Abstract
Objectives
This study aimed to determine the effect of 7-day dietary supplementation of N-acetylcysteine (NAC)/zinc/vitamin C on the time-to-exhaustion (TTE), the cardiorespiratory fitness (CRF) index, and metabolic indicators.
Methods
This study enrolled volleyball student trainees (n = 18 men) who took NAC/zinc/vitamin C (750 mg/5 mg/100 mg) for 7 days at Jouf University, Saudi Arabia. The CRF index and TTE were determined. Serum concentrations of metabolic regulators (insulin, betatrophin, and hepatocyte growth factor), biomarkers of cellular damage/hypoxia, and indicators of lipid and glycemic control were measured.
Results
Supplementation improved the TTE and CRF index, and lowered cytochrome c, C-reactive protein, hypoxia-inducible factor-1α (HIF-1α), total cholesterol, insulin, and glycated hemoglobin values. Before and after supplementation, the CRF index was negatively correlated with body mass index and positively correlated with the TTE. Before supplementation, the CRF index was positively correlated with betatrophin concentrations, and hepatocyte growth factor concentrations were positively correlated with betatrophin concentrations and negatively correlated with the homeostasis model assessment of insulin resistance index. After supplementation, the CRF index was negatively correlated with HIF-1α concentrations and metabolites. Additionally, the TTE was negatively correlated with HIF-1α, cytochrome c, and triacylglycerol concentrations.
Conclusion
Supplementation of NAC/zinc/vitamin C improves metabolic and CRF performance.
Keywords
Introduction
Cardiorespiratory fitness (CRF) is the capacity of the circulatory and respiratory systems to supply oxygen to mitochondria of skeletal muscles. CRF is an objective measure of health that can be tracked over time and compared across populations. 1 Athletic high-intensity training increases cellular damage markers 2 and oxidative stress, which induce fatigue and decreased performance. 3 In contrast, enhancement of antioxidants increases the capacity to perform strenuous physical training and improves tolerance. Dietary thiol donors, such as cysteine and N-acetyl-cysteine (NAC), renew the major water-soluble antioxidant glutathione and attenuate the disadvantages of intense physical exercise.4–7
Although skeletal muscle is a major oxygen consumer, it is adaptively resilient to exercise-induced hypoxic episodes. Stabilization and activation of the oxygen-sensitive, master transcription regulator, hypoxia-inducible factor (HIF)-1α, perfects such adaptation to counteract oxidative stress, vascular endothelial dysfunction, and muscle fiber atrophy and fatigue.8,9 HIF-1α controls glucose transporters and glycolysis, metabolism, endothelial nitric acid synthase/angiogenesis, erythropoietin/erythropoiesis, mitochondrial function, and oxygen consumption, and regulates pH.8,10,11 HIF-1α deletion blunts acute skeletal muscle adaptation, which is lessened with long-term training. However, the overexpression of HIF-1α can be deleterious. Studies have shown higher concentrations of HIF-1α negative regulators in elite athletes than in control subjects.11,12
Serum concentrations of hepatocyte growth factor (HGF), a multi-functional trophic cytokine, are closely correlated with glycemic control and whole-body metabolism. HGH is important for angiogenesis and tissue regeneration. 13 Betatrophin (lipasin or angiopoietin-like 8) is a pancreatic β-cell proliferation inducer and metabolic regulator. Muscle exercise-induced secretion of irisin increases betatrophin production. This production significantly improves glucose and lipid homeostasis and insulin resistance.14–18 However, blood betatrophin and HGF concentrations greatly differ among healthy adults. Additionally, an increase in blood betatrophin concentrations is positively correlated with C-reactive protein (CRP), insulin resistance, and hypertriglyceridemia.19,20 Betatrophin is a potential biomarker for physiological performance that is correlated with physical training.21–23 The trophic effect of endurance and maximal exercise is due to an increase in circulating concentrations of growth factors, including HGF, and mobilization of bone marrow-derived hemopoietic and angiogenic progenitors.24,25 Hypoxic high-intensity training, particularly as a chronic adaptation, induces skeletal muscle satellite cells and angiogenesis, which are associated with HGF and HIF-1α. 26
The above-mentioned findings highlight the important role of HGF, HIF-1α, and betatrophin in physical activity. This study hypothesized that metabolic fitness combined with a healthy antioxidant armament has a positive effect on the CRF index and physical performance. This study aimed to determine the effects of a NAC/zinc/vitamin C antioxidant food supplement on the time-to-exhaustion (TTE), CRF index, and metabolic fitness. Serum indices of systemic inflammation, cellular damage and hypoxia, metabolic regulators, and glycemic and lipid control in novice volleyball players were measured.
