Abstract
Objective
To compare the effectiveness of dynamic stratified potassium supplementation at high concentrations with enteral potassium supplementation in older patients with chronic heart failure and moderate to severe hypokalaemia.
Methods
We performed a single-centre, short-term, randomised, controlled, open-labelled, clinical trial, and patients were randomly allocated to the control or intervention group. The intervention group received intermittent infusions of 30 mmol/100 mL potassium chloride. In the control group, 10% potassium chloride was administered orally in a bolus dose. Short-term efficacy and adverse events were compared.
Results
The intervention group received less potassium than that in the control group. T-wave normalisation and U-wave disappearance occurred sooner in the intervention group than in the control group after potassium supplementation. The rate of increase in potassium concentrations gradually became similar in both groups. The initial blood potassium concentration, method of potassium supplementation, potassium supplement dose, and 24-hour urinary potassium excretion significantly affected the rate of increase in blood potassium concentrations after supplementation.
Conclusions
The efficacy of enteral potassium supplementation is equivalent to that of supplementation with high intravenous potassium concentrations in elderly patients with chronic heart failure and moderate to severe hypokalaemia. High intravenous potassium concentrations may lead to a superior potassium recovery rate.
Introduction
Patients with chronic heart failure (CHF) often have abnormal potassium concentrations. This abnormality is caused in part by elevated aldosterone concentrations after neurohormonal activation, inappropriate use of diuretics, and modern high-sodium diets.1–3 The prevalence of hypokalaemia varies between 19% and 54% in patients with heart failure. 4 A serum potassium concentration <4 mmol/L is related to an increased risk of cardiac arrhythmia in patients with heart failure and may be associated with an increased risk of all-cause mortality in patients with CHF.1,5,6 In a multiple-risk-factor intervention trial, Cohen et al. found that even a 1 mmol/L decrease in serum potassium concentrations would lead to a 28% increase in the incidence of ventricular arrhythmias. 7 Leier et al. recommend maintaining high-normal potassium concentrations of 4.5 to 5.0 mmol/L, which may reduce all-cause mortality in patients with CHF. 8 Therefore, maintaining an ideal concentration of blood potassium is necessary in patients with CHF. 9
Intravenous potassium supplementation is the preferred method of implementation, despite the various adverse effects of inappropriate administration, which include cardiac death. 10 Deep venous infusion of a high concentration of potassium can improve the blood potassium concentration more quickly than other potassium supplementation methods and can improve the abnormal cardiac electrical activity caused by early-stage hypokalaemia. 11 However, data on the efficacy of enteral potassium administration in elderly patients with CHF and moderate to severe hypokalaemia are lacking.
This study aimed to compare the effectiveness of a high concentration of dynamic stratified potassium supplementation with enteral potassium supplementation in patients with CHF and moderate to severe hypokalaemia.
Methods
Participants and eligibility criteria
The present study was a prospective, open-label, randomised, clinical trial. The reporting of this study conforms to the CONSORT statements. 12 We screened all elderly patients with CHF who were admitted to the Emergency Department of the Second Hospital of Hebei Medical University from January 2017 to October 2019. We screened 226 patients, of whom 30 declined to participate and 196 consented to participate. Subsequently, another 23 patients were excluded. The process of recruitment and allocation is shown in Figure 1.
