg, P. gingivalis-infected periodontitis patients.
Trp, tryptophan; Kyn, kynurenine.
Relationship between plasma IDO and Th17 cell, Tregs, and Th17/Treg in Pg-AS patients

Pg-AS, P. gingivalis-infected atherosclerotic patients; IDO, indoleamine 2,3 dioxygenase; Treg, regulatory T cells; Th17, T helper 17 cells.

Correlation between IDO and Th17, Treg, and Th17/Treg in Pg-AS patients. Pearson correlation analysis of (a) IDO and CD4+CD25+FoxP3+/CD4+ T cells frequencies, (b) CD4+IL17+/CD4+T cells frequencies, and (c) Th17/Treg ratio in the Pg-AS patients.
Correlation analysis of P. gingivalis infection with Th17/Treg imbalance and IDO activity in Pg-AS
In patients with AS, the percentage of Th17 cells was positively correlated with the plasma P. gingivalis antibody titre (r = 0.441). Peripheral Tregs were negatively correlated with the P. gingivalis antibody titre (r = −0.251). Additionally, the Th17/Treg ratio was positively correlated with the plasma P. gingivalis antibody titre (r = 0.435). Moreover, the P. gingivalis antibody titre exhibited a negative relationship with IDO activity (r = −0.499; Table 4, Figure 5).
Relationship between P. gingivalis antibody titre and Th17 cell, Tregs, Th17/Treg, and IDO in Pg-AS patients.

Pg-AS, P. gingivalis-infected atherosclerotic patients; IDO, indoleamine 2,3 dioxygenase; Treg, regulatory T cells; Th17, T helper 17 cells.

