Abstract
A giant retrosternal goiter can lead to compression of vital organs in the mediastinum with high risk of acute cardiorespiratory decompensation. Additionally, patients with acromegaly are prone to developing severe airway obstruction and ventilation difficulties during anesthetic induction, leading to hypoxia and an increased partial pressure of carbon dioxide. Therefore, more comprehensive airway management strategies are needed. We herein describe a 57-year-old man with acromegaly and severe tracheal obstruction caused by a giant retrosternal goiter. He presented with a 1-week history of progressive dyspnea and was scheduled to undergo right lobe thyroidectomy and retrosternal goiter thyroidectomy. We created a comprehensive emergency plan for a difficult airway, including regional and topical anesthesia for awake endotracheal intubation, sevoflurane inhalation, small doses of midazolam and sufentanil to increase tolerance, self-made extended-length tracheostomy, video laryngoscope-assisted fiber-optic bronchoscopy, extracorporeal membrane oxygenation, and surgical tracheostomy. Importantly, tetracaine was inhaled through an atomizer, and a laryngotracheal topical anesthesia applicator was used to spray the larynx with 1% tetracaine to reduce stimulation during intubation. The giant goiter was successfully removed through the cervical approach. A carefully designed airway management strategy and close communication among a multidisciplinary operation team are the basis of perioperative anesthetic management for these patients.
Keywords
Introduction
A retrosternal goiter can cause compression of the airway or major blood vessels in the mediastinum, which poses a significant risk for cardiopulmonary complications. Catastrophic hypoxia, vascular complications, and cardiac arrest may occur during the perioperative period. 1 Moreover, patients with acromegaly have a hypertrophic tongue, a loose and collapsed throat and other tissues, a hypertrophic respiratory tract, airway lumen stenosis, and abnormal pulmonary function, readily leading to respiratory tract obstruction.2,3 Establishing an unobstructed airway and allowing good surgical access in the shortest possible time period are important in such patients. We herein describe successful anesthetic management of a patient with a retrosternal goiter and acromegaly who underwent total thyroidectomy (including the retrosternal portion).
Case presentation
