Abstract
Objective
To quantitate plasma interleukin-6 (IL-6) levels in healthy individuals and to clarify how these levels are affected by blood sample handling procedures during short-term storage.
Methods
Ethylenediaminetetraacetic acid (EDTA)-treated plasma samples were simultaneously collected from 14 healthy individuals and stored on ice prior to analysis of the IL-6 levels. White blood cells (WBCs), red blood cells, and platelets were counted immediately after blood collection. IL-6 levels were analyzed every 30 minutes using a commercial electrochemiluminescence immunoassay.
Results
Correlation coefficients between plasma IL-6 levels and WBC counts ranged between 0.605 and 0.554, higher than those for other cell types. The lowest IL-6 value in healthy individuals was estimated at 0.04 pg/mL and the mean values remained under 2 pg/mL over time.
Conclusion
Analysis of IL-6 levels in EDTA-treated plasma samples centrifuged within 1 hour and stored on ice can be performed within 90 minutes of short-term storage if the analytical method has a sensitivity in the range of 10 fg/mL.
Introduction
Cytokines are key components of the innate and adaptive immune system and regulate host responses to infection, inflammation, and trauma. 1 More than 300 different cytokines have been identified. As pivotal players in the progression or regression of pathological processes, these molecules represent biomarkers of several diseases. 2 Some cytokines increase the severity of disease (inflammatory cytokines) whereas others serve to reduce inflammation and promote healing (anti-inflammatory cytokines). Interleukin-6 (IL-6) is an inflammatory cytokine released during the acute phase of inflammation and plays a role in various biological activities including cell growth and differentiation. 3 IL-6 may be a useful biomarker of the severity of infection in patients receiving intensive care for conditions such as coronavirus disease 2019 (COVID-19). 4 , 5
Cytokines are produced by cells including white blood cells (WBCs) and platelets (PLTs) during in vitro storage. 6 Muylle et al. observed that cytokine levels increased with storage time in blood used for transfusion. 7 The techniques used to analyze cytokine levels in blood samples are critically important. Many studies have used serum for analyses of biomarkers; however, plasma is more suitable for cytokine measurements because coagulation is linked to inflammation during immunothrombosis. 8 Hennø et al. suggested that cytokines should be analyzed in blood samples that contain the anticoagulant ethylenediaminetetraacetic acid (EDTA), are stored on ice, and are centrifuged within 4 hours. 9 During diagnosis of infections in emergency medicine, it is important that biomarker levels be stable for 1 hour after blood collection. However, studies of IL-6 levels have been conducted within such a short period because it is difficult to process samples within a few tens of minutes after sample collection. The possibility of temporal fluctuations during this short storage period must be considered.
In addition, many cytokines, including IL-6, are often present at ultra-low levels (<1 pg/mL) in healthy individuals and are thus difficult to measure using conventional techniques. 10 To overcome this limitation, high-sensitivity assays and appropriate data processing are required. The electrochemiluminescent immunoassay (ECLIA) is a powerful analytical tool used for clinical testing. 11 , 12
The primary aim of our study was to estimate plasma IL-6 levels in healthy individual and to assess changes in plasma IL-6 levels over time during short-term storage. We evaluated IL-6 levels in EDTA-treated plasma stored for 90 minutes. IL-6 levels were analyzed four times in 30-minute increments, taking advantage of a facility where the clinical laboratory and sample collection room were adjacent to one another. Ultra-low levels of IL-6 in healthy individuals were estimated using ECLIA and a nonlinear calibration curve. In addition, we explored relationships between IL-6 levels and numbers of WBCs, red blood cells (RBCs), and PLTs.
Materials and Methods
Participants
This study was approved by the Ethics Committees of Shinshu University and Shinshu University Hospital, Japan (No. 4936). Written informed consent was obtained from healthy non-smoking adult male individuals who were not using any medications and had not experienced fever or other symptoms of infectious or inflammatory illness during the 2 weeks prior to blood sampling.
Because few studies have investigated IL-6 levels during short-term storage, it was difficult to determine the minimum required sample size. Thus, the required sample size was estimated based on the results of similar studies. Bayraktaroglu et al. observed significant cytokine accumulation during 5 days of storage of WBC-contaminated platelet concentrates from 11 individuals. 13 Additionally, a study of cytokine concentrations in 16 individuals found significant correlations (correlation coefficients 0.932 to 0.992) between different storage times. 14 Based on effect size guidelines suggested by Cohen, 15 the number of samples for linear multiple regression required to achieve a power of 0.8 at a two-tailed significance level of 0.05) with an effect size between 0.3 and 0.8 was 14.
