Abstract
Objective
To assess the influencing factors of depressive symptoms in malignant tumour patients.
Methods
Participants were 2079 inpatients with malignant tumour (1291: depressive symptoms; 788 no depressive symptoms). Univariable and multivariable logistic regression were used to evaluate sociodemographic and clinical factors influencing depressive symptoms.
Results
Risk factors were family income ≤5000 yuan (odds ratio [OR]: 4.966, 95% confidence interval [CI]: 2.938–8.395) and 5001–10,000 yuan (OR: 3.111, 95% CI: 1.840–5.260); Karnofsky Performance Status of 70 (OR: 2.783, 95% CI: 1.281–6.042) and 80 (OR: 1.834, 95% CI: 1.139–2.953); disease course ≤1 year; palliative treatment (OR: 2.288, 95% CI: 1.292–4.055); progressive disease (OR: 1.876, 95% CI: 1.284–2.739); pain (OR: 1.973, 95% CI: 1.555–2.505); cancer type: lung (OR: 3.199, 95% CI: 1.938–5.279), oesophagus (OR: 3.288, 95% CI: 1.673–6.464), cervix (OR: 1.542, 95% CI: 1.056–2.253) and partial knowledge of disease condition (OR: 2.366, 95% CI: 1.653–3.385). Return to work (OR: 0.503, 95% CI: 0.348–0.727) and physical exercise (OR: 0.437, 95% CI: 0.347–0.551) were protective against depressive symptoms.
Conclusions
Several factors affected depressive symptoms in malignant tumour patients, including income, disease type and course, palliative treatment, return to work and physical exercise.
Introduction
Malignant tumour has become a major cause of global mortality and its management is a huge health burden for health-care systems.1,2 According to the International Agency for Research on Cancer, the cancer burden in China is substantial and the incidence of cancer is still rising.3,4 Although substantial improvements have been made in the clinical treatment of malignant tumour, its overall prognosis remains unfavourable. 5 The diagnosis of malignant tumour and the resulting deterioration in health status are a psychological challenge for patients, as the disease has a substantial effect on patients’ physical appearance, physical ability, family, occupation, economic status and emotions during diagnosis and treatment. 6 More attention should be paid to psychosocial states in cancer patients.
Depression is a common psychological disease that is comorbid with malignant tumour. There is evidence from previous studies that the prevalence of depressive symptoms is substantial in patients with malignant tumours. 7 One study reported that the prevalence of depression among head and neck cancer patients varies from 13% to 40% at diagnosis, 25% to 52% during treatment and 11% to 45% in the first 6 months following treatment. 8 The prevalence of major depressive disorder in breast cancer patients is estimated as 10% to 33%. 9 It has also been reported that the incidence of major depression in malignant tumour patients is approximately 15% in oncological and haematological settings. 10 Cancer treatment causes metabolic and endocrine changes, and chronic pain may also increase the susceptibility to depression of malignant tumour patients.11,12 Depression damages the immune system and promotes the progression of malignant tumours, which leads to worse disease outcomes. 13 Depression also increases patient treatment noncompliance, such as failure to attend consultation appointments, and leads to poorer treatment response. 14 A better understanding of the influencing factors of depression may not only prolong the lives of patients, but also improve their quality of life. 15 Many studies have explored the risk factors for depression in different cancers. Lee et al. showed that higher educational level, stressor severity and anxiety severity were associated with depression in patients with head and neck cancer and lung cancer. 16 Other studies have found that levels of blood cytokines, such as interleukin-10, interleukin-6, interleukin-8 and tumour necrosis factor-α, are associated with depression status in patients with cancer. 17 However, these studies used only clinical or laboratory examination indexes and did not analyse important social factors such as work status. The aim of this study was to identify the influencing factors of depressive symptoms in patients with malignant tumours, focusing on demographic, clinical and social characteristics. The findings may help clinicians to prevent depressive symptoms in patients with malignant tumours.