Subjects and methods
Eighteen volunteer novice male volleyball trainees at Jouf University, Sakaka, Saudi Arabia were consecutively recruited for this study after signing a written informed consent form. Participants were in the middle of their training season and were apparently healthy, without a remarkable medical history. The study was conducted during the academic year of 2019 at the training rooms and play courts of the College of Dentistry under the supervision of Dr Moataz A. Kanoun, Associate Prof. of Physical Education. The study was conducted according to the guidelines of the Declaration of Helsinki, and was approved by the Bioethics Committee of Jouf University (approval #: 30-3/38-39). The age and body mass index ([BMI] weight in kg/height in m2) were prospectively measured. There were seven (39%) tobacco smokers.
The NAC/Z/C antioxidant supplement was composed of 750 mg NAC (NAC750; VitaminVersand24 DE GmbH, Stolberg, Deutschland) and 100 mg vitamin C/5 mg zinc (Limitless C-ZINC; Eva Pharma, Giza, Egypt) tablets, and was taken twice daily for 7 days. The CRF index was assessed using the following equation: CRF index = TTE/hb × 50, where the TTE is the time-to-exhaustion in seconds while stationary running at maximum speed, and hb is the summation of the recovery pulse (using the Mibest OLED Finger Pulse Oximeter; https://www.mibeststore.com/) measured at 1 to 1.5, 2 to 2.5, and 3 to 3.5 minutes post-exhaustion. An index of >96 indicates excellent fitness, 83 to 96 is good, 68 to 82 is average, 54 to 67 is low average, and <54 is poor. 27
Morning fasting peripheral venous blood samples (2.5 mL) were aseptically collected for recovering serum after clotting at room temperature on the first day before supplementation and on the last day of the study. Specific enzyme-linked immunosorbent assay-based quantitative kits were used to determine concentrations of glycated hemoglobin (HbA1c), insulin, CRP, HIF-1α, and cytochrome c (catalog numbers: CEA190Hu, CEA448Hu, SEA821Hu, SEA798Hu, and SEA594Mi, respectively; Cloud-Clone Corp., Wuhan, Hubei, China), as well as betatrophin and HGF (catalog numbers: SK00528-08/SK00331-02; Aviscera Bioscience, Inc., Santa Clara, CA, USA). Enzymatic colorimetric assays were used to measure serum glucose, triacylglycerol (TAG), and total cholesterol concentrations (Human, Gesellschaft fur Biochemica und Diagnostica mbH, Wiesbaden, Germany). The homeostatic model was used to calculate insulin resistance (https://www.thebloodcode.com/homa-ir-calculator/).
Except for smoking, continuous normally distributed data are presented as the mean ± standard deviation. The changes in TTE, the CRF index, and the other investigated parameters before and after NAC/Z/C supplementation were compared using the Wilcoxon matched-pairs signed rank test. Within group correlations (i.e., before and after supplementation) were tested by non-parametric Spearman’s analysis, and r and P values are presented. Smoking was represented by 1 and nonsmoking was represented by 0. Prism 7.0 software (GraphPad Software, Inc, San Diego, CA, USA) was used for analysis. Significance was set at P ≤ 0.05.
Results
Changes in the investigated parameters after supplementation
Table 1 shows the characteristics of the male participants and changes in the investigated parameters before and after providing NAC/Z/C supplementation. The TTE (P = 0.003) and the CRF index (P < 0.001) were significantly higher after supplementation than before supplementation. Concentrations of cytochrome c (P < 0.001), CRP (P = 0.004), HIF-1α (P < 0.001), insulin (P < 0.001), HbA1c (P < 0.001), total cholesterol (P = 0.023), and the homeostasis model assessment of insulin resistance (HOMA-IR) index (P < 0.001) were significantly lower after supplementation than before supplementation. Betatrophin, HGF, glucose, and TAG concentrations were not significantly different after supplementation compared with before supplementation.
Parameters measured before and after 1 week of N-acetyl-cysteine/zinc/vitamin C antioxidant food supplementation to novice volleyball players (total n = 18).