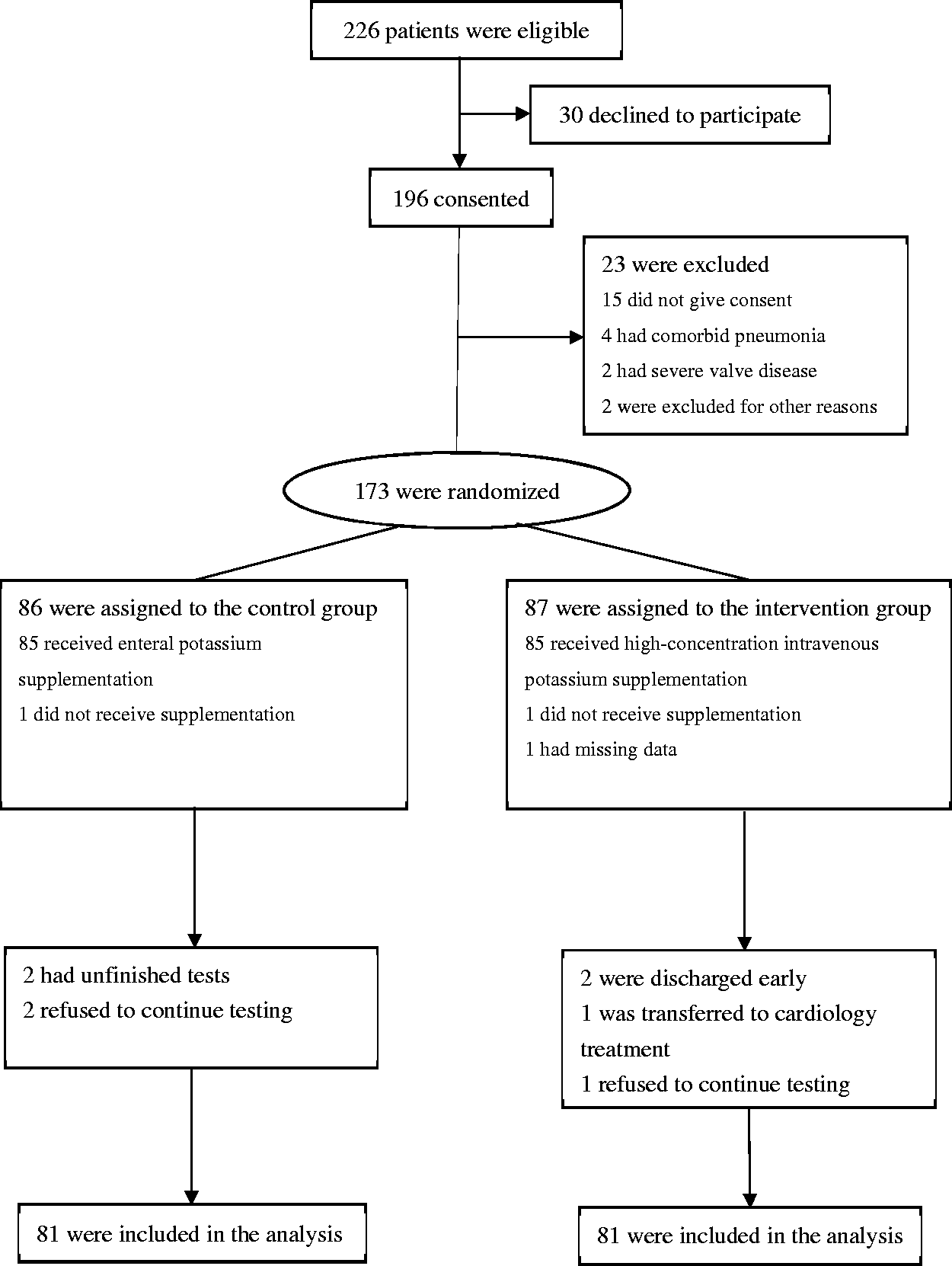
Enrolment and randomization of the patients.
The following inclusion criteria were applied: moderate (2.5–3.0 mmol/L) to severe (<2.5 mmol/L) hypokalaemia; 13 age ≥60 years; CHF of New York Heart Association (NYHA) class II or III; and serum creatinine concentrations ≤221 µmol/L for men and ≤177 µ/L for women. Patients with complications of severe heart, liver, lung, or kidney dysfunction or gastrointestinal absorption dysfunction were excluded. The study complied with the Declaration of Helsinki. The study was approved by the ethics committee of the Second Hospital of Hebei Medical University (approval number: 2017-P037; date of approval: 2017-01-11). We did not originally prospectively register this trial, but it has been registered retrospectively at the Research Registry (https://www.researchregistry.com/: registration number 7165).
Methods and study design
All study subjects signed a written informed consent document. Random number tables were used to allocate research subjects at a 1:1 ratio. The control group was administered 10% potassium chloride oral solution in a bolus dose. The intervention group received intermittent intravenous infusions of 30 mmol/100 mL potassium chloride at a rate of 100 mL/hour 14 from an intravenous infusion pump. Each group was administered three doses of potassium daily. The daily dose of supplemental potassium was adjusted according to the blood potassium concentration and a pre-planned potassium supplementation scheme. The dynamic stratified potassium supplementation doses were calculated according to the serum potassium concentration (Table 1). The speed of the patients’ response to the potassium supplementation regimen was indicated by the rate of increase in potassium concentrations, which was calculated as follows: potassium increase rate = (initial blood potassium concentration after treatment − initial blood potassium concentration before treatment)/initial blood potassium concentration before treatment ×100%. A standard low-fat diet containing the normal amount of potassium was provided to all patients by the hospital’s Nutrition Department throughout the study period.