Methods
Study design and participants
Potential participants in this cross-sectional study were inpatients with malignant tumours from the following departments of the Affiliated Cancer Hospital of Xinjiang Medical University: pulmonary medicine, gastroenterology, breast radiotherapy, chest and abdomen radiotherapy, gynaecology radiotherapy, head and neck radiotherapy, and integrated traditional Chinese and Western medicine. Participants were selected according to the following inclusion criteria: malignant tumour confirmed by pathology or cytology, aged 18 to 85 years, ability to fully understand the questionnaire content and provision of written consent. Excluded patients were those with cognitive impairment and severe weakness owing to disease or treatment; those who did not agree to participate in the study; patients diagnosed with schizophrenia, schizoaffective disorder or bipolar I disorder; patients with depressive symptoms caused by another medical condition or induced by substances (defined using the International Classification of Diseases, Tenth Revision) and patients with severe anxiety and acute confusional states associated with drug toxicity. The reporting of this study conforms to the STROBE guidelines. 18 All participants provided informed consent and the study was approved by the ethics committee of the Affiliated Cancer Hospital of Xinjiang Medical University (approval number: G-201554).
Assessment and diagnosis of depressive symptoms
The Zung Self-Rating Depression Scale (SDS) is a self-report questionnaire that has been previously used to evaluate the presence of depressive symptoms in malignant tumour patients. 19 The SDS comprises 20 items scored on a scale of 1 to 4. The raw SDS score ranges from 20 to 80, and a standard score is calculated by multiplying raw scores by 1.25, resulting in a total standard score from 25 to 100. The severity of depressive symptoms is defined as <53, no depressive symptoms; 53 to 62, mild depressive symptoms; 63 to 72, moderate depressive symptoms; and 72 to 100, severe depressive symptoms. 20
Variables and data collection
Important risk factors were selected based on the sociodemographic and clinical data collected from all patients. Sociodemographic information comprised sex, age, race (Han population or minority population), residence (city or country), education (illiterate, junior, senior or college), marital status (married or single), monthly family income (≤5000, 5001–10,000 or >10,000 yuan), work status (working or not working), physical exercise (engagement in exercise or no exercise). Clinical data comprised Karnofsky Performance Status (KPS) score (60, 70, 80, 90 or 100), cancer type (head and neck, lung, breast, oesophagus, stomach, colorectum, cervix, thyroid or others), disease course (≤1 month, 2–6 months, 7–12 months or >1 year), cancer stage (I, II, III or IV), therapy (re-examination, chemotherapy or/and radiotherapy or palliative treatment), disease state (stable, progression or newly diagnosed), metastasis or not, awareness of condition (no idea, partial knowledge or full knowledge), pain or not and history of hypertension or diabetes.
Definitions of variables
Pain intensity was evaluated using a numerical rating scale and classified into none (0 points), mild (1–3), moderate (4–6) and severe (7–10). 21 Physical activity was defined as any type of planned, structured and repeated physical activity, including any body movement produced by skeletal muscle contraction for improving or maintaining physical fitness. 22 The KPS is a performance status scale that evaluates patient status and is administered by a health-care provider. Patients were assigned to 1 of 10 categories (ranging from 0 (dead) to 100 (normal activity, no evidence of disease). 23 A detailed definition of KPS score is shown in Supplementary Table 1.
Statistical analysis
EpiData 3.1 (EpiData Association, Odense, Denmark) was used to establish a database, and double entry was used to ensure the correctness of the input data. Statistical analysis was conducted using R 4.0.2 software (www.r-project.org). Quantitative variables with normal or approximately normal distribution were expressed as mean ± standard deviation (SD). One-way analysis of variance was used for between-group comparisons. Quantitative variables with skewed distributions were represented by median and interquartile range. Enumeration data were presented as case and frequency (N [%]). The influencing factors of depressive symptoms in patients with malignant tumour were preliminarily analysed using the chi-square test (χ2) and odds ratios (ORs) and 95% confidence intervals (CIs). Statistically significant factors in the univariable analysis were included in a logistic regression analysis using a backward stepwise regression method. The results of the above analysis were used to conduct multivariable logistic regressions for breast cancer, lung cancer, colorectal cancer and cervical cancer to explore the influencing factors of depressive symptoms in specific tumour populations. Of the factors examined in the multivariable logistic regression, the main factors were selected for an efficacy analysis to test the sample size efficacy. All statistical analyses were two-sided, and P < 0.05 was considered statistically significant.
