Abstract
Objective
To present the authors’ experience of Mahaim-type accessory pathways (MAPs), focusing on anatomic localizations.
Methods
Data from consecutive patients who underwent electrophysiological study (EPS) for MAP ablation in two tertiary centres, between January 1998 and June 2020, were retrospectively analysed.
Results
Of the 55 included patients, 27 (49.1%) were male, and the overall mean age was 29.5 ± 11.6 years (range, 12–66 years). MAPs were ablated at the tricuspid annulus in 43 patients (78.2%), mitral annulus in four patients (7.3%), paraseptal region in three patients (5.5%), and right ventricle mid-apical region in five patients (9.1%). Among 49 patients who planned for ablation therapy, the success rate was 91.8% (45 patients).
Conclusion
MAPs were most often ablated at the lateral aspect of the tricuspid annuli, sometimes at other sides of the tricuspid and mitral annuli, and infrequently in the right ventricle. The M potential mapping technique is likely to be a useful target for ablation of MAPs.
Keywords
Introduction
Stanley Kent, an English physiologist, and Wilhelm His Jr, a Swiss cardiologist and anatomist, are well-known for conducting successful and inspiring studies into atrioventricular (AV) conduction and accessory pathways (AP), dating back to 1893.1,2 While Kent had suggested that multiple myocardial connections were present across AV junctions of the normal heart, His described a solitary muscular connection extending from the atrial to ventricular septum. The studies of His were validated over time, with the normal heart shown not to possess multiple myocardial connections across AV junctions. 3 Rather than multiple pathways, Kent had shown node-like structures that did not cross the insulating plane, which he had still believed to provide normal AV conduction.4,5 These findings allowed cardiologists to predict that arrhythmias now defined as ventricular pre-excitation, are the consequence of such multiple myocardial AV connections being present. Even to date, accessory muscular connections that are presumed to be the anatomical substrates of Wolff–Parkinson–White syndrome, are sometimes defined as ‘bundles of Kent’, despite the fact that the connections have been demonstrated to have no anatomic similarity to the structures demonstrated by Kent.2,6 With the use of programmed electrical stimulation, electrophysiological studies in the late 1960’s and early 1970’s revealed that Wolff–Parkinson–White syndrome was one variant of ventricular pre-excitation regarding the structure and location of alternative potential anatomical pathways between the atrial and ventricular muscle masses. 7 A 1975 review by a group of European anatomists and pathologists proposed nomenclature for such potential pathways. 8 While many of these unusual paths remain controversial in terms of their anatomy, the names used for their descriptions have become somewhat deficient. In general, the pathways produce the so-called ‘Mahaim’ pre-excitation. 9 Initially, it was believed that pre-excitation characterised by decremental conduction was produced by connections between the normal AV conduction axis and the crest of the ventricular septum, as described by Mahaim and his colleagues.10,11 However, it was later shown that such pre-excitation, developed from a pathway revealing decremental conduction, could also be constructed by a pathway of atrial origin with morphological characteristics of the structures shown by Kent.12,13 APs that arise from atrial origin were not mentioned by Mahaim or other European morphologists at that time.
Mahaim-type accessory pathways (MAPs) usually originate within the lateral tricuspid annulus and cross the AV junctions, and display decremental conduction, inserting into the distal right ventricular free wall.12–18 Most reported decremental conducting MAPs are right-sided. However, paraseptal and left-sided APs are also reported.19–27 The left-sided decrementally conducting APs originate within the mitral annulus and their distal end inserts into the left ventricle, while the origin and insertion point of paraseptal APs may vary according to its septal side.
Radiofrequency (RF) catheter ablation is the exclusive treatment option for MAPs, offering a definitive therapy with a high success rate and minimal morbidity,14,15 although few case series are noted in the literature.20,28–30 The aim of the present study was to describe the present authors’ ablation experience with this unusual pre-excitation syndrome, focusing on the anatomic localizations of MAPs.