Dynamic stratified potassium supplementation regimen.

Blood samples were collected 3 hours after each supplemental dose. Potassium supplementation was stopped when a blood potassium value ≥4.5 mmol/L was achieved. Patients who developed critically low potassium concentrations (<2 mmol) after initial oral potassium supplementation were allowed to cross over and receive intravenous potassium. The study lasted 5 days.
Outcome measures
The primary objective was to compare the difference in plasma potassium concentrations between the two groups. Secondary effects were compared during the potassium supplementation period.
The side effects of potassium supplementation, such as hyperkalaemia (potassium concentrations >5 mmol/L), arrhythmia, and gastrointestinal reactions, were monitored and recorded during administration and for 3 hours after administration.
Data collection
Data on the patients’ demographic characteristics, medical history, and medication history were collected. In particular, the administration of angiotensin-converting enzyme inhibitors (ACEIs), β-blockers, angiotensin receptor blockers (ARBs), aldosterone inhibitors, and diuretics was recorded and analysed. Serum potassium concentrations were recorded before and at 3, 6, and 9 hours after potassium supplementation. Furthermore, blood serum creatinine concentrations, arterial pH, 24-hour urinary potassium concentrations measured by a clinical chemistry analyser (Hitachi, Hitachinanaka City, Japan), dose levels of potassium, electrocardiographic (ECG) changes measured by an ambulatory ECG acquisition and analysis system (Shenzhen Boying Medical Instrument Technology Co., Ltd., Shenzhen, China), and adverse effects were monitored and recorded during potassium supplementation.
Statistical analyses
Continuous variables are presented as the mean ± standard deviation, and categorical data are presented as percentages. Continuous variables were subjected to paired t-tests for comparisons within groups or unpaired t-tests for comparisons between groups. Categorical data were compared using the chi-square test or Fisher’s exact test. Analysis of variance was used to evaluate the differences between groups. Multivariate linear regression was performed to evaluate the covariates related to the rate of increase in blood potassium concentrations. The significance threshold was set at p < 0.05 for a confidence level of 95%. Statistical analyses were performed using IBM SPSS software, version 20.0 (IBM Corporation, Armonk, NY, USA).
Results
From 1 January 2017 to 30 October 2019, 173 patients aged ≥60 years with CHF and moderate to severe hypokalaemia fulfilled the criteria and were enrolled in the study. There were 86 patients in the control group and 87 in the intervention group. During the study period, 11 patients withdrew from the trial for various reasons. Ultimately, data from 162 patients were collected and analysed (Figure 1). Finally, there were 81 patients in the control group (mean age, 73.8 ± 5.6 years) and 81 patients in the intervention group (mean age, 75.0 ± 6.4 years). The body mass index was not significantly different between the two groups. Sixty-eight (42%) patients were in NYHA class II and 94 (58%) were in class III. The mean ejection fraction was 27% in the control group and 28% in the intervention group, with no significant difference between the two groups (Table 2).
Characteristics of the patients at baseline.

Values are mean ± standard deviation or number.
BMI, body mass index; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; GFR, glomerular filtration rate; NT-proBNP, N-terminal fragment of the prohormone of brain natriuretic peptide; ECG, electrocardiography.
The participants’ baseline characteristics were well matched between the two groups (all p > 0.05, Table 2). Admission laboratory values were also similar between the groups. The mean serum creatinine concentration was 7566.30 ± 1.56137.90 µmol/L in the control group and 7969.84 ± 0.1614.14 µmol/L in the intervention group (p = 0.15). The N-terminal fragment of the prohormone of brain natriuretic peptide was present in serum, but was not significantly different between the groups. The mean arterial pH value was 7.38 in each group. Most patients were treated with multiple cardiovascular medications. The two groups had similar medication histories. The QRS complex lasted <120 ms in 60% of patients, 120 to 149 ms in 22%, and ≥150 ms in 18%. A total of 106 (65%) patients had abnormal T waves or U waves. Thirty-seven (23%) patients had ST segment depression, and 26 (16%) patients had ventricular extrasystoles.
The mean potassium concentration was 2.38 ± 0.42 mmol/L before replacement and 4.59 ± 0.11 mmol/L after replacement. There was no difference in mean serum potassium concentrations at the beginning or at the end of potassium supplementation between the two groups (Table 3).
