Abstract
Objective
C-reactive protein (CRP) is a powerful predictor of and risk factor for cardiovascular disease. However, the relationship between CRP and sudden death (SD) is controversial. Therefore, we performed a meta-analysis to evaluate the association between CRP and SD.
Methods
We conducted a comprehensive search of the databases of PubMed, Web of Science, Embase, Cochrane Library, Wanfang, CNKI, China Biology Medicine disc, and Weipu. Two researchers independently screened the literature, extracted data, and evaluated the data quality. The overall effect size was meta-analyzed using Stata software version 12.0 (StataCorp, College Station, TX, USA).
Results
Twelve prospective studies involving 36,646 patients were included in the present meta-analysis. The data revealed that patients with higher CRP concentrations had a greater risk of SD (hazard ratio, 1.19; 95% confidence interval, 1.09–1.29). When the hazard ratio of SD was calculated by multivariate analysis of nine studies, CRP was confirmed to be an independent predictive factor for SD (hazard ratio, 1.05; 95% confidence interval, 1.03–1.07).
Conclusions
This meta-analysis confirmed that CRP is an independent predictor of SD. These results support the recommendation of recording the CRP concentration for risk assessment of SD in clinical practice.
Keywords
Introduction
Sudden death (SD) is one of the most serious human conditions. The etiologies of SD are divided into cardiovascular system causes, respiratory system causes, central nervous system causes, abdominal causes, endocrine system causes, iatrogenic causes, and miscellaneous causes. The main cause of SD worldwide is cardiovascular disease (CVD), accounting for approximately 90% of cases of SD. 1 Among all cases of SD, sudden cardiac death (SCD) accounts for approximately 85%. 2 Globally, four to five million people die of SCD per year. 3 In Western countries, SCD is the cause of approximately 20,000 deaths in Australia and 350,000 deaths in the United States every year. 4 SCD accounts for approximately 50% of all cardiovascular deaths, and most cases of SCD are associated with coronary artery disease (CAD).3,5 SD or arrhythmias account for almost 39% of total deaths among patients being treated with dialysis in the United States. 6 Many traditional risk factors, such as smoking, diabetes, blood pressure, and race, can lead to SD. 7
It is important to note that the definition of SD varies among the literature. The World Health Organization defines SD as death caused by natural disease within a short time in people who apparently appear to be in good health or basically stable condition. However, what duration from onset to death can be defined as SD? At present, there is no universally recognized standard for the specific duration. This duration is 6 hours as defined by the World Health Organization, whereas other researchers have defined SD as death that occurs within 1 hour after the onset of symptoms or in patients found dead within 24 hours.3,8
At present, the prevention of SD is a great challenge and involves recognizing and treating individuals at high risk. 9 The left ventricular ejection fraction is the only commonly used predictive indicator of SCD in the current guidelines.3,8 However, recent research has shown that heart rate variability is the most frequently analyzed index in the technical literature of cardiovascular risk stratification, and many models and methods have been developed.10,11 In addition, although the use of an implantable cardioverter defibrillator (ICD) for primary prevention reduces the incidence of SCD, such therapy is costly and associated with procedural complications, infections, device malfunctions, inappropriate discharges, and diminished quality of life.10,12,13 Furthermore, the effectiveness of the use of ICDs in patients undergoing hemodialysis is uncertain because few data can infer its potential hazards and benefits, and whether the setting of defibrillation thresholds needs to be improved remains unclear.9,14 Additionally, many noninvasive electrocardiogram-derived medical tests, such as Holter analysis, have also been suggested to assess risk stratification for SD in patients with CAD. 15 With respect to biomarkers, the guidelines only indicate that measurement of natriuretic peptides (brain natriuretic peptide or N-terminal prohormone of brain natriuretic peptide) can be useful by adding prognostic information to standard risk factors for predicting SCD in patients with structural heart disease. 3 Although these biomarkers can predict SD, many individuals who undergo SD are not identified through their analysis. 5
C-reactive protein (CRP), a nonspecific inflammatory marker, is not only a strong predictor of and risk factor for CVD but is also a valuable prognostic biomarker that is directly involved in the course of CVD, such as atherosclerosis. 16 Several recent studies showed that the serum CRP concentration was associated with an increased risk of SD.5,17,18 Some studies also demonstrated that CRP was related to adverse events, such as myocardial infarction, atrial fibrillation, hypertension, and type 2 diabetes mellitus. 18 However, the role of CRP in predicting the occurrence of SD remains controversial. Biasucci et al.19,20 found a significant association between the CRP concentration and malignant ventricular arrhythmias in their initial observation, but they did not observe a significant association between the CRP concentration and SD in their subsequent prospective study. Therefore, we conducted the present meta-analysis to provide more evidence of using the CRP concentration for SD prediction and improvement of SD risk stratification.