A 57-year-old man with severe impairment of lung function of unknown cause, diabetes mellitus, and hypopituitarism was hospitalized because of a 1-week history of shortness of breath and sleep apnea. Physical examination on admission revealed that the right thyroid lobe was about 6 cm in diameter, was regular in shape, and had a smooth surface. The patient showed features of acromegaly, including a widened nose, protruding eyebrow arch, lengthened mandible, plump lips, and swollen hands and feet. Computed tomography (CT) showed a cystic mass (6.2 × 5.1 cm) in the right lobe of the thyroid gland protruding downward into the mediastinum. The trachea was pushed to the left with lumen restriction, and the esophagus was compressed (Figure 1). Additionally, two slightly low-density nodular shadows were seen in the right lobe of the thyroid gland. Ultrasound examination showed that the shape of the thyroid was abnormal. The largest left lobe was located in the middle part of the gland and measured approximately 23 × 14 × 16 mm. The largest right lobe was located in the lower part of the gland and behind the sternum and measured approximately 78 × 48 × 70 mm. The shape of the thyroid gland was regular, the boundary was distinct, and no obvious blood flow signal was detected. Pulmonary function examination via spirometry showed that the patient had very severe mixed ventilation dysfunction (maximal ventilatory capacity, 27.9 L/min [24% of predicted value]; forced expiratory volume in 1 s (FEV1), 0.93 L [30.7% of predicted value]; and FEV1/forced expiratory volume, 49.82% [59.5% of predicted value]). Examination also revealed severe obstruction of large airway airflow, moderate to severe obstruction of small airway airflow, abnormal increased airway resistance, abnormal decreased airway conduction, severe emphysema, moderately decreased diffusion function, severely decreased ventilation reserve function, and overventilation. Laboratory examination revealed the following: thyroglobulin, 601.70 µg/L (reference range, 1.4–78.0 µg/L); parathyroid hormone, 5.47 nmol/L (reference range, 0.03–2.47 nmol/L); growth hormone, 11.08 ng/mL (reference range, 1.60–6.90 ng/mL); adrenocorticotropic hormone, 79.76 ng/L (reference range, 5–78 ng/L); serum cortisol (8:00
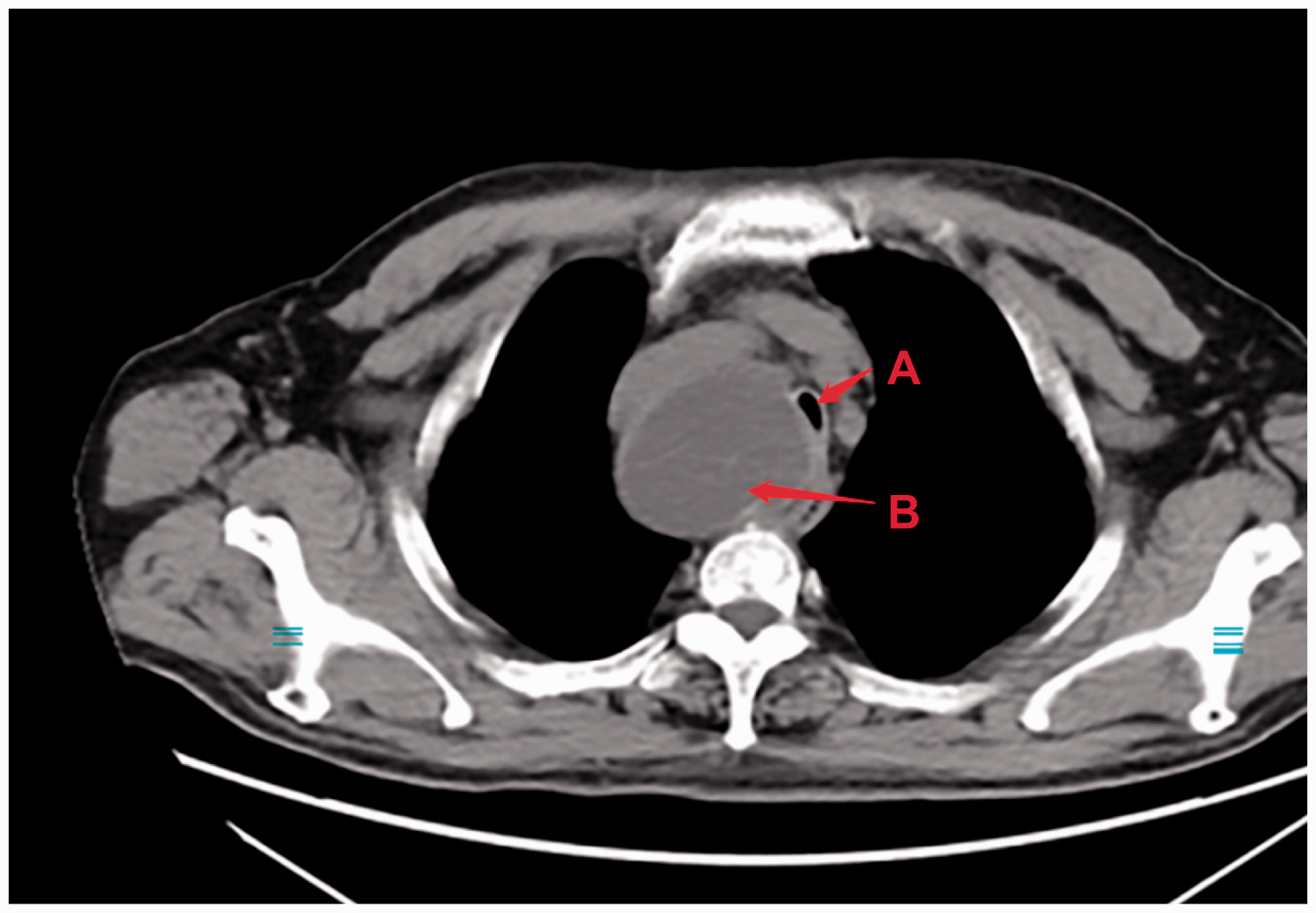
Preoperative computed tomography image of the patient showed that the giant goiter in the chest had resulted in severe tracheal obstruction. Arrow labeled “A”: tracheal obstruction. Arrow labeled “B”: goiter.
