Abstract
Catheter ablation of accessory pathways can be challenging depending on the location of these pathways, and accessory pathways are rare through the aortic cusps. We report a patient who underwent radiofrequency catheter ablation for manifestation of a left anterior accessory pathway from the left coronary sinus of Valsalva near the aortic–mitral continuity. Anterior accessory pathways can be safely and effectively ablated from the aortic cusps with favorable long-term outcomes.
Keywords
Introduction
Accessory pathways (APs) crossing the tricuspid or mitral annulus and inserting into the atrium and ventricle are remnants of early atrioventricular (AV) connections. These aberrant pathways occur because of incomplete embryological development of AV annuli and failure of fibrous separation between atria and ventricles. Catheter ablation has become the preferred treatment for patients with manifestation of a left anterior AP. 1 We report a case of successful ablation of a left anterior AP from the left coronary cusp (LCC) near the aortic–mitral continuity.
Case report
A 56-year-old man was referred to our institute with recurrent palpitations. A physical examination, chest radiography, and echocardiography showed no evidence of structural heart disease. A 12-lead electrocardiogram performed at the time of admission showed a regular sinus rhythm with ventricular pre-excitation (Figure 1) and wide complex tachycardia. The delta wave was positive in leads I, II, III, and aVF and in all precordial leads, and negative in aVL and aVR. This finding indicated a left anterior AP.
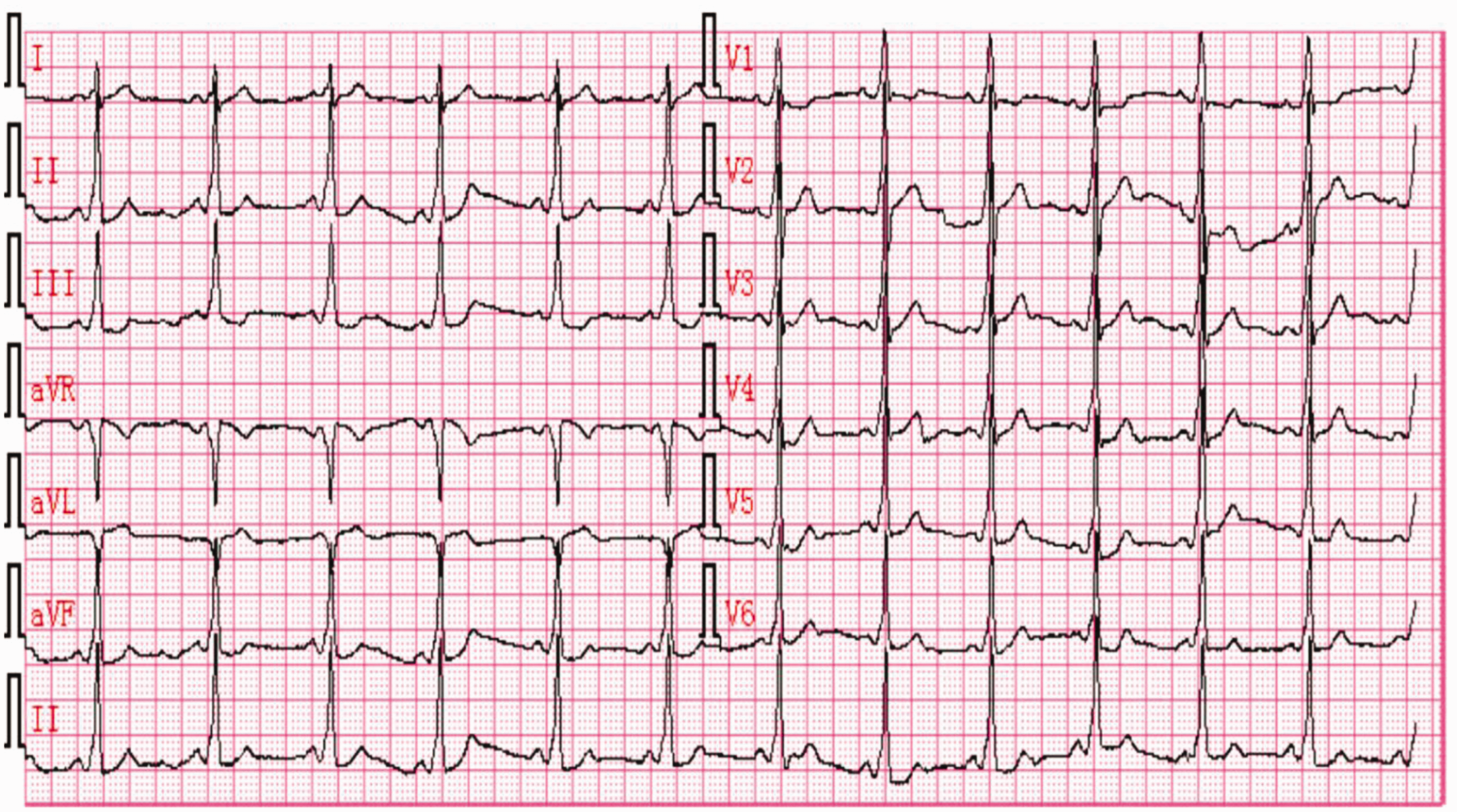
Twelve-lead electrocardiogram showing sinus rhythm with manifestation of pre-excitation. The delta wave morphology indicates a left anterior accessory pathway
An electrophysiological study was performed after obtaining informed written consent. A decapolar catheter was advanced into the coronary sinus and a quadripolar catheter was positioned into the right ventricular apex (RVA).