Patients and methods
Study population
In this multicentre retrospective analysis, consecutive patients who underwent electrophysiological study (EPS) and who were evaluated and diagnosed with MAPs, between January 1998 and June 2020 in the Cardiology Departments of Gulhane Research and Training Hospital and Liv Hospital, were enrolled. Data regarding epidemiological characteristics, clinical features, medical treatments, baseline electrocardiogram (ECG)/electrophysiological (EP) characteristics, EP properties of MAPs, and results, were extracted from medical records and analysed. There were no exclusion criteria. The Clinical Research Ethics Committee of Gulhane Training and Research Hospital approved the study (approval No. 2020-432). Requirement for informed patient consent for the study was waived due to the retrospective nature of the study and the use of anonymised patient data. The reporting of this study conforms to STROBE guidelines. 31
EPS protocol
All patients, or their legal proxy, provided informed consent for EPS, which was conducted in the fasting state with antiarrhythmic drugs discontinued at least one week before the procedure. The procedure was performed under local anaesthesia, and intravenous (i.v.) sedative drugs were used whenever necessary. Patients underwent ablation procedures under local anaesthesia in a fasting state. Drugs administered for the procedure included propofol, midazolam and fentanyl. Three catheters were introduced through the right femoral vein, and positioned at the high right atrium (HRA), coronary sinus, and His region. The catheter at the His or HRA position was shifted to the right ventricle when necessary. For left-sided procedures, a retrograde transaortic approach was performed. Standard 12-lead ECGs were recorded along with bipolar intracardiac ECG filtered at 30–500 Hz. In addition to intracardiac recordings, 12 surface ECG leads were recorded simultaneously. Programmed electrical stimulation was performed from the right atrium and right ventricular apex to determine the patterns of ventricular antegrade conduction and atrial retrograde conduction. The refractory periods of the AV node and AP were noted. The relationship between Mahaim potential, right bundle, and His bundle was evaluated during sinus rhythm and after tachycardia induction. Programmed ventricular extra stimulus was used for testing V–A conduction both in basal circumstances and after an i.v. atropine bolus (1 mg or 0.02 mg/kg body weight).
The presence of MAP conduction was confirmed with the following criteria: (1) baseline sinus rhythm without pre-excitation, minimal pre-excitation or manifest pre-excitation with left or right bundle branch block morphology; (2) presence of progressive ventricular pre-excitation during programmed atrial stimulation associated with an increase in the AV interval along with shortening in the His–ventricular (HV) interval; (3) absence of retrograde conduction over the AP during ventricular stimulation; and (4) reversal of His bundle activation during both supraventricular tachycardia and antegrade pre-excitation, indicating retrograde His bundle activation.
Significant findings relating to MAPs include 12-lead ECG findings of minimal pre-excitation as subtle abnormalities, suggesting pre-excitation is a QRS complex width within the normal range but with a short HV interval and an rS configuration in lead III. 29
Mahaim fibres originate from several different anatomic localizations of the heart. Right-sided decrementally conducting MAPs connect the right atrium or AV node and right ventricle free wall. 32 The distal site of AP insertion is near the insertion of the moderator band into the anterior papillary muscle. 33 On the other side, the left-sided decrementally conducting APs connect the mitral annulus and the left ventricle, while the origin and insertion point of paraseptal APs depend on its septal side.19–27
Ablation procedure
After MAP assessment using the EP properties described above, an RF Marinr™ 7F, 4-mm deflectable tip ablation catheter (Medtronic, Minneapolis, MN, USA) was introduced through the femoral vein for mapping and ablating the AP. Mapping began at the coronary sinus ostium level. The tricuspid annulus was mapped clockwise in the left anterior oblique and right anterior oblique fluoroscopy projections. At the Mahaim potential recording site, RF energy was delivered with an Atakr™ RF generator (Medtronic).
Mahaim potentials were localized through the coronary sinus at the lateral mitral annulus site in some unusual cases. These patients underwent ablation procedures with a transaortic retrograde approach.
A stimulation study was performed after RF ablation to establish the absence of antegrade conduction over the MAP and non-inducibility of the tachycardia. The absence of AP conduction and inducible tachycardia during a vigorous stimulation protocol with isoproterenol infusion for 30 min, after successful RF ablation, was considered a procedural success.
Patients were hospitalized for at least one day after undergoing RF ablation. The following day, they received 100 mg oral aspirin, and were discharged. Oral aspirin treatment at 100 mg daily was continued for one month. Antiarrhythmic drugs were not prescribed for successful procedures.
Statistical analyses
In this descriptive study, categorical variables are presented as absolute numbers and percentages, and continuous variables are presented as mean ± SD. Statistical analyses were performed using SPSS software for Windows®, version 20.0 (IBM, Armonk, NY, USA).
Results
The records of 7704 patients who underwent EPS were evaluated. Among the 7704 cases, 3837 patients (49.8%) were diagnosed with AV-nodal re-entrant tachycardia (AVNRT), 2195 (28.5%) with atrial flutter/fibrillation/tachycardia, 891 (11.6%) with manifest APs and 791 (10.3%) with concealed APs. Out of 1682 patients with APs, 55 (3.3%) were diagnosed with Mahaim fibre-mediated pathway, and were included in the present study.