Relationship between P. gingivalis antibody titres and Th17, Treg, Th17/Treg, and IDO in the Pg-AS patients. Spearman correlation analysis of P. gingivalis antibody titres and (a) Th17, (b) Treg, (c) Th17/Treg, and (d) IDO in the Pg-AS patients.
Discussion
T cell-mediated acquired immunity is implicated in the pathogenesis of AS. The immune imbalance between Th17 cells and Tregs plays a key role during this process. Treg cells, known as suppressor T cells, are a subset of CD4+T cells that play a protective role in AS. Th17 cells are another subset of CD4+T lymphocytes, and they play a proatherogenic role in AS. Recently, several in vivo and in vitro studies15–17 have indicated that during AS, Th17/Treg imbalance is characterised by a decrease in Treg cells and an increase in Th17 cells. Consistent with our findings, the Th17/Treg ratio included increased Th17 frequency and decreased Treg frequency in atherosclerotic patients. Considering that the Th17/Treg balance is essential for regulating immune balance and little is known about whether Th17/Treg imbalance is associated with P. gingivalis in AS patients, we analysed the relationship between P. gingivalis infection and T cell immune response in AS.
We found that P. gingivalis-infected atherosclerotic patients had significantly decreased numbers of peripheral Tregs and an increased percentage of peripheral Th17 cells. This result was further confirmed by the downregulation of FoxP3 (Treg cell-specific transcription factor) and upregulation of RORγt (Th17 cell transcription factor) in P. gingivalis-infected atherosclerotic patients. In addition to the number of cells, T cell functional cytokines were examined. TGFβ1 and IL-10 are the main effective factors that are secreted by Treg cells, and they play important roles in mediating suppressive activity. IL-17A is a proinflammatory factor that is produced specifically by Th17 cells. 18 TGFβ1 and IL-10 expression was decreased and IL-17A expression was increased in P. gingivalis-infected AS patients. These findings were consistent with our previous finding in apolipoprotein E knockout mice that were orally infected with P. gingivalis. 19 Inflammatory status, including anti-P. gingivalis IgG antibody titres and periodontal parameters, was higher in Pg-AS patients than in AS patients. Moreover, there was a positive correlation between Th17 cell levels, the Th17/Treg ratio, and P. gingivalis antibody titre. Our findings suggest that P. gingivalis may be associated with Th17/Treg imbalance in AS patients.
The differentiation of CD4+T cell subsets from naïve T cells is regulated by specific cytokines. IDO is a potential controlling factor that plays a role in controlling Th17 and Treg cell transitions through regulating Trp. IDO is mainly expressed in antigen-presenting cells. The main mechanism by which IDO regulates T cells is by inhibiting T cell proliferation and differentiation through regulating Trp metabolism. Trp is an essential amino acid for T cell differentiation. Kyn is a metabolite of Trp. Kyn can activate general control nonderepressible 2 to inhibit Th17 cell differentiation and induce Treg cell proliferation. 20 Currently, immune regulation by IDO in infectious and autoimmune diseases is well recognized.21,22 Animal experiments also showed that the atherosclerotic plaque area in IDO-deficient mice was increased and IL-10 expression in the spleen and lymph nodes was decreased. 23 Additionally, IDO-expressing aortic plasmacytoid dendritic cells may induce Treg cells and effectively slow the progression of AS. 24 IDO is an immunosuppressive factor and plays a protective role in AS. In our study, IDO activity was reduced in atherosclerotic patients, especially in P. gingivalis-infected atherosclerotic patients. There was a negative correlation between IDO and the Th17/Treg ratio in atherosclerotic patients. Regression analysis showed that IDO activity was negatively associated with the P. gingivalis antibody titre. Palm et al. demonstrated that P. gingivalis downregulated IDO mRNA expression in human gingival fibroblasts. Gingipain activity, which is a P. gingivalis virulence factor, is especially crucial in the process. 11 IDO is important in P. gingivalis-differentiated myeloid dendritic cells that drive T cell responses in vitro. 25 The plasma anti-P. gingivalis IgG titre represents the quantity of bacteria that is exchanged between bacterial biofilms and the bloodstream. Higher P. gingivalis antibody titres may increase the effect of bacteria on endothelial cells and promote plaque formation, which is a potential clinical marker for the risk of AS in periodontal patients.26,27 Elevated levels of antibodies against P. gingivalis indicate advanced periodontal disease as well as advancement. 28 Our study suggested that P. gingivalis may severely affect IDO activity, which ultimately induces Th17 cells and inhibits Tregs.
No significant differences were found between healthy controls and periodontitis patients. Wang et al. 29 showed that the Th17 cell percentage, IL-17 concentration, and IL-17 and RORrt mRNA expression were increased in gingivitis. No difference was found in Treg and FoxP3 mRNA expression. The role of glycolysis and amino acid and fatty acid metabolism was discussed in Th17/Treg balance. However, periodontitis pathogens were not involved in this process. Periodontitis is a chronic infectious disease that starts with inflammation of the gingiva and is characterized by alveolar bone resorption. The role of the main pathogens, such as P. gingivalis, should be considered in the context of periodontitis. However, the immune status in gingivitis and periodontitis was different, especially at different ages. 30 In our study, the Th17/Treg imbalance was associated with P. gingivalis in AS but not in healthy subjects. This finding suggests that the role of P. gingivalis in AS accelerates the process rather than causing its occurrence.
TGFβ1 and IL-10 are anti-inflammatory cytokines that are produced by inflammatory cells such as Tregs. Both Tregs and Th17 cells require TGFβ1 during the early stage of differentiation. Multiple studies have shown that TGFβ1 participates in the development of AS and has antiatherogenic effects in the early stages of the disease and proatherogenic effects in the later stages. 31 Disruption of TGFβ1 in T cells accelerates the lesion progression. 32 In our study, the TGFβ1 level was decreased in Pg-AS patients, suggesting a stronger inflammatory reaction in these patients. A lower TGFβ1 level suggested that P. gingivalis infection may impair TGFβ1 to inhibit Treg differentiation and promote AS. Additionally, the frequency of Th17 cells was increased, while TGFβ1 was decreased in Pg-AS patients. Bettelli et al. showed that CD4+ lymphocytes may achieve the Th17 cell phenotype in the presence of low concentrations of TGFβ1 and proinflammatory cytokines such as IL-6. 33 TGFβ1 alone induced FoxP3 expression and Treg cell development. However, proinflammatory cytokines may inhibit the induction of Treg cells and simultaneously promote Th17 cell differentiation. Overall, further decreasing the low levels of TGFβ1 and increasing levels of proinflammatory cytokines may promote Th17 cells, which supports our finding that TGFβ1 (a Treg-related cytokine) decreased and IL-17 (a Th17-related cytokine) increased.
There were some limitations in the present study. First, we analysed the frequency of Th17 cells and Tregs in blood. However, the absolute number of Th17 cells and Tregs was not measured. These numbers should be determined in a future study. Second, this study was conducted at one centre, and the population and sample size may introduce sample bias. With a relatively small number of participants, the severity or stage of periodontitis was not considered in this study. Third, in vitro studies are needed to confirm the mechanism by which P. gingivalis decreases IDO expression and converts Tregs into Th17 cells.
This observational study showed that P. gingivalis infection may reduce IDO activity and promote Th17/Treg imbalance during AS progression. IDO is a potential risk predictor for periodontitis-associated AS.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221080877 - Supplemental material for Th17/Treg balance and indoleamine 2,3 dioxygenase activity in periodontitis-associated atherosclerotic patients
Supplemental material, sj-pdf-1-imr-10.1177_03000605221080877 for Th17/Treg balance and indoleamine 2,3 dioxygenase activity in periodontitis-associated atherosclerotic patients by Jie Yang, Ting Hao, Yu Liu, Jin Huang, Wenlei Wu, Juan Wu and Weibin Sun in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
All authors declare that there is no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (82001111 to Jie Yang, 514721115 to Weibin Sun, 51972167 to Wenlei Wu) and the Jiangsu Province Natural Science Foundation of China (BK20200149 to Juan Wu).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