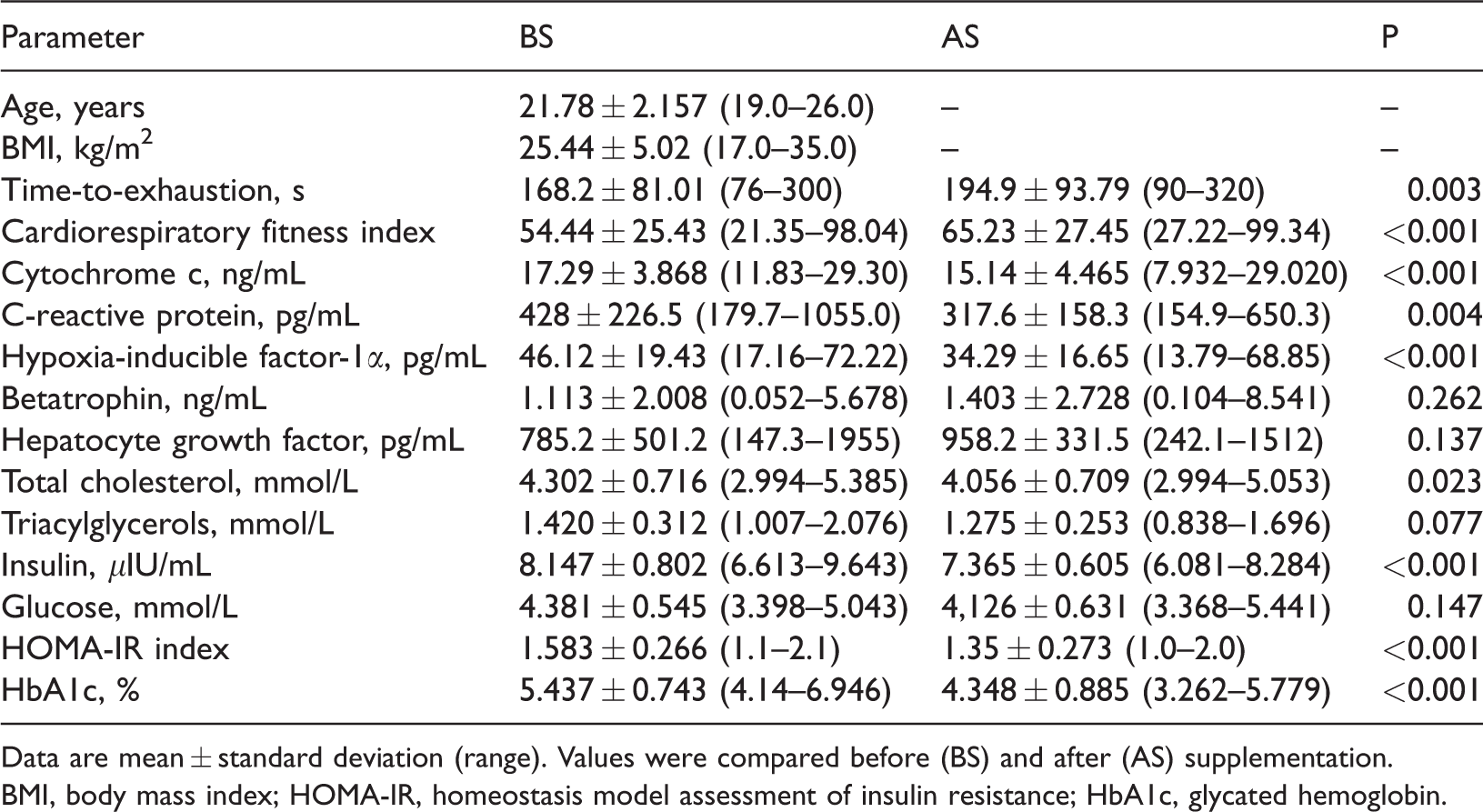
Data are mean ± standard deviation (range). Values were compared before (BS) and after (AS) supplementation.
BMI, body mass index; HOMA-IR, homeostasis model assessment of insulin resistance; HbA1c, glycated hemoglobin.