Baseline characteristics of the 55 included patients are summarised in Table 1. Of the 55 patients, 27 (49.1%) were male, and the mean age was 29.5 ± 11.6 years (range, 12–66 years). A total of 47 patients presented with documented wide QRS tachycardia that was of either left bundle branch morphology (43 patients [78.2%]) or right bundle branch morphology (four patients [7.3%]). In contrast, seven patients (12.7%) presented with palpitations, and one patient (1.8%) with syncope. Only half of the patients had been prescribed antiarrhythmic drugs before RF ablation therapy. A resting 12-lead ECG revealed a minimal pre-excitation during sinus rhythm in 17 (30.9%) of the patients, while the remaining patients had normal sinus rhythm without pre-excitation (Figure 1). Among 55 patients, eight patients (14.5%) had rS pattern in lead III combined with the absence of septal q wave in lead I. In five patients, Ebstein’s disease was diagnosed in preprocedural examinations.
Baseline demographic and clinical characteristics of 55 patients with Mahaim-type accessory pathways who underwent electrophysiological study.
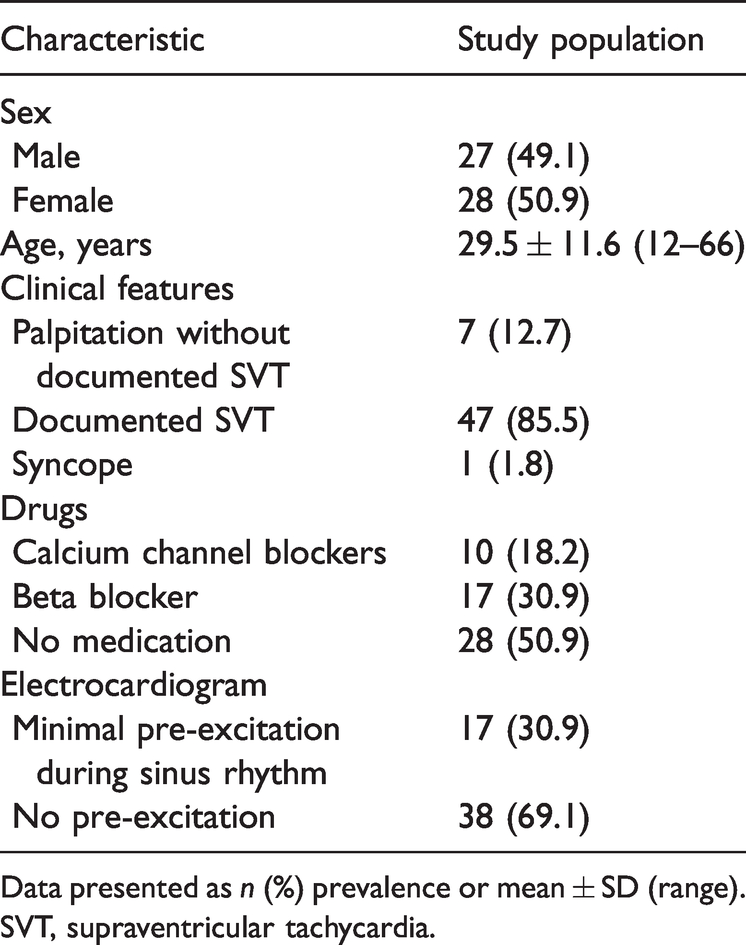
Data presented as n (%) prevalence or mean ± SD (range).
SVT, supraventricular tachycardia.

Representative electrocardiogram from a patient with Mahaim-type accessory pathway showing: (I) subtle pre-excitation (red arrow); (II) normal range width QRS complex (94 ms); (III) rS configuration in lead III (black arrow); and (IV) absence of septal q wave in lead I (asterix).
In terms of treatment, MAPs were ablated at the tricuspid annulus in 43 patients (78.2%), mitral annulus in four patients (7.3%), paraseptal region in three patients (5.5%), and right ventricle midapical region in five patients (9.1%), respectively (Figure 2).

Representative Mahaim-type accessory pathway (MAP) ablation targets in the present study population of 55 patients. Solid circles show radiofrequency ablation targets around the tricuspid and mitral anulus. CS, coronary sinus; H, His; MV, mitral valve; TV, tricuspid valve. *MAP in the right ventricular free wall region.