Sample collection and analyses of IL-6 levels
To examine the effects of short-term storage on IL-6 levels, venous blood was collected into a vacuum blood collection tube (4- × 5-mL tubes containing EDTA as an anticoagulant; Terumo Co., Tokyo, Japan). Prior to blood collection, 14 healthy participants and medical staff were on standby. To standardize the analysis times, venous blood samples were collected from all participants at the same time by assigning one staff member to each participant. Blood samples were stored on ice to exclude the effects of temperature differences prior to analyses. Immediately after collection, blood samples were carried to a laboratory and centrifuged using a refrigerated centrifuge (Model 6200, Kubota Co., Tokyo, Japan) at 2000 × g for 10 minutes at 4 °C to obtain plasma. The completion of this centrifugation step was defined as 0 minutes (Figure 1). The numbers of WBCs, RBCs, and PLTs were measured using a blood cell analyzer (XN-9000, Sysmex Co., Tokyo, Japan). Next, 30 µL of plasma was analyzed every 30 minutes using an ECLIA platform (Cobas 8000 [e602], Roche Diagnostics K.K., Tokyo, Japan) and 96-spot IL-6 ECLIA plates (Elecsys IL-6, Roche Diagnostics K.K.).

Experimental protocol for plasma IL-6 analyses.
Clinical information and results from sample analyses were collected and entered in a structured database by research staff who had no direct contact with study participants.
Calibration curve
IL-6 levels were determined using a nonlinear calibration curve. A four-parameter logistic model has been developed as a powerful technique for nonlinear calibration.
16
,
17
This method offers improved statistical estimation because of the bias-variance trade-off that occurs when the true curve is asymmetric. The calibration curve was fitted using a four-parameter logistic model. In this model, y (signal from the ECLIA platform), was expressed in terms of the IL-6 level, x (pg/mL), as follows:
Statistical analyses
All analyses were performed using Statistical Package for Social Sciences (SPSS) version 25 (Advanced Analytics, Inc., Tokyo, Japan). Changes in IL-6 levels over time were evaluated using one way analysis of variance (ANOVA). Unless stated otherwise, all data were expressed as means ± standard deviations (SDs). Values of P < 0.05 were considered statistically significant.
Results
Table 1 summarizes the clinical characteristics of participants and cell counts immediately following centrifugation of plasma. Fourteen healthy adult male individuals participated in the study. The mean ± SD age, height, weight, and body mass index (BMI) was 34.8 ± 2.8 years (range: 32 to 39 years), 173.5 ± 6.3 cm, 69.5 ± 10.3 kg, and 23.1 ± 3.2 kg/m2, respectively. Mean ± SD BMI was lower than the threshold for Asians for overweight/obese. 18 Mean ± SD counts of WBCs, RBCs, and PLTs were 4.97 ± 1.09 × 103/µL, 5.29 ± 0.22 × 106/µL, and 24.0 ± 4.6 × 104/µL, respectively. Mean ± SD counts of neutrophils, lymphocytes, monocytes, eosinophils, and basophils were 2.81 ± 0.84 × 103/µL, 1.66 ± 0.32 × 103/µL, 0.27 ± 0.06 × 103/µL, 0.19 ± 0.12 × 103/µL, and 0.04 ± 0.02 × 103/µL, respectively. Neutrophils accounted for 56% of WBCs. The mean ± SD hematocrit was 47.5 ± 1.7%. These results were within the normal range for healthy Japanese individuals. 19
Clinical characteristics of participants and cell counts in plasma samples immediately following centrifugation.
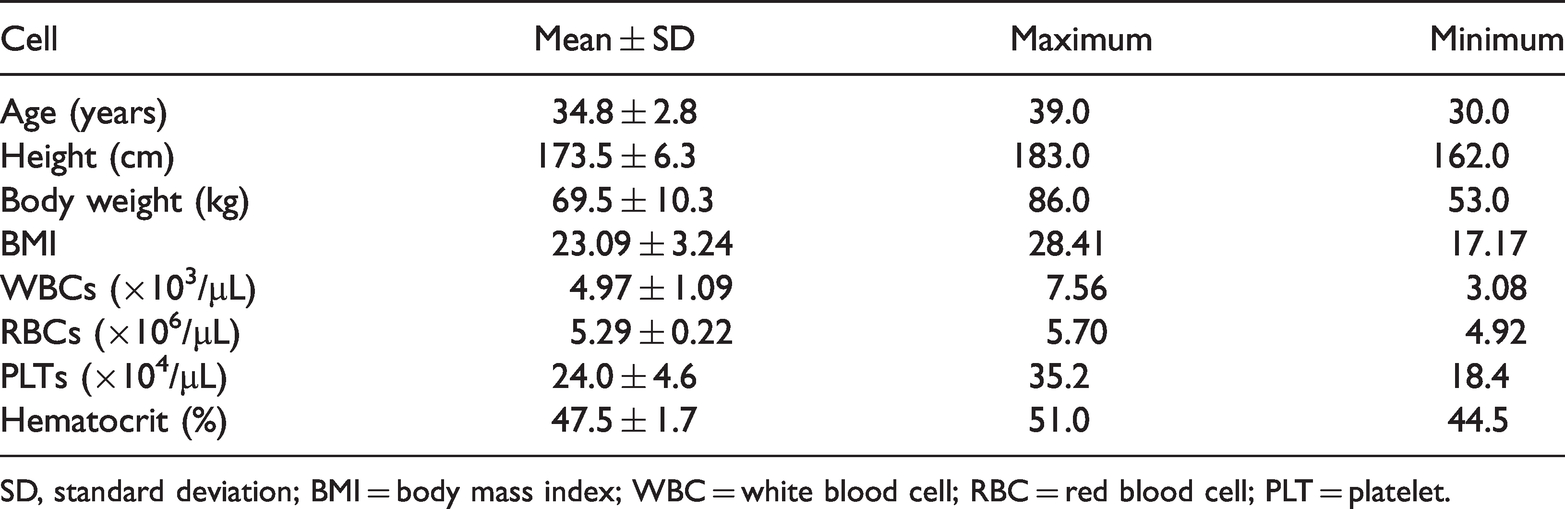
SD, standard deviation; BMI = body mass index; WBC = white blood cell; RBC = red blood cell; PLT = platelet.