Methods
Search strategy
A comprehensive search was conducted of the databases of PubMed, Web of Science, Embase, and Cochrane Library according to the PRISMA guideline 21 until 9 November 2020. There were no language restrictions for retrieval. The following subject terms were combined and applied to the search: (“sudden death” or “sudden unexpected death” or “sudden cardiac death” or “SD” or “SCD”) and (“C-reactive protein” or “high-sensitivity C-reactive protein” or “CRP” or “hsCRP”). Moreover, we manually screened the references of the included studies for additional potentially eligible studies. The study selection process is shown in Figure 1.
Study selection process.
Inclusion and exclusion criteria
The included studies were required to meet the following criteria 1 : (1) the end point covered SD, and the relationship between SD and CRP was investigated 2 and (2) the study design was prospective. Studies were excluded if they met one of the following conditions 1 : (1) the data were incomplete or could not be transformed 2 or (2) the type of research was a letter, review, case report, animal experiment, commentary, or gray literature.
Data extraction
Identification of all studies and extraction of all related data were conducted by two independent authors (R.Z. and W.W.) who had undergone specialized training. The data were then reexamined by another investigator (W.W.). The extracted information was shown as follows: first author, publication year, study design, country, recruitment period, sex, age, number of patients, primary disease, number of SD cases, CRP detection method, follow-up, optimal CRP cut-off values, hazard ratio (HR) with 95% confidence interval (CI), and end point. The quality of the included studies was assessed using the Newcastle–Ottawa Quality Assessment Scale. A score of ≤5 was considered low quality, 6 or 7 was considered moderate quality, and 8 or 9 was considered high quality. Any disagreements were resolved through consensus.
Statistical analysis
Stata software version 12.0 (StataCorp, College Station, TX, USA) was used for the statistical analysis. The relationship between SD and CRP was assessed by calculating the HR with 95% CI. When authors had provided both multivariable and univariable analyses, the former was selected. Heterogeneity between studies was statistically assessed by the χ2 and I2 tests. When p < 0.10 or I2 > 50%, indicating significant heterogeneity among studies, we chose a random-effects model. When p > 0.10 and I2 < 50%, a fixed-effects model was used. A sensitivity analysis was also conducted to check the stability of the results by sequentially omitting each inclusive study. Additionally, we performed Begg’s test and a funnel plot to determine the presence of publication bias across studies. A two-sided p value of <0.05 was considered statistically significant.
In error, we did not prospectively register this trial. However, we retrospectively registered it at INPLASY (registration number INPLASY2021110074).
Results
Study characteristics
Twelve eligible prospective studies published from 2007 to 2019 were included in this meta-analysis. Among the 36,646 people included in these studies, SD occurred in 1532 (4.2%). Among the 12 studies, 1 was a multicenter cohort study involving 43 countries, 8 were studies from Western countries (Israel, Poland, Italy, Germany, Finland, and the United States), and 3 were studies from Eastern countries (including China and Japan). The sample size of each study ranged from 202 to 17,095. The mean/median follow-up time ranged from 15.9 to 157.2 months. Among the 12 eligible studies, 3 established a cut-off value of the CRP concentration and treated it as a dichotomous variable, whereas 6 set the CRP concentration as a continuous variable. According to the classification of primary disease, five studies explored the relationship between CRP and SD in patients with heart disease, six studies involved patients undergoing dialysis, and one study involved older adults. The detailed characteristics of the included studies are shown in Table 1.