Correlations between indices of systemic inflammation, cellular damage and hypoxia, metabolic regulators, and glycemic and lipid control
Correlation analysis among parameters in the participants before supplementation is shown in Table 2. Age was significantly positively correlated with the TTE (r = 0.456, P = 0.029) and the CRF index (r = 0.440, P = 0.039), but negatively correlated with TAG concentrations (r = −0.457, P = 0.028). BMI was negatively correlated with the CRF index (r = −0.410, P = 0.05), and positively correlated with insulin concentrations (r = 0.544, P = 0.010) and HbA1c values (r = 0.439, P = 0.034). Smoking was negatively correlated with the TTE (r = −0.473, P = 0.024,) and betatrophin concentrations (r = −0.703, P = 0.001). The TTE was positively correlated with the CRF (r = 0.934, P < 0.001) and betatrophin concentrations (r = 0.405, P = 0.05). Cytochrome c concentrations were positively correlated with HIF-1α concentrations (r = 0.634, P = 0.002). Betatrophin concentrations were positively correlated with HGF concentrations (r = 0.577, P = 0.006). Insulin concentrations were positively correlated with the HOMA-IR index (r = 0.690, P = 0.001) and HbA1c values (r = 0.474, P = 0.024). Glucose concentrations were positively correlated with the HOMA-IR index (r = 0.765, P = 0.001) and total cholesterol concentrations (r = 0.516, P = 0.014). The HOMA-IR index was positively correlated with total cholesterol concentrations (r = 0.525, P = 0.013). HGF concentrations were negatively correlated with the HOMA-IR index (r = −0.499, P = 0.018), insulin concentrations (r = −0.399, P = 0.05), and TAG concentrations (r = −0.413, P = 0.05).
Correlations of demographic and anthropometric characteristics with other parameters before antioxidant supplementation in the participants.

Data are r/P values.
BMI, body mass index; TTE, time-to-exhaustion; CRF, cardiorespiratory fitness; CytC, cytochrome c; CRP, C-reactive protein; BT, betatrophin; HGF, hepatocyte growth factor; HIF-1α, hypoxia-inducible factor-1α; Ins, insulin; HOMA-IR, homeostasis model assessment of insulin resistance; HbA1c, glycated hemoglobin; TC, total cholesterol; TAGs, triacylglycerols, ns, nonsignificant.
Correlation analysis among parameters in the participants after supplementation is shown in Table 3. Age was significantly positively correlated with the TTE (r = 0.399, P = 0.050) and the CRF index (r = 0.453, P = 0.029). BMI was negatively correlated with the CRF index (r = −0.399, P = 0.05), and positively correlated with CRP (r = 0.425, P = 0.039), glucose (r = 0.488, P = 0.020), HbA1c (r = 0.436, P = 0.035) and TAG (r = 0.430, P = 0.037) values. Smoking was negatively correlated with the TTE (r = −0.478, P = 0.023) and betatrophin concentrations (r = −0.561, P = 0.008). The TTE was negatively correlated with cytochrome c (r = −0.409, P = 0.05), HIF-1α (r = −0.401, P = 0.05) and TAG (r = −0.436, P = 0.035) concentrations, and positively correlated with the CRF (r = 0.890, P < 0.001). The CRF was negatively correlated with HbA1c (r = −0.439, P = 0.034), TAG (r = −0.491, P = 0.019), glucose (r = −0.410, P = 0.05), and HIF-1α (−0.403, P = 0.05) concentrations. Cytochrome c concentrations were positively correlated with HIF-1α (r = 0.566, P = 0.007) and total cholesterol (r = 0.454, P = 0.029) concentrations. Betatrophin concentrations were negatively correlated with HIF-1α concentrations (r = −0.440, P = 0.034). HGF concentrations were positively correlated with HIF-1α concentrations (r = 0.399, P = 0.05) and negatively correlated with TAG concentrations (r = −0.447, P = 0.032). HIF-1α concentrations were positively correlated with glucose concentrations (r = 0.408, P = 0.050). The HOMA-IR index was positively correlated with HbA1c (r = 0.436, P = 0.035), glucose (r = 0.813, P < 0.001), and insulin (r = 0.619, P = 0.003) values. Glucose concentrations were positively correlated with HbA1c concentrations (r = 0.409, P = 0.05) and total cholesterol values (r = 0.421, P = 0.041).
Correlations of demographic and anthropometric characteristics with other parameters after antioxidant supplementation in the participants.

Data are r/P values.
BMI, body mass index; TTE, time-to-exhaustion; CRF, cardiorespiratory fitness; CytC, cytochrome c; CRP, C-reactive protein; BT, betatrophin; HGF, hepatocyte growth factor; HIF-1α, hypoxia-inducible factor-1α; Ins, insulin; HOMA-IR, homeostasis model assessment of insulin resistance; HbA1c, glycated hemoglobin; TC, total cholesterol; TAGs, triacylglycerols, ns, nonsignificant.