Results
Background characteristics of participants
In total, 2178 patients with malignant tumour were enrolled in this study. After excluding patients with impaired cognition owing to disease or treatment; severely weak patients; those who did not agree to participate in the study; those diagnosed with schizophrenia, schizoaffective disorder or bipolar I disorder; those with depressive symptoms caused by another medical condition or induced by substances; and those with severe anxiety and acute confusional states associated with drug toxicity, 2079 participants remained (response rate: 95.45%). All participants were divided into a depressive symptoms group (n = 1291) and a non-depressive symptoms group (n = 788) according to the presence of depressive symptoms. The screening process is shown in Figure 1. The men:women sex ratio was 1:1.82. The average age of all patients was 54.62 ± 11.43 years and 86.24% of patients were married. Of participants, 1473 (70.85%) were Han, 54.65% were educated to junior high school level or below, 1285 lived in the city and 57.53% had a monthly family income below 5000 yuan. Of participants, 1036 engaged in physical exercise. Only 233 patients returned to work and 1846 did not return to work or retired. Most of the 2079 participants had KPS scores of 80 (33.09%) or 90 (52.86%). There were 321 patients with lung tumour, 512 patients with breast cancer, 305 patients with colorectal cancer and 305 patients with cervical cancer. Of patients, 53.39% had a disease course longer than 6 months and 26.84% patients had disease progression. Regarding treatment, 51.23% patients had received radiotherapy, chemotherapy or concurrent chemoradiotherapy. Of patients, 639 were at stage IV, 725 had tumour metastasis and 60 had no knowledge of their disease state. A total of 235 patients had hypertension and 130 had diabetes. Detailed patient data are shown in Table 1.
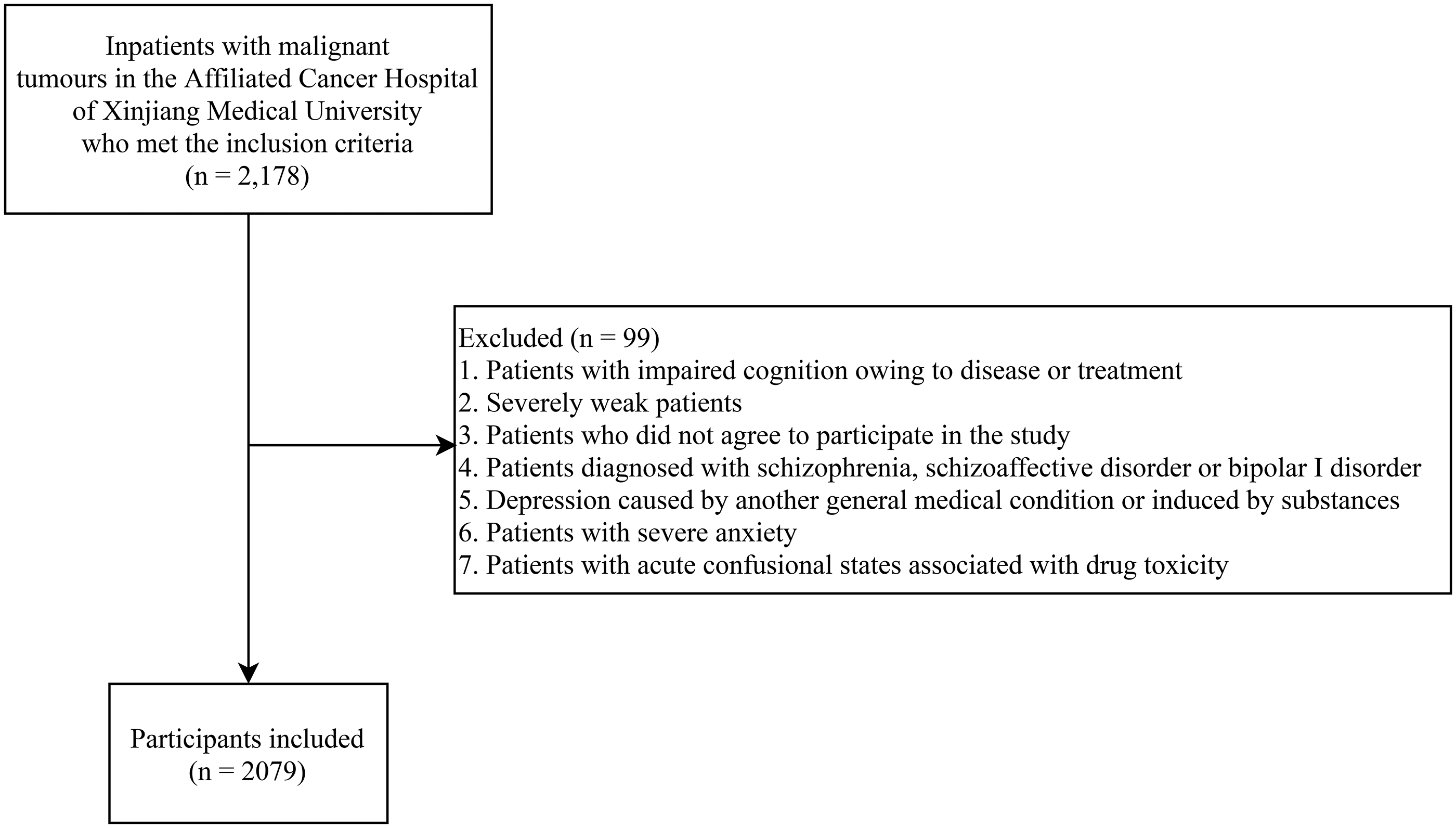
Participant screening process.
