Abstract
Mediastinal unicentric Castleman disease (UCD) frequently manifests as a hyper-enhancing lymph node mass and is often surgically curable. However, because of excessive vascularisation and adhesion to important surrounding structures, surgery is often associated with severe haemorrhage that is often difficult to control thoracoscopically. Therefore, thoracotomy is often preferred, which increases the trauma to the patient and affects postoperative recovery. Here, we describe the case of a 30-year-old male patient with a large upper mediastinal lymph node (7 × 5 × 4 cm) that was compressing his superior vena cava. The distribution of nutritive arteries of the mass was analysed in detail, and the main branches were embolised prior to surgery. With the assistance of preoperative isovolumetric haemodilution, we achieved complete resection through single-port thoracoscopy, with only minor haemorrhage, which enabled the patient to recover rapidly. This multidisciplinary collaborative model, based on single-port thoracoscopic surgery, may be of wide practical use for the treatment of mediastinal UCD.
Keywords
Introduction
Giant mediastinal lymph node hyperplasia, also named Castleman disease (CD), is a rare type of benign lymph node hyperplasia that was originally described by Castleman and Towne in 1954. 1 Unicentric CD (UCD) usually manifests as a hyper-enhancing lymph node mass that is often surgically curable.2,3 However, because of local infiltration and excessive vascularisation, surgery is often associated with severe haemorrhage or a failure to achieve complete resection.4,5 Here, we describe a case of rapid recovery from the surgical resection of giant mediastinal lymph node hyperplasia.
Case presentation
The reporting of the present case conforms to the CARE guidelines. 6 A 30-year-old male patient was admitted to our hospital because of chest tightness after exercise of 1 month’s duration. He had been diagnosed with chronic hepatitis B >10 years earlier, for which he was taking entecavir. His hepatitis B virus DNA titre was within the normal range. Computed tomography (CT) revealed a well-defined mass in his right upper mediastinum that was compressing his superior vena cava (Figure 1a and b). Subsequent CT thoracic aortic angiography demonstrated that the mediastinal mass was mainly supplied by the brachiocephalic trunk, and more specifically, most of the large and tortuous vessels surrounding the mass originated from the right subclavian artery (Figure 1c). Using arteriography of the thoracic aorta and subclavian artery, we determined that the bilateral branches of the subclavian artery and the T3 and T4 intercostal arteries were the main supply arteries. His two distal intercostal arteries, right internal mammary artery and left thyrocervical trunk were embolised (Figure 2), but we were unable to successfully embolise the right subclavian artery branch because of its narrow lumen.
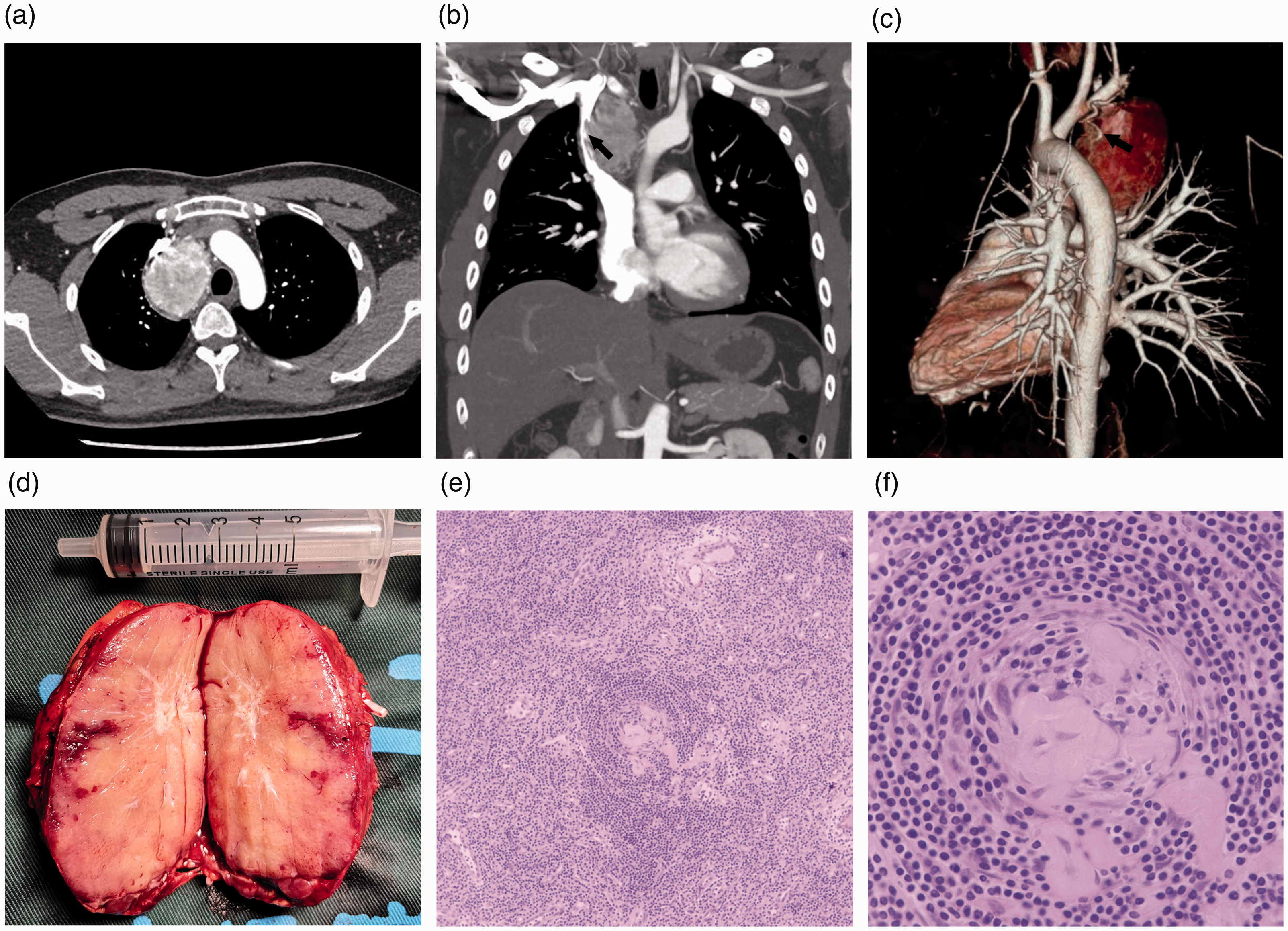