Detailed characteristics of the studies included in this meta-analysis.

SD, sudden death; HR, hazard ratio; CI, confidence interval; CRP, C-reactive protein; M, male; F, female; NOS, Newcastle–Ottawa Quality Assessment Scale; CHD, coronary heart disease; NR, not reported; ELISA, enzyme-linked immunosorbent assay; SCD, sudden cardiac death; BMI, body mass index; LVEF, left ventricular ejection fraction; MI, myocardial infarction; HDL-C, high-density lipoprotein cholesterol; CVD, cardiovascular disease; LDL-C, low-density lipoprotein cholesterol; HCM, hypertrophic cardiomyopathy; ACS, acute coronary syndrome; HF, heart failure; CAD, coronary artery disease; TIA, transient ischemic stroke; eGFR, estimated glomerular filtration rate; VDRA, vitamin D receptor activator; NYHA, New York Heart Association.
Associations between CRP and SD
The present meta-analysis of 12 studies showed that patients with higher CRP concentrations had a higher risk of SD (HR, 1.19; 95% CI, 1.09–1.29; p ≤ 0.001; I2 = 73%) (Figure 2(a)). Because of the obvious heterogeneity among studies, we adopted a random-effects model and conducted a subgroup analysis. When the HR of SD was calculated by multivariate analysis of nine studies, CRP was confirmed to be an independent predictive factor for SD (HR, 1.05; 95% CI, 1.03–1.07; p = 0.001; I2 = 73.1%) (Figure 2(b)). Subgroup analyses were performed based on the primary disease, and the results showed that patients with cardiac disease and a higher CRP concentration had a greater risk of SD (HR, 1.24; 95% CI, 1.05–1.47). This trend was also observed in patients undergoing dialysis (HR, 1.20; 95% CI, 1.05–1.36) (Figure 3(a)). In addition, the subgroup analysis based on outcomes suggested that an elevated CRP concentration was associated with a higher risk of SD (HR, 1.16; 95% CI, 1.01–1.32; p = 0.126; I2 = 51.8%) and SCD (HR, 1.21; 95% CI, 1.08–1.37; p ≤ 0.001; I2 = 74.9%) (Figure 3(b)). Additionally, a further