IL-6 levels were estimated using an ECLIA and a four-parameter logistic model. Of a total of 56 data points, 7 data points could not be analyzed because the IL-6 levels were below the lower limit of the calibration curve. Changes IL-6 levels over time in samples stored for 90 minutes after collection are shown in Figure 2. IL-6 levels ranged from 0.04 to 8.82 pg/mL in healthy individuals. The mean ± SD IL-6 levels at 0, 30, 60, and 90 minutes were 1.85 ± 2.22, 1.75 ± 2.19, 1.41 ± 2.18, and 1.45 ± 2.15 pg/mL, respectively. The mean values tended to decrease slightly; however, no statistically significant differences were observed by one-way ANOVA.
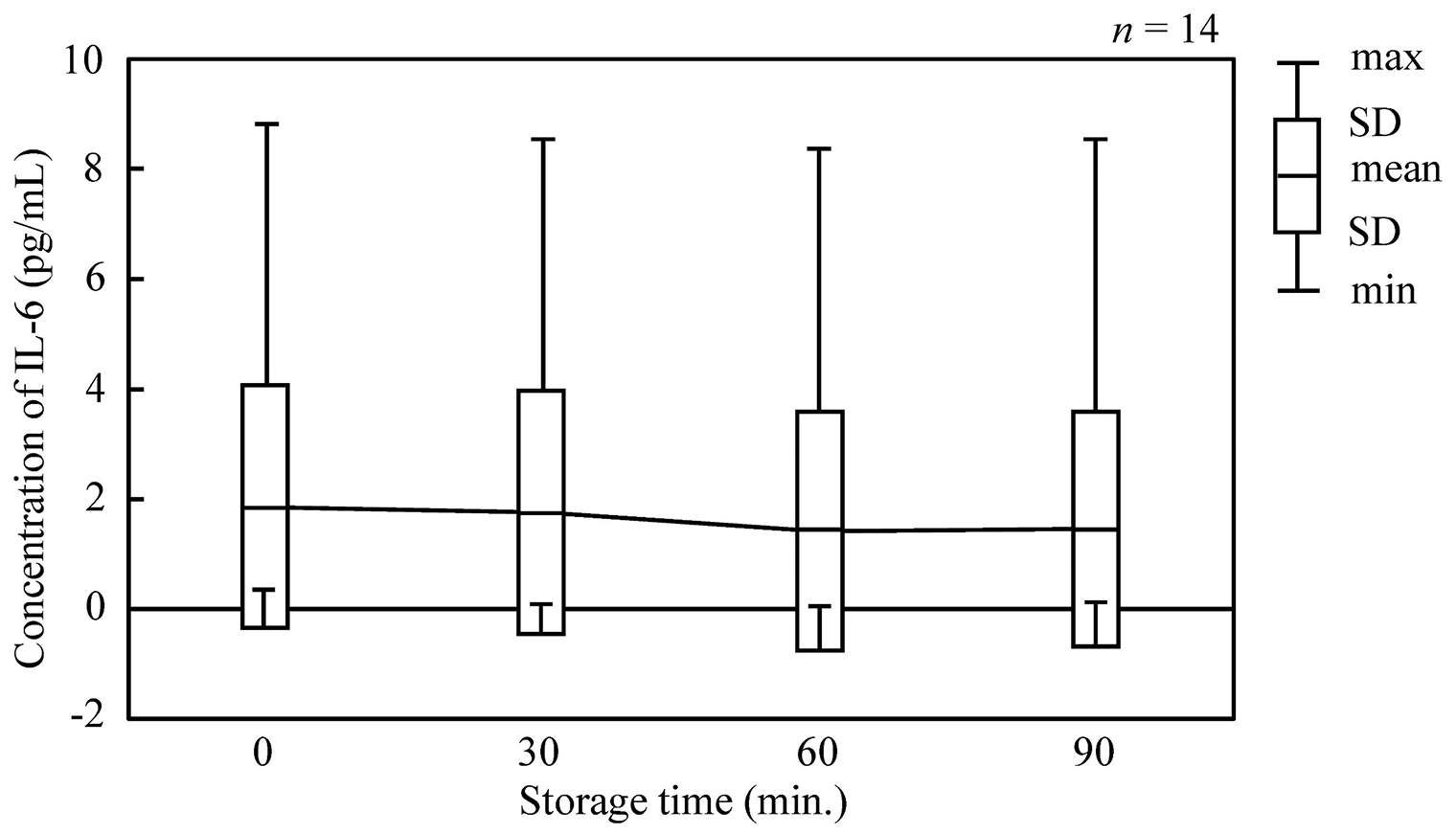
Time-course changes in plasma concentrations of IL-6 after blood collection.