Imaging manifestations, gross appearance and histology of the mass. (a and b) Computed tomography (CT) images demonstrating a homogenously enhanced, well-circumscribed mass that is compressing the superior vena cava (arrow). (c) CT-based three-dimensional reconstruction showing thick and tortuous blood vessels (arrow) around the mass. (d) Gross intraoperative appearance of the surgically excised mass. (e and f) Photomicrographs of haematoxylin and eosin-stained sections of the mass (100× and 400× respectively). The histologic architecture of the lymph node is almost destroyed, vascular proliferation and hyalinisation of the vessel walls are present, and in some areas, there are thickened mantle zones, lined by multiple layers of lymphocytes.

Contrast examination before and after embolisation of the nutritive vessels. (a) T3 intercostal artery; (b) T4 intercostal artery; (c) left thyrocervical trunk; (d) right internal thoracic artery.
Surgery was scheduled for the day after the arteriography. A double-lumen endotracheal tube was placed and the patient was positioned in the left lateral decubitus position. Single-lung ventilation was applied, and a 4-cm long incision was made in the fourth intercostal space along a right anterior axillary line. Via single-port thoracoscopy, we disconnected the thin nutritive vessels by ultrasonic scalpel and coagulation hook, gradually freed up the thicker branches connecting to the mass and clamped these using vascular clips, and finally removed the tumour completely. Because the mass was hard and lacked the flexibility of lung tissue, we could not remove it via a normal-length incision. Therefore, to avoid the mass rupturing, we extended the incision appropriately. The mass was 7 × 5 × 4 cm in size (Figure 1d), and was later confirmed to be hyaline vascular Castleman disease by postoperative haematoxylin and eosin-staining (Figure 1e and f), immunohistochemistry, and in situ hybridisation (ISH) (Figure 3). However, immunostaining for human herpesvirus (HHV)-8 and ISH for Epstein–Barr virus (EBV) were both negative, which is consistent with the findings of most previous studies that neither HHV-8 nor EBV is involved in hyaline vascular CD. 7

Immunohistochemistry and in situ hybridisation (ISH) of sections of the mass. (a and b) CD20 and CD79α staining to indicate B cells. (c and d) CD3 and CD5 staining to indicate T cells. (e and f) Follicular dendritic cells stained with CD21 and CD23. (g) Ki-67 staining for active cell proliferation. Staining is principally in the germinal centres. (h) Immunostaining for HHV-8 was negative. (i) In situ hybridisation for the Epstein–Barr virus encoding region was negative. All 40× magnification.
In addition to the vascular embolism, acute normovolaemic haemodilution and autologous blood transfusion were also used. After the induction of anaesthesia and before the surgical incision was made, blood was slowly drawn from the left jugular vein of the patient and an equal amount of balanced salt solution was simultaneously provided via a peripheral vein until the haemoglobin concentration had decreased from 167 g/L to 130 g/L. Blood reinfusion was performed when the surgical wound had stopped haemorrhaging. A precise thoracoscopically-guided intercostal nerve block was performed. The entire procedure lasted ∼1 hour, and the total blood loss was ∼200 ml. The patient was discharged on the third postoperative day, when he demonstrated an absence of cough, haemothorax, chylothorax, and severe pain. During the following year there were no complications or recurrence.