Baseline data for 2079 cancer patients.

KPS, Karnofsky Performance Status.
Univariable analysis for malignant tumour patients with depressive symptoms
A univariable analysis was conducted to compare the characteristics of malignant tumour patients with or without depressive symptoms. The results showed significant differences between the depressive symptoms group and the non-depressive symptoms group on sex (χ2 = 13.918, P < 0.001), age (χ2 = 61.662, P < 0.001), place of residence (χ2 = 31.634, P < 0.001), education (χ2 = 125.227, P < 0.001), marital status (χ2 = 19.798, P < 0.001), monthly family income (χ2 = 199.74, P < 0.001), work status (χ2 = 92.701, P < 0.001), physical exercise (χ2 = 214.075, P < 0.001), KPS score (P < 0.001), cancer stage (χ2 = 119.365, P < 0.001), disease course (χ2 = 57.251, P < 0.001), therapy (χ2 = 102.383, P < 0.001), disease state (χ2 = 191.774, P < 0.001), tumour metastasis (χ2 = 134.579, P < 0.001), pain (χ2 =84.292, P < 0.001), condition awareness (χ2 = 128.204, P < 0.001), history of hypertension (χ2 = 5.598, P < 0.001) and type of cancer (χ2 = 10.317, P < 0.001) (Table 2).
Univariable analysis of factors associated with depressive symptoms in cancer patients.
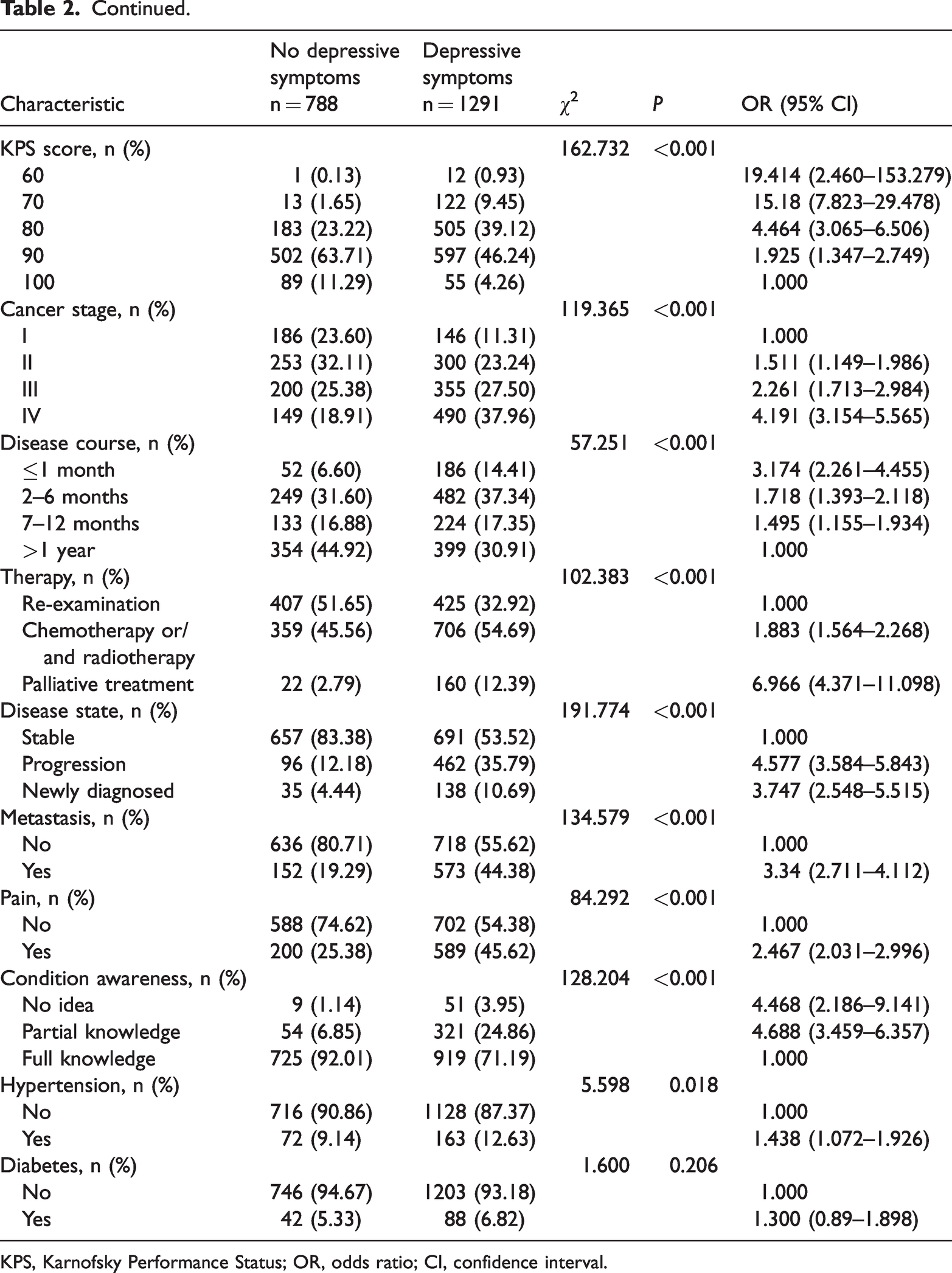
KPS, Karnofsky Performance Status; OR, odds ratio; CI, confidence interval.