After diagnostic EPS, six patients out of 55 declined ablation therapy, of whom, two had nodofascicular Mahaim, and thus, a risk for complete AV block subsequent to RF ablation. Of the 49 patients who planned ablation therapy, 45 were ablated successfully (success rate 91.8%). In four patients, the APs could not be ablated due to atrial fibrillation (two patients) and prolonged procedure time (two patients). In the two patients with prolonged procedure time, the APs were originating from the right ventricular apical region. Ablation of four patients with MAPs originating from the mitral annulus site was successful using the retrograde aortic route without necessitating septal puncture. Long sheaths were routinely used in most cases (81.8%) to facilitate the stability of the ablation catheter. Representative intracardiac recordings are shown in Figures 3–5. Representative fluoroscopic images from two patients during EPS are shown in Figure 6.

Representative intraoperative electrocardiogram from a patient with Mahaim-type accessory pathway (MAP) showing typical wide QRS complex tachycardia with left bundle branch pattern. The tricuspid lateral MAP was ablated.

Representative electrocardiogram from a patient with Mahaim-type accessory pathway. The catheter at the lateral tricuspid annulus demonstrating the M potential preceding the QRS complex by 20 msn. Successfully delivered radiofrequency eliminated accessory pathway conduction.

Representative electrocardiogram from a patient with Mahaim-type accessory pathway (MAP). Programmed atrial extra stimulus produced right bundle branch aberrancy. Detailed mapping revealed left posteroseptal MAP. The AH interval was increased, whereas the HV interval was shortened concomitant to prolongation of the AV interval.

Representative fluoroscopic images of successful ablation sites in two patients with Mahaim-type accessory pathway (MAP), showing: (a) a patient with right-sided AP in whom the ablation (Abl) catheter was delivered through the right femoral vein at the lateral tricuspid annulus as standard approach (RAO view); and (b) a patient with left-sided AP in whom a retrograde transaortic approach was performed for reaching the lateral mitral annulus (LAO view). AP, accessory pathway; CS, coronary sinus catheter; HRA, high right atrium.
Nine patients had other types of supraventricular tachyarrhythmia in addition to Mahaim during EPS. Six had other types of atrioventricular re-entrant tachycardia, two had AVNRT, and one had atrial tachycardia. All the accompanying tachyarrhythmias were ablated in the same session successfully.
Atrial fibrillation developed in two patients (3.6%) during the RF procedure. While direct cardioversion was performed in one of the two patients, medical cardioversion was performed in the other patient. Sinus rhythm was restored in both patients. No other complications developed in patients who underwent RF ablation. Electrophysiological data from patients analysed in the current study are summarised in Table 2.
Electrophysiological data from 55 patients with Mahaim-type accessory pathways who underwent electrophysiological study.

Data presented as mean ± SD (range) or n (%) prevalence.
AH interval, atrial–His interval; AT, atrial tachycardia; AVNRT, AV-nodal re-entrant tachycardia; AVRT, atrioventricular re-entrant tachycardia; HV interval, His–ventricular interval; RF, radiofrequency; SVT, supraventricular tachycardia.
Discussion
To the best of our knowledge, this is the largest evaluation of patients with MAP who underwent an electrophysiological study. The main findings of the present study were as follows: (1) MAPs were mainly ablated at the lateral aspect of the tricuspid annulus, (2) the success rate of catheter ablation was 91.8%, (3) radiofrequency catheter ablation is a safe and effective approach, and (4) M potential was likely to be a useful target for the ablation of MAPs. 34
The MAP is typically conducted anterogradely; however, retrogradely conducting APs have been reported. Although the first description of MAPs was ‘nodoventricular’, subsequent investigations revealed these decrementally conducting APs were mainly located at the lateral tricuspid annulus site.12,35,36
Consistent with previous publications,20,37,38 MAPs were found to be primarily located at the lateral tricuspid annulus site in the present study (40 patients [72.7%]). In addition, 15 patients (27.3%) had MAPs located at the mitral annulus and tricuspid annulus, other than the lateral aspect. Left-sided MAPs are very rare, with few case reports published in the literature. Wide QRS complex tachycardia with right bundle branch morphology in these patients may be misinterpreted as ventricular tachycardia. Four patients in the present study had MAPs located along the mitral annulus, and ablation was successfully performed in these patients via a retrograde transaortic approach. A comparison of right-sided and left-sided Mahaim pathways in a series of patients revealed that left-sided Mahaim pathways (n = 4) were all clustered in the left lateral annulus. 39 Conversely, the present study found only two out of four left-sided Mahaim pathways located at the left lateral annulus.