There was no decremental ventriculoatrial conduction or gap phenomenon of the AP during RVA pacing from a pacing cycle length of 600 to 300 ms. Programed atrial stimulation showed an anterograde AP refractory period of 300 ms. Incremental atrial pacing was followed by progressive lengthening of the AV interval and an increased degree of pre-excitation, without decremental AP conduction. Wide complex tachycardia was induced when the QRS became fully pre-excited during the programed incremental atrial stimulation, and no AV nodal jump was observed.
At baseline and during tachycardia, the coronary sinus was activated from distally to proximally, with the earliest ventricular activation located at the anterolateral wall of the mitral annulus. In this procedure, a 3.5-mm, externally irrigated radiofrequency catheter (Biosense Webster, Diamond Bar, CA, USA) was introduced into the left ventricle via a retrograde aortic approach to map the earliest ventricular activation at the anterolateral part of the mitral annulus near CS1, 2. This event was preceded by onset of a delta wave. Subsequently, we attempted an alternative transseptal approach. The pre-excitation was not eliminated when we applied radiofrequency energy directly at the earliest ventricular activation site that preceded onset of the delta wave only by 2 ms in the anterior part of the left atrium (Figure 2a). Further analysis showed that the locations of the left AP pathway and the aortic sinus of Valsalva were close anatomically and radiologically. After confirming the ostial locations of both coronary arteries and identifying the contour of the aortic sinus of Valsalva by injecting contrast agent, we mapped the aortic cusps. Despite fusion of the atrial and ventricular potential and ventricular activation preceding onset of the delta wave by only 22 ms, the target site in the LCC near the aortic–mitral continuity showed the earliest ventricular activation (Figure 2b). Additionally, no His potential was recorded at this site. The delta wave disappeared within 2 s of radiofrequency ablation at this site and disappeared after 30 s at 25 W under continuous monitoring using a CARTO-3 dimension system (Biosense Webster) and fluoroscopy (Figure 3).