Logistic regression analysis for depressive symptoms in malignant tumour patients
The data showed that the following factors increased the risk of depressive symptoms in malignant tumour patients: monthly family income ≤5000 yuan (OR = 4.966, 95% CI: 2.938–8.395, P < 0.001) and 5001 to 10,000 yuan (OR = 3.111, 95% CI: 1.840–5.260, P < 0.001), KPS score of 70 (OR = 2.783, 95% CI: 1.281–6.042, P = 0.010) and 80 (OR = 1.834, 95% CI: 1.139–2.953, P = 0.013), disease course ≤12 months (P < 0.05), palliative treatment (OR = 2.288, 95% CI: 1.292–4.055, P = 0.005), progressive disease (OR = 1.876, 95% CI: 1.284–2.739, P = 0.001), pain (OR = 1.973, 95% CI: 1.555–2.505, P < 0.001), cancer type lung (OR = 3.199, 95% CI: 1.938–5.279, P < 0.001), oesophagus (OR = 3.288, 95% CI: 1.673–6.464, P < 0.001) and cervix (OR = 1.542, 95% CI: 1.056–2.253, P = 0.025) and partial knowledge of disease condition (OR = 2.366, 95% CI: 1.653–3.385, P < 0.001). Return to work (OR = 0.503, 95% CI: 0.348–0.727, P < 0.001) and physical exercise (OR = 0.437, 95% CI: 0.347–0.551, P < 0.001) reduced the risk of depressive symptoms in patients with malignant tumours (Table 3).
Stepwise logistic regression analysis of factors associated with depressive symptoms in cancer patients.

KPS, Karnofsky Performance Status; SE, standard error; OR, odds ratio; CI, confidence interval; AIC, Akaike information criterion.