The location and extent of AV ring specialized tissues has been demonstrated in histologic analyses by Yani et al., 40 showing their relationship with the normal AV conduction system consisting of the sinus node, the AV conduction system, and the Purkinje network. These specialized structures are AV rings which take their origin from inferior extensions of the AV node, passing rightward and leftward to encircle the orifices of the tricuspid and mitral valves, and reuniting to form an extensive retroaortic node. However, the right-sided pathways likely originate from the expanded primary ring, while the rings described by Yanni et al., 40 included both the primary ring and the AV canal myocardium. 13 The AV ring tissues are remnants of the sequestrated atrial components of the primary ring and they are similar to the structures initially illustrated by Kent. 4 They can form the atrial components of some of the unusual APs responsible for decremental conduction in the setting of pre-excitation. However, they are generally found in the vestibule of the tricuspid valve, while a small group of APs are derived from the mitral vestibule. They originate from the initial AV canal myocardium, which itself is able to produce AV delay even prior to the formation of the AV conduction axis.
Several techniques have been used for MAP ablation, including: (1) pathway (Mahaim-M) potential mapping, (2) activation mapping of the earliest local ventricular potential, (3) shortest stimulus-QRS interval mapping, and (4) bump mapping. 37 Because MAPs insert at the right ventricle free wall away from the tricuspid annulus, searching for the earliest ventricular activation along the tricuspid annulus cannot be an ablation target. These pathways do not conduct retrogradely; thus, mapping the earliest atrial activation is also impossible. The present standard ablation approach was based on searching for M potential.33,38 This technique has been widely used and became a preferred method for mapping and ablation. Inadvertent rigid catheter manipulation, which may cause conduction block lasting minutes to hours, must be avoided. Since most of the Mahaims are mapped on the lateral tricuspid annulus or right free wall underneath the valve, long sheaths help to maintain catheter stability.21,41,42
Long AV pathways with distal (nonannular) insertion often show wide dissemination over a large area of ventricular muscle, and targeting distal branches is time-consuming. It is possible to ablate some, as assessed by changes in the pre-excitation pattern, but complete elimination may not be achievable.43,44 In the present study, successful ablation was performed in three out of five patients with AV decremental pathways with a long course, representing a 60% success rate.
Patients with MAP often have accompanying APs or dual node physiology with AV nodal re-entrant tachycardia. 41 In the present study, nine patients were found to have other types of supraventricular tachyarrhythmias in addition to MAP during the electrophysiological study. Six patients with AV re-entrant tachycardia, two with AV nodal re-entrant tachycardia, and one with atrial tachycardia were induced and ablated successfully. All nine patients had MAP located at the lateral tricuspid annulus site.
The ECG during sinus rhythm was considered to be normal in the majority of patients with MAP, and such patients usually do not show overt pre-excitation. Nevertheless, some ECG patterns may contribute to the existence of Mahaim fibres. For example, an rS pattern in lead III combined with the absence of a septal q wave was shown to be specific for patients with a Mahaim fibre. 45 The specificity of an rS pattern in lead III associated with the absence of a septal q wave was close to 90%, albeit with a sensitivity of 45%. 45 In the present study population, the above-mentioned ECG patterns were investigated and only a small group of patients were found to demonstrate compatible findings, suggesting that its sensitivity is much lower.
There are several limitations to the present study. First, the results are limited by the retrospective nature of the study design. The epidemiological characteristics of the patients and findings of electrophysiological procedures were retrieved from an institutional archive, which may have resulted in loss of information. Secondly, as this was a multicentre study including data from only two hospitals in the same city, the generalizability of the results may be limited. However, the data may be extrapolated to other centres in Turkey, because the included hospitals receive referrals from elsewhere. Thirdly, due to the rarity of MAP, the present study group was too small to assess hard epidemiological data, although the study population was large compared with previously published studies.
In conclusion, the present study demonstrates that MAPs are often ablated at the lateral aspect of the tricuspid annuli, but may also be seen in other sides of the tricuspid and mitral annuli and, infrequently, in the right ventricle. The ablation of long AV pathways originating from the right ventricle with distal (nonannular) insertion was more challenging because of its wide dissemination over a large area, which caused prolonged procedure times. Consequently, the success rate was lower in this group. The M potential mapping technique was used for the MAP ablation procedure, and is likely to be a useful target for the ablation of MAPs. The coexistence of accompanying APs or dual node physiology were common.
Clinical Perspective
MAP is an uncommon and unique arrhythmia. It usually originates in the lateral tricuspid annulus site, displays decremental conduction, and inserts into the distal right ventricular free wall. MAPs are ablated at the tricuspid annulus, mitral annulus, paraseptal region, and right ventricle midapical region, respectively. RF ablation is the preferred method in the treatment of MAP. Among several different ablation techniques, the M potential mapping technique is likely to be a useful target for ablation of MAP.