Laboratory data.

A comprehensive contingency plan was established before the operation, and the multidisciplinary operation team comprised thyroid surgeons, cardiothoracic surgeons, and anesthesiologists who focused on the management of difficult airway and acute cardiopulmonary compensation disorders after anesthetic induction. To ensure airway safety, surgeons able to perform surgical tracheostomy and clinicians on the extracorporeal membrane oxygenation (ECMO) team were on standby in case of an emergency (Figure 2). An airway management plan was prepared, including regional and topical anesthesia for awake endotracheal intubation, sedative and analgesic drugs, and video laryngoscope-assisted fiber-optic bronchoscopy (FOB) (Figure 2).

Airway management plan. First step: regional and topical anesthesia for awake endotracheal intubation. Second step: sevoflurane inhalation technique and a combination of various anesthetics. Third step, video laryngoscope-assisted FOB. The surgeons for surgical tracheostomy and the ECMO team were on standby.
The patient’s vital signs upon entering the operating room were as follows: blood pressure, 106/56 mmHg; heart rate, 76 beats/minute; and oxygen saturation, >90%. First, the anesthesia team provided the patient with sufficient aerosolization of local anesthetic on the tracheal mucosa: the patient was instructed to take a deep breath and inhale 40 mg of tetracaine through a MADgic® atomizer (Teleflex Inc., Wayne, PA, USA) for about 20 minutes (Figure 3(a)). In addition, a laryngotracheal topical anesthesia applicator with 1% tetracaine was used to spray the larynx to further reduce stimulation during intubation (Figure 3(b)). Second, we started sevoflurane inhalation at a concentration of 0.5% because of the anticipated difficult airway and increased the concentration every three to four respirations. After the sevoflurane concentration had been increased to 2%, the patient lost consciousness while maintaining spontaneous respiration. This sevoflurane inhalation technique helped us avoid the dangerous situation of an inability to ventilate. Next, midazolam (1 mg) and sufentanil (5 µg) were administrated intravenously to improve tolerance, alleviate discomfort, and achieve successful intubation. The patient underwent assisted manual ventilation when inadequate gas exchange occurred. Third, the patient was intubated with spontaneous respiration via video laryngoscope-assisted FOB. Importantly, based on the CT scan, we chose an endotracheal tube with a 5.5-mm inner diameter (ID) (Teleflex Inc.) to intubate the patient so that its tip could pass through the narrowest part of the trachea (8 mm as measured on the CT scan); however, the length of the tube was not sufficient to pass the teeth. Therefore, we made an extended endotracheal tube by connecting a 5.5-mm ID endotracheal tube (removing the connector) to the top part (about 5 cm) of a 6.5-mm ID tube using 3M™ Tegaderm™ Transparent Film (3M, Saint Paul, MN, USA) to tape them together (the connection was confirmed to be very tight after trying to pull the tubes apart with high force). In this way, the endotracheal tube could pass through the narrow part of the trachea compressed by the giant retrosternal goiter, and the tip of the endotracheal tube was located 29 cm from the teeth (Figure 4).

The patient was treated with sufficient tracheal surface anesthesia. (a) The patient inhaled tetracaine through an atomizer. (b) A laryngeal anesthesia tube was used to spray the larynx with 1% tetracaine. The red arrow shows the laryngeal anesthesia tube.