Changes in serum potassium concentrations after potassium supplementation.
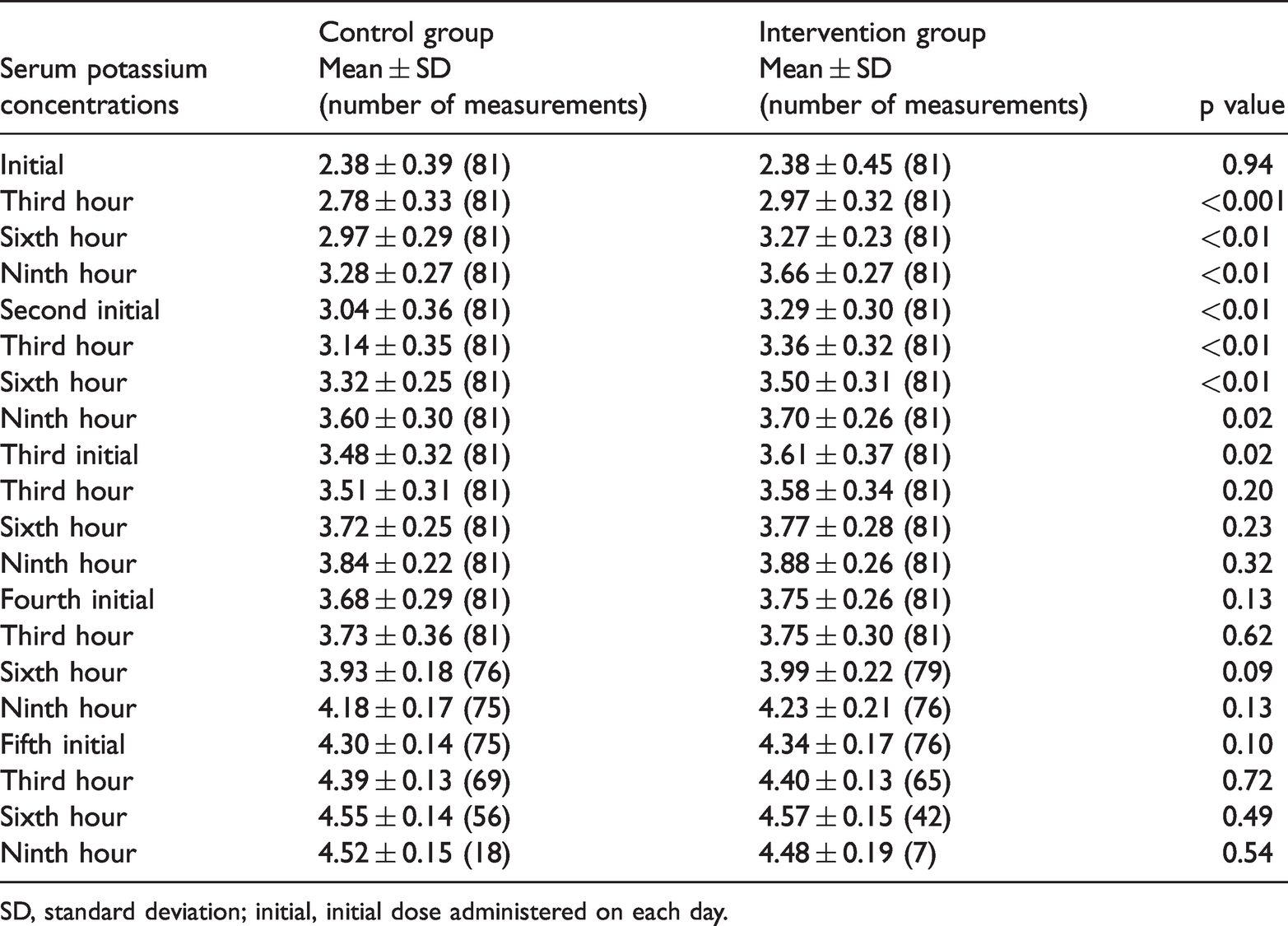
SD, standard deviation; initial, initial dose administered on each day.