The correlation coefficients, R, between IL-6 levels and cell counts are shown in Figure 3. Correlation analyses revealed significant differences (P < 0.05) at 0 and 60 minutes for WBCs and at 0 and 30 minutes for RBCs. PLTs showed no significant differences over time. IL-6 levels correlated most directly with numbers of WBCs, although the R values varied only slightly over time (between 0.605 and 0.554). For PLTs, R values increased from 0.300 to 0.377 over time, while for RBCs, R values decreased slightly from 0.628 to 0.450.

Correlations between plasma levels of IL-6 and cell counts.
Discussion
To standardize the analyses in our study, we collected all venous blood samples at the same time by assigning one staff member to each participant. IL-6 levels were measured using a highly sensitive ECLIA method, and calibration curves were fitted using a four-parameter logistic model.
It was expected that WBC, RBC, and PLT counts should be quite stable in plasma. Thus, cell counts were compared with IL-6 levels. We calculated correlation coefficients for comparisons between plasma IL-6 levels and cell counts and found that for the correlation between IL-6 and WBCs, R ranged from 0.554 to 0.605, higher than for other cell types. The correlation coefficients for RBCs gradually decreased over time, presumably because RBCs do not secrete IL-6. This finding is consistent with the results of previous studies. It is not clear why the correlation coefficient between IL-6 levels and RBC counts increased at the 30-minute time point in this study. Because cell counts were only conducted immediately after centrifugation, changes in correlations over time are only influenced by changes in IL-6 levels. In addition, the correlation coefficients suggested remarkable effects of PLT counts on changes over time in IL-6 levels. Because of an effect of temperature, Hennø et al. recommended that whole blood samples be placed on ice (0 °C) following collection and immediately centrifuged. 9 The results of our study showed that IL-6 levels are stable in plasma stored on ice for short periods (<90 minutes) if EDTA is added to the blood sample and it is centrifuged within 1 hour.
Quantitation of IL-6 levels in healthy individuals revealed ultra-low mean values (< 2 pg/mL). If one wished to designate the results from healthy individuals as negative, it may be appropriate to set the cut-off as 1.5 pg/mL. However, the minimum value for plasma IL-6 levels was estimated to be as low as 0.04 pg/mL. Therefore, analytical devices need to achieve a sensitivity in the range of 10 fg/mL to reliably quantify plasma IL-6 levels.
There were several limitations to our study. We studied only small volume plasma samples from healthy individuals. In future work, we intend to conduct similar analyses in patients with higher IL-6 levels and in cerebrospinal fluid.
Conclusion
High plasma levels of IL-6 suggest the presence of disease, and thus this molecule serves as a potential biomarker of disease severity. 20 However, several factors, including storage time and temperature, as well as analytical accuracy, affect the reliability of IL-6 analyses. It can be particularly challenging to analyze plasma IL-6 levels shortly after blood collection. In this study, we present guidelines for analyzing IL-6 levels in EDTA-treated plasma samples centrifuged within 1 hour and stored on ice. Measurement can be performed within 90 minutes during short-term storage if the analytical method has a sensitivity in the range of 10 fg/mL.
In addition, we assessed how handling and processing clinical blood samples affects ultra-low plasma levels of IL-6. Although our findings are based on a small number of participants, our data suggest that highly sensitive assays and appropriate data processing are required for analyses of IL-6 levels. Analytical methods to precisely measure ultra-low concentrations of IL-6 in the range of 10 fg/mL merit further development.
Footnotes
Acknowledgment
We thank the healthy individuals who participated in this study for their support.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by a grant from the Japan Society for the Promotion of Science (grant number 20H04514 to M. Yamaguchi).