Subgroup analysis of different tumour types
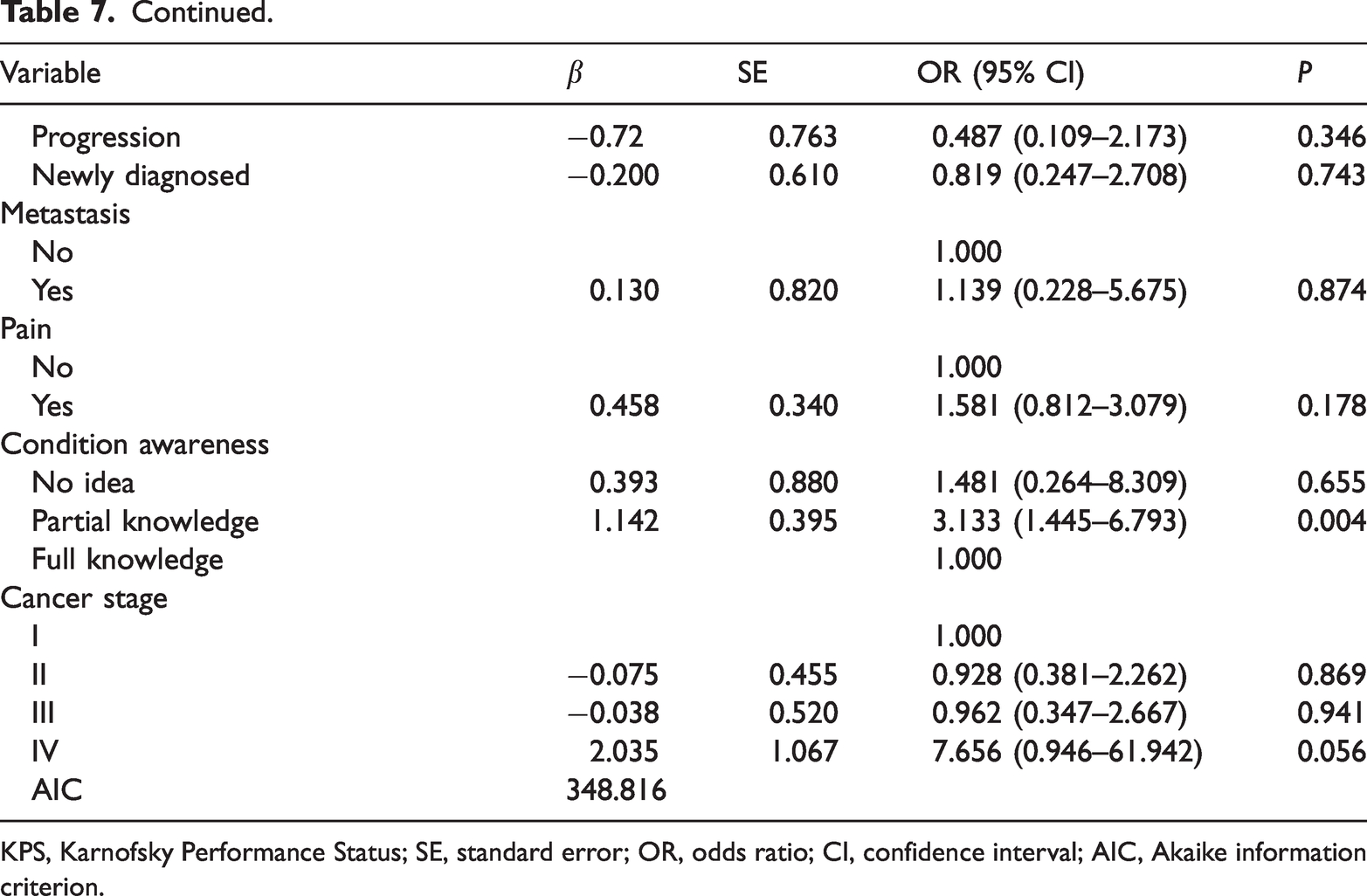
As shown in Table 4, monthly family income ≤10,000 yuan (P < 0.05), KPS score of 80 (OR = 3.552, 95% CI: 1.685–7.488, P < 0.001), progressive disease (OR = 2.994, 95% CI: 1.144–7.836, P = 0.025), pain (OR = 2.143, 95% CI: 1.377–3.335, P < 0.001) and partial knowledge of disease condition (OR = 3.017, 95% CI: 1.275–7.141, P = 0.0102) increased the risk of depressive symptoms in breast cancer patients. Return to work (OR = 0.452, 95% CI: 0.240–0.848, P = 0.013) and physical exercise (OR = 0.478, 95% CI: 0.307–0.745, P = 0.001) were protective factors for depressive symptoms in breast cancer patients. In lung cancer patients, progressive disease (OR = 3.801, 95% CI: 1.422–10.165, P = 0.008) was a risk factor for depressive symptoms whereas return to work (OR = 0.216, 95% CI: 0.046–1.007, P = 0.051) and physical exercise (OR = 0.270, 95% CI: 0.121–0.602, P = 0.001) were protective factors for depressive symptoms (Table 5). For patients with colorectal cancer, pain was a risk factor for depressive symptoms (OR = 2.053, 95% CI: 1.097–3.842 P = 0.024) and physical exercise reduced the risk of depressive symptoms (OR = 0.357, 95% CI: 0.193–0.663, P = 0.001) (Table 6). In addition, monthly family income ≤10,000 yuan (P < 0.05), disease duration of 7 to 12 months (OR = 9.045, 95% CI: 1.427–57.318, P = 0.019) and partial knowledge of their disease condition (OR = 3.133, 95% CI: 1.445–6.793, P = 0.004) increased the risk of depressive symptoms in cervical cancer patients (Table 7).