subgroup analysis indicated that that regardless of whether the CRP concentration was treated as a continuous variable or dichotomous variable, patients with an elevated CRP concentration had a higher risk of SD (HR, 1.15; 95% CI, 1.07–1.23 and HR, 2.77; 95% CI, 1.69–4.55, respectively) (Figure 3(c)).
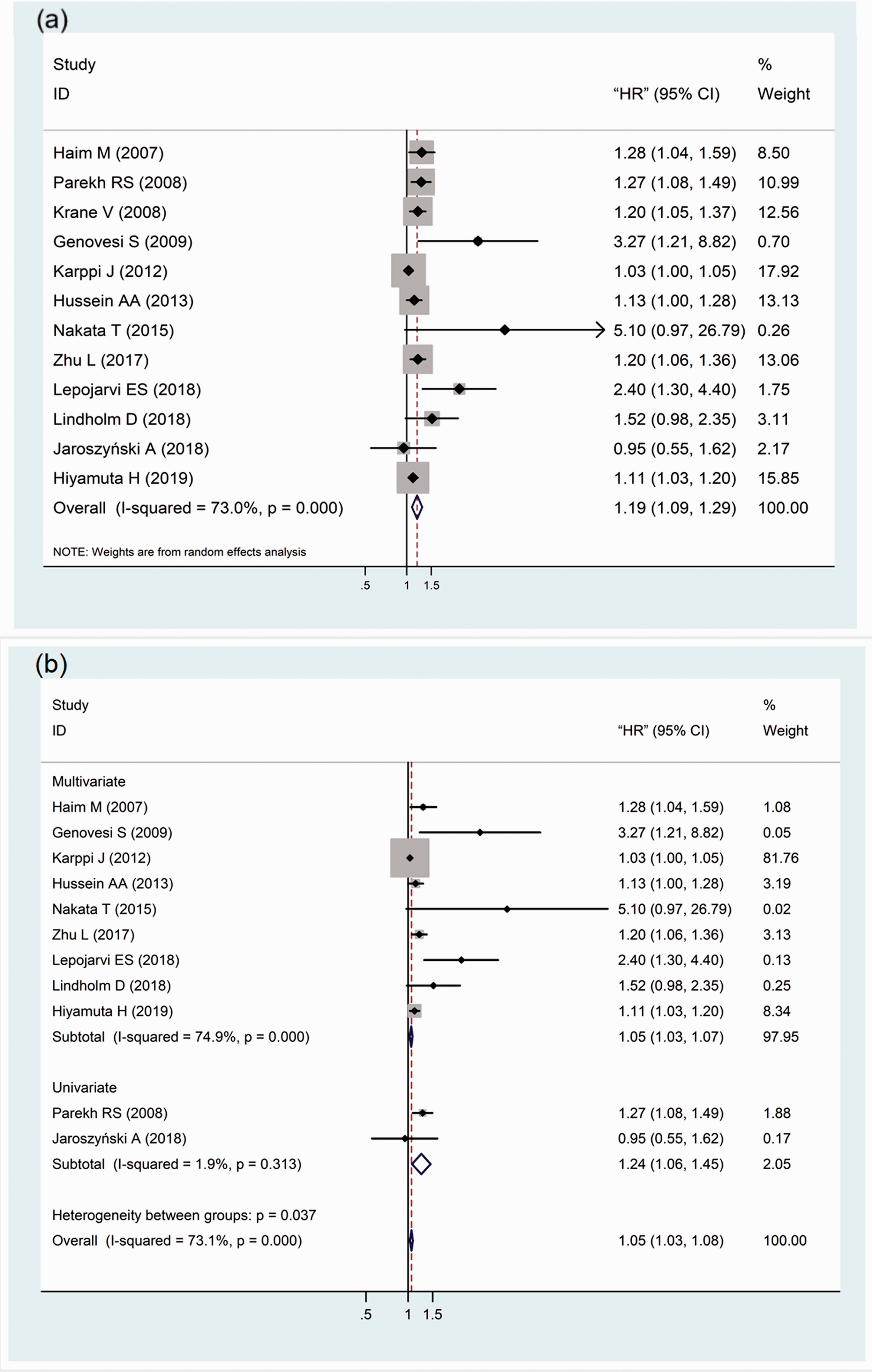
Forest plots for the association between CRP and SD. (a) Association between CRP and SD. (b) Subgroup analysis: HR calculated by multivariate analysis and univariate analysis. CRP, C-reactive protein; SD, sudden death; HR, hazard ratio; CI, confidence interval.

Forest plots for the association between CRP and SD. (a) Subgroup analysis of the association between CRP and SD according to primary disease. (b) Subgroup analysis of CRP and SD according to outcome. (c) Subgroup analysis of CRP and SD according to cut-off value of CRP. CRP, C-reactive protein; SD, sudden death; HR, hazard ratio; CI, confidence interval.
We also carried out a subgroup analysis according to the age of patients, follow-up time, country, and sample size (Table 2). The subgroup analysis according to age showed that an increased CRP concentration was associated with an increased risk of SD both in patients aged <60 and >60 years (HR, 1.21; 95% CI, 1.03–1.42 and HR, 1.23; 95% CI, 1.11–1.35, respectively). In the subgroup analysis according to follow-up time, however, statistical significance was only found in patients followed up for more than 36 months (HR, 1.19; 95% CI, 1.09–1.29). In the subgroup analysis according to country, the stratified analysis showed that an increased CRP concentration was associated with an increased risk of SD in both Western and Eastern patients (HR, 1.20; 95% CI, 1.02–1.35 and HR, 1.16; 95% CI, 1.02–1.32). Finally, the hierarchical analysis according to sample size showed that a sample size of >1000 could better reflect the predictive effect of CRP on SD (HR, 1.17; 95% CI, 1.07–1.28).