Post-supplement potassium concentrations met the target level (≥4.5 mmol/L) in 154 (95%) patients after 2184 episodes of supplementation during the study period. Three patients in the intervention group and five in the control group were excluded because they failed to meet this preset standard. During potassium supplementation, serum potassium concentrations showed a gradual upward trend in both groups. The mean blood potassium concentration in the intervention group was higher from the third hour on the first day until the first potassium dose on the third day of supplementation than that in the control group (all p < 0.05, Table 3). As blood potassium concentrations gradually approached their predetermined target value, the necessary supplemental dose gradually decreased during the study period. The total dose of supplemental potassium in the intervention group (427.40 ± 99.00 mmol) was significantly lower than the total dose in the control group (460.41 ± 48.48 mmol, p = 0.008).
There was no significant difference in urinary excretion between the two groups, except on the second day. Urinary potassium excretion on the second day was significantly higher in the intervention group (87.33 ± 11.69 mmol) than in the control group (82.81 ± 14.59 mmol, p = 0.03).The total amount of urinary potassium excretion during the study period was 408.20 ± 50.02 mmol in the control group and 413.54 ± 53.67 in the intervention group, with no significant difference between the groups.
The intervention group showed a higher rate of increase than that in the control group on the first day (p = 0.002). Interestingly, the increase in rate in the control group was higher than that in the intervention group on the second day (p = 0.003) (Table 4).
Rate of increase in blood potassium concentrations after potassium supplementation.

Values are mean ± standard deviation.
Initial, initial dose administered on each day.
There was no significant difference in the total potassium supplement dose or total urinary potassium excretion depending on whether patients were taking β-blockers, ACEIs, ARBs, thiazide diuretics, aldosterone antagonists, or none of those medications. Patients who were taking loop diuretics had a higher urinary potassium excretion and required a higher total dose of supplemental potassium than those not taking these drugs (both p < 0.05) (Table 5).
Potassium supplementation and urinary potassium excretion in the two groups.

Values are mean ± standard deviation (n).
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker.
Patients were divided into four subgroups according to the group or the history of medication. The rate of increase potassium concentrations did not show significant heterogeneity in the subgroup analyses. There were no significant differences in the number of patients who took β-blockers, ACEIs or ARBs, loop diuretics, thiazide diuretics, aldosterone antagonists, or none of those drugs between the four groups (Table 6).
Intergroup comparison of the rate of increase in potassium concentrations.

We performed multivariate linear regression analysis to identify the factors that affect the rate of increase in potassium concentrations. The variables of sex, age, BMI, hypertension, diabetes, ejection fraction, glomerular filtration rate, β-blockers, ACEIs or ARBs, diuretics, aldosterone antagonists, initial blood potassium concentration, method of potassium supplementation, dose of supplemental potassium, and 24-hour urinary potassium excretion were included in the multiple linear regression model. Among these variables, the initial blood potassium concentration, method of potassium supplementation, dose of supplemental potassium, and 24-hour urinary potassium excretion significantly affected the rate of increase in blood potassium concentrations after potassium supplementation (all p < 0.01) (Table 7).
Multivariate linear regression analysis of the factors affecting the rate of increase in potassium concentrations.

The ECG abnormalities in the two groups showed a trend in improvement after potassium supplementation treatment. On the second day after potassium supplementation, ventricular extrasystole and T-wave and U-wave changes disappeared more quickly in the intervention group than in the control group (both p < 0.05) (Table 8).
Changes in electrocardiographic characteristics during potassium supplementation.

No mortality was observed in the study. Eleven (13.6%) adverse events were observed in the control group, comprising worsening heart failure requiring intravenous diuretics, episodes of new arrhythmia, and gastrointestinal reactions. There were six adverse events in the intervention group, which comprised episodes of worsening heart failure requiring intravenous diuretics, episodes of new arrhythmia, and gastrointestinal reactions. There was no significant difference in the rate of adverse events between the two groups (Table 9).
Adverse events.

aWorsening signs or symptoms of heart failure requiring intravenous diuretics.
bNew and clinically significant atrial or ventricular dysrhythmia.
Discussion
During the 5-day period in this study, 95% of users reached the target potassium concentration without obvious side effects using each of the two potassium supplementation methods. A previous study showed an increased risk of sudden death in older male patients with heart failure. 15 Fluid retention is a classic effect of heart failure that results from impaired cardiac contraction. 16 Therefore, fluid volume should be controlled when treating patients with heart failure. Inappropriate administration of potassium in patients with CHF may aggravate heart failure and lead to cardiac arrest, hyperkalaemia, arrhythmia, or even death. 17 Therefore, supplementation of high potassium concentrations is the most rational way to achieve high-normal potassium concentrations. In this trial, we found that serum potassium concentrations showed a gradual upward trend in both groups. During the study period, no serious complications, such as cardiac arrest, hyperkalaemia, arrhythmia, or death, occurred in either group.