Logistic regression analysis of related factors for breast cancer population.

KPS, Karnofsky Performance Status; SE, standard error; OR, odds ratio; CI, confidence interval; AIC, Akaike information criterion.
Logistic regression analysis of related factors for lung cancer population.

KPS, Karnofsky Performance Status; SE, standard error; OR, odds ratio; CI, confidence interval; AIC, Akaike information criterion.
Logistic regression analysis of related factors for colorectal cancer population.
KPS, Karnofsky Performance Status; SE, standard error; OR, odds ratio; CI, confidence interval; AIC, Akaike information criterion.
Logistic regression analysis of related factors for cervical cancer population.

KPS, Karnofsky Performance Status; SE, standard error; OR, odds ratio; CI, confidence interval; AIC, Akaike information criterion.
Power analysis and sample size determination
The G* Power software (Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany) and the existing sample size were used for power analysis to test the efficiency of the backward stepwise regression model. The results showed that of the main study variables (monthly family income, work status, physical exercise, tumour metastasis and pain), the lowest statistical power value was 0.959 for tumour metastasis, indicating that the sample size was sufficient for good performance of the backward stepwise regression model.
Discussion
This study collected clinical data from 2079 malignant tumour patients and evaluated the risk factors for depressive symptoms in those patients. The results showed that monthly family income ≤10,000 yuan, KPS score of 70 and 80, disease course ≤1 year, palliative treatment, progressive disease, pain and partial knowledge of disease condition were risk factors for depressive symptoms in malignant tumour patients. Return to work and physical exercise were protective factors for depressive symptoms in patients with malignant tumour.
In this study, higher family income levels were associated with a lower risk of depressive symptoms. Patients with higher incomes experience less worry about the economic burden of cancer treatment and fewer depressive symptoms, and their family burden may be lower than that of patients with lower incomes. 24 Sharp and her colleagues found that 49% of 654 cancer patients had a heavy treatment-related economic burden and family income was an independent risk factor for depressive symptoms in malignant tumour patients. 25 The KPS score reflects patient function; lower scores indicate worse health status. 26 Several studies have indicated that lower KPS score is an independent prognostic factor for survival in cancer patients;27,28 thus, low function may increase patients’ fear and anxiety. In this study, KPS scores of 70 and 80 increased the risk of patient depressive symptoms by 1.783 and 0.834 times, respectively. Additionally, depressive symptoms were related to disease course. Patients with a disease course ≤1 year had a higher risk of depressive symptoms. This may be because patients with a longer disease course have passed the initial period of disease denial, have begun to accept the reality of the disease and are willing to actively cooperate with treatment. 8 Palliative treatment provides symptomatic support for the clinical symptoms of cancer patients; although it relieves clinical symptoms, it cannot control the further development and metastasis of malignant tumours. 29 In this study, patients receiving palliative treatment had a higher risk of depressive symptoms. This study also showed that progressive disease and tumour metastasis affected the risk of depressive symptoms in patients with malignant tumour, which supports previous study findings. 30 This may be because patients with progressive disease and tumour metastasis experience greater psychological anxiety and despair. According to data from a meta-analysis, 55% of patients receiving anticancer treatment and 64% with advanced metastatic or terminal disease experience pain. 31 Studies have shown that pain substantially affects the psychology of patients, and moderate to severe pain leads to severe depressive symptoms in patients. 32 The present findings showed that pain increased the risk of patient depressive symptoms by 0.973 times, indicating that pain is an independent risk factor for depressive symptoms in patients with malignant tumours. Patients experiencing pain may have inflammation, which is associated with a high risk of depression because inflammation can induce neurogenesis and synaptic plasticity impairment. 33 Clinicians should pay attention to inflammation in cancer patients to prevent inflammation-induced stress and depressive symptoms. 34 A study by Ghoshal et al. indicated that patients with cancer prefer to have full knowledge of their diagnoses and prognoses. 35 Deeper understanding of their disease increases patient compliance. Patients who are unaware or partly informed about their disease may be fearful and therefore have a higher risk of cancer-related negative emotions than those who are informed. In the present study, different cancer types had different effects on the risk of depressive symptoms. Compared with breast cancer patients, lung, oesophageal and cervical cancer patients had a higher risk of depressive symptoms. Previous studies have estimated that the prevalence of depressive symptoms in lung, oesophageal and cervical cancer patients is very high.16,36 This may be because the incidence of these kinds of tumours is also very high, which may lead to a high prevalence of depressive symptoms in people with these cancers.