Discussion
Oxidative homeostasis balances the endogenously generated and exogenously acquired prooxidant free radicals and oxidants versus the enzymatic, and endogenous and exogenously acquired nonenzymatic antioxidants (e.g., vitamins C and E, lipoate, CoQ, glutathione, carotenoids, and zinc). Antioxidant supplementation has beneficial outcomes with physical activity, particularly in high-intensity, short-duration, vigorous, and exhaustive exercise, and for physical inactivity compared with low-to-moderate intensity and long-term exercises.28–30 NAC is a non-essential precursor of cysteine and glutathione; the major water-soluble antioxidant in muscle. NAC also enhances potassium homeostasis and potentiates sarcoplasmic Na+/K+- and Ca2+-ATPases.31,32 However, counteracting the reactive oxygen species deprives cells from their physiological roles. This is the reason for the outcomes of antioxidant supplementation with substantial physical activity being controversial. High doses of antioxidant supplementation lead to prooxidant effects and systemic and muscle harmful effects by inhibiting the anabolic signaling pathways (JNK, p65phox, and Akt/mTOR/p70s6K/p38MAPK) that blunt the expression of adaptation proteins. These effects apply to vitamins C and E and NAC among others.32–35
CRF declines in untrained individuals, and fitness is inversely correlated with age, BMI, metabolic syndrome, diabetes, and dyslipidemia as risk factors for cardiovascular disease. 36 In this study, NAC/Z/C antioxidant supplementation deceased cellular damage and hypoxia, as well as inflammatory markers (cytochrome c, HIF-1α, and CRP), and improved metabolic efficiency (decreased total cholesterol, HbA1c, insulin, and HOMA-IR values). These metabolic effects were paralleled by a nonsignificant increase in serum concentrations of betatrophin and HGF and a decrease in glucose and TAG concentrations. The large inter-individual variation might have nullified the significance of differences among these variables. However, strengthened metabolic fitness was found as shown by a significant improvement in the TTE and CRF index. An improved metabolic capacity and oxygen supply enable skeletal muscle to adapt to long-term aerobic performance at higher intensities. 11 Vitamin C suppresses endothelial cell apoptosis by increasing the bioavailability of intracellular nitric oxide, which prevents cytochrome c release and blocks apoptosis signaling. 37
A meta-analysis of previous studies that measured changes in circulating HIF-1α concentrations after endurance exercise showed various profiles of a reduction, increase, and no change. Expression of muscle HGF is mostly increased after endurance exercise. Serum betatrophin concentrations are mostly decreased after endurance exercise. Most previous studies on endurance exercise showed increased gene expression for cytochrome c in muscles. Endurance exercise mostly acutely reduces serum insulin concentrations. 38 HIF-1α, which is induced by acute exercise, unfavorably inhibits oxygen consumption and aerobic mitochondrial metabolism. Such HIF-1α activation is blunted by its negative regulators that are enhanced by long-term aerobic endurance exercise training.11,24,26,39 HbA1c values indirectly indicate a dominant inflammatory/oxidative stress status and correlate with CRP concentrations.40,41 Therefore, a reduction in HbA1c, CRP, and cytochrome c values in the present study reflects the metabolic efficacy of the antioxidant cocktail used.
In the present study, age was significantly positively correlated with the TTE and CRF index before and after supplementation. However, age was only significantly negatively correlated with serum TAG concentrations before supplementation. BMI was negatively correlated with the CRF index before and after supplementation. BMI was also positively correlated with serum insulin and HbA1c values before supplementation, and positively correlated with serum CRP, glucose, HbA1c and TAG values after supplementation. Smoking was negatively correlated with the TTE and serum betatrophin concentrations before and after supplementation. Previous studies have shown negative correlations of CRF and physical activity with BMI and smoking.42–46 However, although smoking and physical activity are largely incongruent behaviors, their relationship is often attenuated or reversed in male adolescents and for moderate (vs. vigorous) exercise.47,48 This finding is consistent with our findings in young adult participants.
Aerobic and muscle fitness are independently, inversely associated with serum CRP concentrations.
49
In contrast, independently of lifestyle behavior and BMI, an improvement in and/or high CRF reduces CRP concentrations.
50
In the present study, correlation analysis before supplementation showed the following significant positive correlations: 1) TTE with the CRF index and serum betatrophin concentrations, 2) serum cytochrome c with HIF-1α concentrations, 3) serum betatrophin with HGF concentrations, 4) serum insulin concentrations with HOMA-IR and HbA1c values, 5) serum glucose concentrations with the HOMA-IR index and total cholesterol concentrations, and 6) the HOMA-IR index with serum total cholesterol concentrations. HGF concentrations were negatively correlated with the HOMA-IR index, and insulin and TAG concentrations. Mitochondrial dysfunction, which improves with physical activity and fitness, has been implicated in the development of metabolic disorders, such as insulin resistance and type 2 diabetes mellitus.