This study showed that supplementation of high potassium concentrations in the intervention group normalised blood potassium more quickly than the enteral potassium supplement provided to the control group. Under normal conditions, the digestive tract can absorb only 77% of the potassium that is consumed. 18 Furthermore, once absorbed, potassium has a half-life of <7 hours.19,20 Poor enteral absorption in patients with CHF may account for the slow recovery of blood potassium with enteral potassium supplementation. Therefore, enteral therapy may not be appropriate for some patients (e.g., those with poor enteral absorption). Previous studies have shown that an early transition from hypokalaemia to normokalaemia in heart failure at 1 month is associated with improved cardiovascular outcomes.2,21,22 Some authors also encouraged enteral potassium supplementation in less critically ill patients 14 on the basis that such treatment is rational.
Patients with CHF typically show hypersecretion of catecholamines and aldosterone,8,23,24 both of which increase urinary potassium excretion. The use of ACEIs/ARBs and β-blockers could prevent hypokalaemia by opposing the neurohumoral activation associated with heart failure.9,25 Potassium depletion is often caused by an increased loss of potassium through the kidneys due to non-potassium-sparing diuretic therapy. 26 In this trial, we analysed the relationship of diuretics with potassium supplementation and urinary potassium excretion. We found no significant difference in the total supplemental potassium required or the total urinary potassium excretion depending on whether patients used β-blockers, ACEIs, ARBs, thiazide diuretics, aldosterone antagonists, or none of those drugs. However, patients who took loop diuretics had a higher urinary potassium excretion and required higher potassium supplement doses than patients who do not take such drugs. Multivariate linear regression analyses showed that β-blockers, ACEIs, ARBs, and diuretics had no significant effect on the rate of increase in serum potassium concentrations. This finding is consistent with previous studies, which showed that ACE inhibitors in patients with CHF were insufficient for protecting against potassium loss.8,27 Some authors recommended using thiazide diuretics only at low doses and adding a potassium-sparing diuretic drug when higher diuretic doses are required to maintain long-term blood potassium stability. 28
Mild to severe hypokalaemia can aggravate ventricular arrhythmia in middle-aged and elderly patients, and this leads to a poor outcome.1,7 Ventricular tachyarrhythmias are highly prevalent in heart failure, with 50% to 80% of patients showing non-sustained ventricular tachyarrhythmias on ambulatory cardiac monitoring. 4 We found that the normalisation of T waves and the disappearance of U waves were faster after central venous potassium supplementation than after control oral treatment, although longer-term follow-up will be necessary. Previous studies have shown that deep venous infusion of high potassium chloride concentrations decrease the frequency of ventricular arrhythmias. 11 For severe potassium disorders especially accompanied by life-threatening cardiac conduction disturbances and neuromuscular dysfunction, intravenous potassium supplementation is still the first choice of treatment. 29 This effect may be related to the instantaneous increase in serum potassium concentrations in the heart. 30 Early detection and treatment allow clinicians to prevent morbidity and mortality from cardiac arrhythmia. 31
Gastrointestinal reactions, which comprised nausea, vomiting, and diarrhoea, were the most common adverse events, accounting for 41.2% (7/17) of the total events. The incidence of adverse events was not significantly different between the two groups over the 5-day study period.
Limitations
The present study has some limitations as follows. One limitation is that our trial was a single-centre study. Another limitation is that heart failure subtypes and acute hypokalaemia and chronic hypokalaemia were not distinguished in this study. Additionally, there were no specific dose data on diuretics. With regard to potassium intake, although a standard low-fat diet was provided, the amount of potassium that patients acquired from the diet could not be accurately calculated. ECGs were also recorded without continuous dynamic monitoring. Finally, the economic benefits of enteral administration were not analysed.
Conclusions
This study shows that the efficacy of enteral potassium supplementation is essentially equivalent to that of high concentrations of intravenous potassium supplementation in elderly patients with CHF and moderate to severe hypokalaemia without gastrointestinal absorption dysfunction or renal dysfunction. However, the rate of potassium normalisation may be superior with high intravenous potassium concentrations.
Footnotes
Acknowledgement
We thank all participating colleagues and nurses for their assistance in data collection.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This work was funded by the province of Hebei Health and Family Planning Commission Science and Technology Project (20180360).