Associations between C-reactive protein and sudden death.

HR, hazard ratio; CI, confidence interval; NR, not reported.
Sensitivity and publication analysis
A sensitivity analysis was performed to assess the impact of each study on overall heterogeneity. This sensitivity analysis indicated low heterogeneity among the studies included in the meta-analysis (Figure 4). We also evaluated the publication bias of all included studies by Begg’s test and a funnel plot, and no publication bias was found (Figure 5). This indicated that the results of the meta-analysis were statistically reliable.

Sensitivity analysis for this meta-analysis.

Publication bias in the meta-analysis (Beggʼs test, p = 0.193).
Discussion
The prevalence of SD varied from 0.88% to 14.02% in the studies included in the present meta-analysis. The prevalence of SCD was 2.58% in patients with cardiac disease, and the prevalence of SD was 4.91% in patients undergoing dialysis. We infer that the reason for the higher prevalence of SD in patients undergoing dialysis might be related to the inflammatory state caused by the dialysis process and the limited application of invasive tests such as coronary angiography. Nevertheless, CRP is an inflammatory marker, and CRP testing is widely available and relatively inexpensive. CRP measurement is the preferred first test to support a diagnosis of inflammatory or infectious conditions. Furthermore, as a directly measurable marker, the CRP concentration peaks rapidly, approximately 48 hours after the inflammatory stimulus. When the stimulus for production stops, the CRP concentration decreases quickly. 22 All CRP assays measure the same protein, although laboratories differ in their measurement methods. CRP is most commonly measured in its high-sensitivity form, which is a reliable marker of subclinical and systemic inflammatory states. 23 High-sensitivity CRP is a designation given to laboratory assays that measure CRP concentrations of <3 mg/L. It can be used to stratify patients for CVD risk. In addition, CRP is more closely associated with the risks of plaque rupture and vascular thrombosis than with the extent of the underlying atherosclerotic burden. Moreover, it plays a critical role in understanding statin therapy as a treatment with both anti-inflammatory and lipid-lowering effects. 24
Most cases of SD occur in patients with CAD because CAD may increase the instability of unstable plaques in coronary arteries.5,25 Our results also showed that patients with cardiac disease who had a higher CRP concentration had a greater risk of SCD and that patients with CAD accounted for 80.4% of all patients with cardiac disease. When a high-risk vulnerable plaque ruptures, coronary artery thrombosis occurs, inducing a proarrhythmic effect and eventually resulting in SCD.26–28 Inflammation is considered a crucial factor in this process, and the serum concentration of CRP is an accurate objective index of the overall inflammatory activity throughout the body.29,30 Furthermore, Haverkate et al. 30 demonstrated that among patients with stable angina pectoris, those with a CRP concentration in the top quintile had an almost two-times higher risk of SD than those with a low CRP concentration. Moreover, another study also showed that the plasma concentration of CRP was directly and significantly correlated with an elevated incidence of SD. 31 Several studies have shown that CRP can promote cardiac fibrosis by nuclear factor kappa B signaling pathways in patients with hypertrophic cardiomyopathy and that myocardial fibrosis is one of the major determinants of SCD.18,32,33 Therefore, our results confirmed that CRP is a good predictor of SD in patients with cardiac disease.