(a) Intra-cardiac electrograms from the mapping catheter at the “earliest” site on the mitral annulus showing that a ventricular potential preceded onset of the wave by 2 ms. CARTO left lateral view and left anterior oblique view post-ablation showing the site of ablation (red dots) in the left atrium aspect of the mitral annulus. (b) The ventricular potential from the mapping catheter at the earliest site preceded onset of the wave by 22 ms with fused “A” and “V”. CARTO left lateral view and left anterior oblique view post-ablation showing the target ablation site (blue dot) in the left coronary cusp near the aortic–mitral continuity

Left panel: an electrocardiogram that was conducted following successful ablation shows sinus rhythm with a normal PR interval without pre-excitation. Right panel: radiograms in the right anterior oblique (30°) and left anterior oblique (45°) projections show the ablation sites. A distal electrode of the ablation catheter was positioned at the base of the left coronary sinus of Valsalva
With incremental atrial pacing and a His bundle potential followed by atrial potentials in response to ventricular pacing, the AV nodal Wenckebach block cycle length was 280 ms, which matched that observed before ablation. No tachycardia was induced even during infusion of isoproterenol as high as 5 µg/minute. The patient was discharged without medication and has remained well with no pre-excitation (Figure 3) and no recurrence of tachycardia during a 12-month follow-up.
Discussion
We report a rare case of AP connection where the successful site of ablation was located inside the LCC near the aortic–mitral continuity region. We were not able to locate the AP potential at the target. However, the presence of a local intra-cardiac potential at the successful site showed continuous electrical activation with atrial activity followed by an early and fragmented ventricular potential preceding onset of the delta wave.
Godin et al. 2 reported AP ablation in the LCC. In their case, there was antegrade pre-excitation during sinus rhythm and the AP participated in an orthodromic AV reciprocating tachycardia. An unusual decremental AP mimicking tachycardia of the left ventricular outflow tract was also successfully ablated in the LCC. 3 However, in this case, no retrograde conduction via the AP was documented. A third case of successful ablation of AP inside the LCC was reported where an AP was located at the aortic–mitral continuity area. 4 In our case, an AP located at the aortic–mitral continuity was non-decremental with antegrade conduction during sinus rhythm and involved in antidromic AV reciprocating tachycardia. Additionally, the locations of the left AP pathway and the aortic sinus of Valsalva were close anatomically and radiologically, as reported previously. 5 Catheter ablation of the AP from the aortic cusps was also recently reviewed. 6 This review included 16 case studies and 36 patients in whom 94.5% of the AP was ablated either in the noncoronary cusp or right coronary cusp, and only 5.5% of the AP was ablated in the LCC.
The LCC is related to the posterior wall of the right ventricular outflow tract, but is located more posteriorly, and is continuous with the anterior leaflet of the mitral valve as an aortomitral continuity. The anatomical substrate of the AP found in our patient remains unclear. An AV AP is rarely found in the area of fibrous continuity between the aortic and mitral valves. This is because this area usually contains a wide gap between the atrial and ventricular myocardium to accommodate the aortic outflow tract.7,8 Anatomically, the posterior part of the aortic valve is composed of the LCC and the noncoronary cusp, and is surrounded by the inter-atrial septum, which gives rise to the septal mitral leaflet. These are collectively called the aortic–mitral continuity. Additionally, expansion of fibrous tissue at either extreme of the area of continuity forms the right and left fibrous trigones. However, fibrous tissue from each of the fibrous trigones is not continuous around the orifice9,10 and disjunction at the annulus is found as frequently in normal valves as in prolapsed valves. 11 At the base of the LCC, a crescent of ventricular musculature extends into the aorta between the leaflets of the aortic and mitral valves.3,12,13
Although there is no evidence of connection between the LCC and atrial myocardium in histological examinations, successful ablation of atrial tachycardia inside the LCC demonstrates their proximity. 14 Nevertheless, the atrial and ventricular myocardium might be connected across the anterior region of the mitral annulus near the aortic–mitral continuity. Findings from our case suggest that an AP located in this area can be successfully ablated from the LCC.
In conclusion, an AV AP can be present and successfully ablated at rare locations, such as the left aortic sinus of Valsalva near the aortic–mitral continuity, after failure to achieve ablation along the mitral annulus.
Footnotes
Ethics statement
No ethical approval was required. The authors obtained written informed consent for publication from the patient.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