One previous study demonstrated that depressive symptoms in cancer patients improved over 18 months after return to work. 37 The present findings support this and indicate that return to work can reduce the risk of depressive symptoms in patients with malignant tumours. This may be because work increases family income and relieves the financial pressure caused by cancer treatment. Cancer patients who return to work experience a richer environment and more social interactions. This can improve neurogenesis in the dentate gyrus of the hippocampus, which is required for learning, memory and emotional behaviours, and reduce the risk of depression in cancer patients. 38 Additionally, work distracts patients and gives them less time to think about their disease. Physical exercise was another protective factor of depressive symptoms in patients with malignant tumours in this study. Physical exercise reduces the risk of depressive symptoms and improves the quality of life of cancer patients. 39 Physical exercise reduces the risk of depressive symptoms in cancer patients because it improves immune function and relieves patients’ anxiety and tension; this further improves sleep and appetite, promotes digestion and increases the body’s tolerance to pain.40,41 Using a mouse model, Crupi et al. showed that brain inflammation, impairment and hippocampal plasticity are associated with anxiety and depression. 42 Cognitive function and neurogenesis may be impaired in cancer patients because of chemotherapy, which may cause mood disorders and depressive symptoms in these patients. 43 Exercise improves brain function and brain plasticity, especially adult hippocampal neurogenesis, and so could be a useful novel treatment method for depression. 44 Patients with maligant tumours should engage in appropriate exercise to improve brain function, sleep quality and appetite to prevent the occurrence of depressive symptoms. If possible, patients with malignant tumours should go back to work and take part in more social activities to prevent the occurrence of depressive symptoms.
The present study identified several factors that affected depressive symptom status in patients with cancer. These findings may help in quickly identifying cancer patients with a high risk of depressive symptoms. Patients diagnosed with cancer may experience psychosocial and physical pressure. 45 Depressive symptoms lead to poor treatment response, increase hospital admission rate and are associated with poorer prognosis and higher cancer mortality; most worryingly, they can also lead to mortality and suicide. 46 Therefore, clinicians must take timely preventive measures against depressive symptoms in cancer patients with a high risk of such symptoms, and reduce the occurrence rate of depressive symptoms using psychological counselling. Malignant tumour patients with depressive symptoms should receive appropriate psychosocial interventions, psychological treatment or antidepressants, which may help to improve adherence to cancer treatments and reduce the poor prognosis rate in cancer patients. Social support should be provided for patients with lower incomes and heavy family burdens.
There were some study limitations. First, the study included a relatively small number of patients (especially for some subgroups) from a single institution, which may have reduced the statistical power. The findings should be generalized with caution. Second, depressive symptoms were assessed using a self-report patient rating scale, which may have resulted in uncontrolled bias. Third, some important variables associated with depression were not assessed. For example, specific temperament traits predict perceived stress, 47 indicating that people with different temperament traits may have different risks of depression. Additionally, one previous study showed that temperament traits correlate with depression status in cancer patients receiving palliative care. 48 Fourth, there are currently no structured interviews or validated assessment tools for neuropsychiatric disorders, which may reduce the validity of the exclusion criteria for these patients and may have reduced the statistical power of our study. To confirm the present findings, more well-designed studies are required using longer scales and a wider range of reliable variables.
Conclusion
In this study, we evaluated the factors affecting the risk of depressive symptoms in malignant tumour patients. The results showed that monthly family income ≤10,000 yuan, KPS scores of 70 and 80, disease course of ≤1 year, palliative treatment, progressive disease, pain, partial knowledge of disease condition, return to work and physical exercise were influencing factors for depressive symptoms in patients with malignant tumours. These findings may help clinicians to conduct timely preventive interventions for cancer patients with a high risk of depressive symptoms. Additionally, these results could inform the use of appropriate treatments, including psychosocial interventions and antidepressants, for malignant tumour patients with depressive symptoms.
Footnotes
Declaration of Conflicting Interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was supported by the Xinjiang Uygur Autonomous Region 13th Five-Year Plan key discipline construction project.