61
Mitochondrial dysfunction and free cytoplasmic cytochrome c, which is involved in oxidative stress and O2/energy shortage, activate caspase 9 as the primary event in apoptosis.
62
Circulating HGF concentrations are high in healthy athletes, irrespective of the type of exercise. 24 This finding may explain the present finding of no significant increase in HGF concentrations, which showed large inter-individual variation, because basal concentrations were already high. Large individual variations in serum HGF concentrations have been previously reported.20,38 HGF induces bcl-2 and inhibits translocation of bax from the cytosol to the mitochondria to prevent endothelial cell apoptosis, which is induced by high glucose concentrations. 64 HGF attenuates oxidative stress and cytochrome c release in retinal pigment epithelium cells treated with buthionine-(S,R)-sulfoximine, which depletes glutathione. 65 In contrast, HGF concentrations are negatively correlated with aerobic and muscle fitness, and positively correlated with CRP, interleukin-6, and tumor necrosis factor-α concentrations. 49
The present study showed no significant increase in serum betatrophin concentrations after supplementation, with large inter-individual variation. Serum betatrophin concentrations were positively correlated with the TTE and HGF concentrations before supplementation, and negatively correlated with HIF-1α and smoking after supplementation. Betatrophin concentrations show large inter-individual differences and are positively correlated with age, glucose, TAG, total cholesterol, and HbA1c values in healthy adults and patients.
20
The present study showed better metabolic cytoprotective action, anti-inflammatory effects, and resilience to hypoxia after supplementation, as reflected by a significant improvement in the TTE and CRF index in the participants. This finding was observed despite the fact that glycemic and lipid profile parameters were within the normal physiological range. Ten years previously, our research group was one of the first groups worldwide to characterize the changes in circulating concentrations of HIF-1α as a hypoxic cellular stress and damage biomarker, which is consistent with the present results.70–73 In the present study, the distinct improvement in the performance of the participants regarding the TTE, the CRF index, and metabolic fitness compared with previous studies on well-trained athletes,28–30 could be attributed to the following. 1) This study used a strong antioxidant cocktail, 2) the participants’ athletic performance was novice in nature, and 3) the cardiorespiratory fitness (n = 9; 50% with a poor index) and the metabolic fitness (n = 11; >61% with a BMI of ≥25 kg/m2) of the participants were poor, which suggested physical inactivity and/or unhealthy nutrition. However, validation of the utility of the biomarkers used in this study requires larger studies in the future.
Conclusions
The antioxidant NAC/Z/C food supplement applied in this study led to a significant improvement in the TTE and CRF index because it supported cellular viability (cytochrome c and HIF-1α), reduced inflammation (CRP) and insulin resistance (HbA1c and HOMA-IR), and improved the lipid profile (total cholesterol) and glycemic control indices (insulin and HbA1c). These findings were correlated with a nonsignificant increase in metabolic regulators of cytoprotection (betatrophin and HGF), and a decrease in glucose and TAG concentrations. BMI and smoking negatively affected these benefits. HIF-1α concentrations were positively correlated with cytochrome c and glucose concentrations, and negatively correlated with the CRF index and TTE, which suggest that HIF-1α is a marker of damage. The marked improvements observed in this study are reasonable in consideration of the initial low physical fitness and CRF of our participants, and this suggests unhealthy nutrition. Larger and longer term studies are required in the future to confirm the generalization of our results and the utility of the biomarkers used, particularly for trained athletes.
Footnotes
Acknowledgements
The author is highly indebted to Prof. Tarek H. El-Metwally (Biochemistry Division, Pathology Department, College of Medicine, Jouf University, Sakaka, Saudi Arabia) for help during the planning and execution of lab work of this study and to Prof. Hossam M. Omar (Assiut University, Assiut, Egypt) for assistance of final drafting of the manuscript. The training and field data collection were sponsored by Dr. Moataz A. Kanoun (Physical Education Department, Jouf University, Sakaka, Saudi Arabia) and Dr. Mohammad Salahuddin (Department of Physiology, College of Medicine, Jouf University, Sakaka, Saudi Arabia).
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: The authors were supported by the Deanship of Scientific Research, the Vice Presidency for Postgraduate Studies and Scientific Research, Jouf University, Sakaka, Saudi Arabia for funding of this work (project fund number: 39/564).