The self-made extended endotracheal tube with 5.5-mm inner diameter (ID). The extended tracheal tube was composed of a 5.5-mm ID endotracheal tube (connector was removed; arrow labeled “A”) and the top part (about 5 cm) of a 6.5-mm ID endotracheal tube (arrow labeled “B”).
We did not choose a long right-angle endotracheal tube because its angle would have prevented the bronchoscope from passing through. We also prepared an introducer and different sizes of endotracheal tubes (upscaled and downscaled) in case of an emergency. Notably, FOB was performed both before and after intubation. This examination helped us to confirm that the endotracheal tube could pass the narrowest part of the trachea, localize the tip position, and ensure that the tube could pass through the stenosis. The tube position was also confirmed by the end-tidal carbon dioxide concentration, which was within the normal range under mechanical ventilation (tidal volume, 400 mL; respiratory rate, 13 breaths/minute; and oxygen concentration, 56%). The front end of the endotracheal tube had just reached the surface of the cystic mass when the endotracheal tube was placed at 27 cm (Figure 5(a)). When the depth of the endotracheal tube was adjusted to 29 cm, FOB showed that it had crossed the mass, and the final location was 1 cm above the carina (Figure 5(b)). The mass was just the footprinted trachea; it was cystic mass in structure, surrounded by an outer membrane, and homogeneous in texture. Therefore, it was safe to leave the endotracheal tube in place. Because the mass was very large and had compressed part of the trachea near the carina, we maintained an adequate distance to keep the tip of the tube away from the carina. We used FOB to reconfirm the location of the endotracheal tube tip after surgical positioning.

Fiber-optic bronchoscopy-guided endotracheal intubation. (a) The front end of the endotracheal tube was placed on the surface of the cystic mass when the endotracheal tube was inserted at 27 cm. The red arrow shows the cystic mass. (b) When the depth of the endotracheal tube was adjusted to 29 cm, fiber-optic bronchoscopy showed that it had crossed the mass, with its final location at 1 cm above the carina. The red arrow labeled “1” shows the opening of the left main bronchus, and the red arrow labeled “3” shows the tracheal carina.
Anesthesia was maintained with 1.5% sevoflurane, dexmedetomidine infusion at 0.4 µg\kg\hour, propofol infusion at 3 mg\kg\hour, and remifentanil infusion at 0.2 µg\kg\hour. We did not use neuromuscular blocking drugs to avoid the influence of muscle relaxation on the intraoperative neuromonitoring of the recurrent laryngeal nerve (RLN) during thyroid surgery. The operation was successfully completed in 1.5 hours. Additionally, we used FOB for the pre-extubation airway examination and ensured that the compression effect of the mass had been removed. An airway exchange catheter was kept in place for protected extubation in case of a difficult airway. The patient was safely transferred back to the ward after he was fully awake. He had no post-thyroidectomy airway damage due to tracheomalacia or laryngeal edema. Histopathological examination confirmed that the goiter was a nodular goiter with a hematoma. After treatment in the ward, the patient was safely discharged on the third day postoperatively.
Discussion
In the present case, a giant goiter compressed the trachea and damaged the patient’s ventilatory function. Respiratory anatomic abnormalities and stenosis are often present in patients with acromegaly. Thus, tracheal intubation may be difficult. These patients are prone to developing severe airway obstruction and ventilation difficulties during anesthetic induction. Our patient had both a retrosternal goiter and acromegaly; thus, he had more risk factors for airway obstruction. Although both dexmedetomidine and sufentanil are effective as adjuvants for awake fiber-optic intubation under airway topical anesthesia combined with midazolam sedation, respiratory depression is still a potential risk when using a sufentanil regimen. 4
Comprehensive preoperative assessments, including medical history collection, physical examination, and imaging examination, are considered very important for identifying patients at risk of respiratory complications. It is necessary to determine whether the airway is narrow and collapsing; additionally, inspiratory respiration difficulty related to body position changes indicates the possibility of airway softening. CT studies (tracheal compression of >50% or pericardial effusion) and pulmonary function tests are important methods for identifying respiratory risk factors.5,6 Chest CT shows whether tracheal and bronchial compression is present and facilitates accurate measurement of the airway diameter. It is also helpful to determine the exact level and degree of tracheobronchial tree compression. 7 Injury to the RLN often leads to abnormal opening and closing of the glottis, which is easily ignored as an important cause of postoperative asphyxia. Attention should be paid to the presence of hoarseness and water choking before the operation for early detection of RLN injury. If preoperative endoscopy can be performed, injury to the RLN will be more apparent. The presence of RLN injury further increases the risk of surgical injury. 8 Monitoring the RLN can improve the recognition rate of RLN injury, relieve the cause of injury, allow for timely implementation of remedial measures, and reduce the incidence and severity of postoperative complications. A tracheostomy is a medical procedure—either temporary or permanent—that involves creating an opening in the neck to place a tube into a person’s windpipe. The tube is inserted through an incision in the neck below the vocal cords, allowing air to enter the lungs.