Discussion
CD, also known as angiofollicular lymph node hyperplasia or giant lymph node hyperplasia, is a rare lymphoproliferative disease that is characterised by benign, localised enlargement of lymph nodes.8,9 It includes a spectrum of pathological variants, including unicentric hyaline vascular CD and the less common plasma cell CD, multicentric CD, and HHV-8-associated CD. The mediastinum is the most common location for UCD, with less common extrathoracic sites being the neck, axilla, abdomen, and pelvis. 10
Unlike plasma-cell CD, unicentric hyaline vascular CD is rarely associated with systemic symptoms. Multicentric CD is histologically closely related to the plasma-cell type, and is often associated with systemic symptoms, including fever, night sweats, fatigue, anaemia and arthralgia. Laboratory measurements may indicate hypergammaglobulinaemia, thrombocytosis and/or rapid erythrocyte sedimentation rate. Nevertheless, hyaline vascular CD is frequently not associated with obvious symptoms and is often an incidental finding during physical examination. 11
The treatment of multicentric CD may require steroid therapy, chemotherapy, antiviral medication or the use of antiproliferative regimens. However, UCD lesions can often be completely resected. 2 Open surgery is the conventional approach used for UCD because the lesions are hypervascular and often adhere closely to the surrounding tissues, which include important mediastinal structures, and therefore a careful surgical approach is necessary.5,12 Failure to fully resect the lymph node that is primarily affected is the only significant predictor of mortality, and a diagnostic “wedge” resection is associated with significantly worse outcomes than complete excision of the diseased lymph node. 13 Accordingly, evaluation using selective angiography is usually necessary to avoid uncontrollable massive haemorrhage: characterisation of the hypervascularisation in this way provides an indication of the risk of haemorrhage during surgery. Chest CT, performed in three phases, is an important means of evaluating the vascularisation of mediastinal tumours. 14 In the present case, the chest tightness after exercise was probably caused by vascular compression because the mass was situated between the azygous vein and superior vena cava, and close to the trachea and subclavian artery. To date, there have been no detailed reports of the distribution of the nutritive arteries of UCD lesions in the location identified in the present patient. Here, we have comprehensively described the arterial supply of the mass, which is a critical component of the preoperative evaluation of such patients, and the present case report therefore provides a useful addition to the existing literature.
Compared with conventional multi-port thoracoscopy, single-port video-assisted thoracic surgery has become a more popular type of thoracic surgery, owing to advantages such as the lower postoperative pain and superior cosmetic effect associated with it. 15 We have reviewed the existing reports of the use of multi-port thoracoscopy for the resection of intrathoracic CD lesions (Table 1), and in the present case, we used single-port thoracoscopy to resect the mass. An incision in the fourth intercostal space, along an anterior axillary line, is ideal to avoid blind spots in the surgical field. During the procedure, special attention should be paid to the protection of the azygos vein, superior vena cava, and tracheal wall. In addition to opening the pleura over the azygos vein, we also widened the gap between the azygos vein and the root of the right pulmonary artery. When dealing with the nutritive blood vessels surrounding the mass, we used the aspirator to carefully retract the azygos vein and superior vena cava. Furthermore, the curved aspirator can be used to remove the small amount of haemorrhage, to ensure that the tumour bed is clearly identifiable.
Clinical findings, surgical details, and outcomes of previously reported cases of CD that were managed via thoracoscopy.

HV, hyaline vascular-type Castleman disease; PC, plasma cell-type Castleman disease; VATS, video-assisted thoracic surgery; SVC, superior vena cava.
Acute normovolaemic haemodilution is a blood conservation technique that is used to reduce the red blood cell volume lost during surgery. 16 It is the only method that provides fresh autologous blood, in which platelet and clotting factor function are normally kept intact and few red blood cells are lost. 17 It is usually used in vascular surgery, such as for peripheral arterial occlusive disease, central retinal vein thrombosis, and subarachnoid haemorrhage. 18 However, to date, there have been no reports of its use during the surgical treatment of giant mediastinal lymph node hyperplasia. In the present case, the total volume of blood withdrawn was 500 ml and the patient’s body mass was 60 kg, and therefore little effect on the patient would be expected. 19
Conclusion
To avoid uncontrollable haemorrhage, it is extremely important to evaluate the vascular supply of CD masses thoroughly and to perform effective nutritive artery embolisation. In this way, single-port thoracoscopic surgery is rendered safe and reliable. Normovolaemic haemodilution and autologous blood transfusion also represent a useful adjunct. These measures help ensure the safety of the procedure, reduce postoperative pain, and promote the recovery of the patients. We believe that this multidisciplinary collaborative model, based on single-port thoracoscopic surgery, may represent a practical means of treating mediastinal UCD.
Footnotes
Ethics statement
The study protocol was approved by the Ethics Committee of the Second Affiliated Hospital, School of Medicine, Zhejiang University (approval number: 2021-0754). Written informed consent was obtained from the patient described in this study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was supported by a grant from the National Natural Science Foundation of China (Grant No. 81601987).
Author contributions
Conceptualisation: Haihua Gu and Junqiang Fan. Data curation: Haihua Gu, Tianshu Liu, Ling Zhu, and Lufeng Zhao. Writing-original draft: Haihua Gu. Writing-review & editing: Haihua Gu and Junqiang Fan.