Patients undergoing dialysis have a high risk of developing chronic inflammation secondary to the non-physiological essence of the dialysis process, vascular access, infections, and multiple comorbid states. 7 One might argue that uremia or the hemodialysis procedure causes a different inflammatory state corresponding to approximately 5- to 10-fold higher CRP concentrations than in the general population. 34 Dialysis sessions can lead to severe arrhythmias, and SD in patients undergoing dialysis is partly related to these patients’ cardiovascular lesions. 35 This increased SD risk may be mediated through the development of premature atherosclerosis and cytokine-induced plaque instability. 7 However, according to recent studies, the mechanism of SD in patients undergoing dialysis is different from that in patients with cardiac disease because the arrhythmia in patients undergoing dialysis is not sudden ventricular tachycardia or atrial fibrillation but instead bradycardia according.36–38 Additionally, inflammation directly induces fatal arrhythmia because of sympathetic overactivity and myocardial fibrosis even in the absence of atherosclerosis. 17 Nakata et al. 39 pointed out that a CRP concentration of >2.38 mg/mL may be the threshold for identifying high-risk SD cases, whereas Genovesi et al. 35 set the optimal CRP cut-off value at 1.9 mg/dL. This may be due to population differences in different regions. Parekh et al. 7 also showed that an increased CRP concentration was associated with a higher risk of SCD, even taking competing risks from other causes of death into account.
Strategies to decrease dialytic arrhythmic risk include adding sessions or prolonging dialysis times, routinely monitoring the serum potassium concentration before dialysis, minimizing exposure to extremely low-potassium and low-calcium dialysates as much as possible, and lowering dialysate temperatures. Other treatments, such as placement of ICDs, should be used with caution because of the lack of data regarding their potential hazards and benefits. Additionally, whether the setting of defibrillation thresholds needs to be improved and whether bradycardia should be backed up remain unclear.7,14 Moe et al. 24 suggested that management of bone and mineral disorders is also important for the prevention of SD.
Our meta-analysis demonstrated that CRP was a predictor of SD and that higher CRP concentrations were associated with an increased long-term risk of SD. Subgroups of patients categorized by age showed that regardless of the patients’ age and nationality, the association between CRP and SD was statistically significant, indicating the advantage of CRP measurement in predicting SD. These data can be explained by the fact that patients aged >60 years often have multiple chronic cardiovascular conditions and a higher burden of inflammation, which is related to structural myocardial changes.8,40 Patients aged <60 years are more likely to have acute coronary thrombosis, which is the main cause of SD.40,41 Our analysis also suggested that a follow-up time of at least 36 months is necessary before observing the potential predictive effect of CRP. This may be attributed to the increased number of SD cases appearing during longer follow-up. Moreover, the association between CRP and SD was observed only in studies with a sample size of >1000, which indicates that a large sample size is required to explore the relationship between CRP and SD.
The findings of our meta-analysis not only extend the clinical value of an increased CRP concentration from atherogenic implications to SD risk assessment but also strongly support the inflammatory hypothesis of atherothrombosis, which is persistently accelerated during progression of kidney dysfunction or due to hemodialysis procedures, resulting in unexpected cardiovascular death.
Limitations
Our meta-analysis has two main limitations. First, only 12 relevant studies were included. Thus, large-sample, high-quality cohort studies are required to confirm our conclusions, particularly with respect to the association between CRP and SD. Second, the definition of SD varies from country to country and lacks standardization. This suggests that health institutions in all countries should continue to strengthen information exchanges and clearly define SD to facilitate further research development in this field.
Conclusions
This is the first meta-analysis to explore the relationship between CRP and SD. Our results confirmed the predictive role of CRP on SD both in patients with cardiac disease and in patients undergoing dialysis. However, the identification of new risk markers of SD remains one of the most important research fields for the scientific community, and identification of more risk markers is needed to effectively prevent SD. Each country should generate a multidisciplinary expert network to allow the interchange of knowledge and establishment of a universal definition of SD.
Footnotes
Acknowledgements
R.Z. and W.W. contributed to the data extraction, statistical analysis, and drafting of the manuscript. J.L. and W.W. rechecked the results and revised the manuscript. Z.G. designed the study and took responsibility for the integrity of the data and accuracy of the data analysis. X.T., Y.H., Z.S., and L.Y. participated in the data extraction.
Declaration of conflict of interest
The authors have no conflicts of interest to declare.
Data availability statement
All the data used to support the findings of this study are included within the article. Please contact the corresponding author for data requests.
Ethics statement
The need for approval was waived because all data were from published literature and available from public databases. The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the Innovative Team of Jiangsu Province (from: Su Wei Science and Education 2017 [No. 1]).