During intubation in the present case, the patient’s airway was exceptionally narrow, with the narrowest part measuring only 3 mm (lateral axis) × 10 mm (vertical axis). Any stimulation may have caused a tracheal spasm. Intubation in such cases must be successful on the first attempt, and the distal end of the cannula must cross the tracheal stenosis. Matters needing attention during intubation in our case were awake endotracheal intubation and video laryngoscope-assisted FOB. Hypertrophy and stenosis may exist in the glottis and subglottis of patients with acromegaly. A tracheal catheter with a slightly smaller inner diameter should be selected to pass through the tracheal stenosis and reduce damage to the goiter. If the endotracheal tube can be advanced to the distal uncompressed airway in the absence of occlusion, oral optical fiber intubation can be considered while maintaining spontaneous respiration; this will help to prevent complications during anesthesia. In the present case, a self-made extended 5.5-mm ID endotracheal tube was used for intubation, and FOB confirmed that the tube passed through the tracheal stenosis.
FOB can be used to exclude tumor invasion to the tracheal wall and evaluate the risk of tracheal compression, softening before induction, and scheduled surgery. More importantly, it can help guide the endotracheal tube through the occlusion site. 9 Researchers have found that cross-sectional CT imaging of the tracheal deviation does not seem to affect tracheoesophageal compression symptoms. 10 This may be due to the cystic and substantial nature of benign goiters, causing the tube to pass the tracheal stenosis and arrive at the distal trachea. This may also explain why our patient’s symptoms were not as severe as suggested by CT. A mediastinal mass may be a tumor, cyst, or aneurysm and may originate from any component of the pleura, lung, or anterior mediastinum. Anesthetic precautions with an anterior mediastinal mass may vary according to the individual patient’s anatomical structure. 6 Even when spontaneous ventilation is maintained, it is challenging to maintain airway patency and prevent a severe reduction in oxygen saturation in these patients, especially during the induction and extubation processes.11,12 The use of ECMO in patients with a giant retrosternal goiter is another option. For patients with severe airway obstruction caused by a mediastinal mass, ECMO can provide necessary tissue oxygenation and secure a definitive airway in a controlled environment. 11
Conclusions
We have herein described successful anesthetic management in a patient with high-risk airway obstruction accompanied by a giant retrosternal goiter and acromegaly. A careful airway management plan involving elements such as ECMO team and surgical tracheostomy standby, a combination of various anesthetics, creative application of a self-made extended endotracheal tube, and video laryngoscope-assisted FOB is of critical importance to prevent death due to difficult airway management.
Footnotes
Authors’ contributions
XHS, CC, and RHZ conducted a review of the literature. XHS, CC, CG, CLJ, and TZ prepared the body of the manuscript. CC, CG, CLJ, and TZ critically reviewed the publication. All authors endorsed the final form of the manuscript.
Consent for publication
The patient provided written consent for the publication of this case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics approval
This report was approved by the Ethics Committee of West China Hospital, Sichuan University.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
