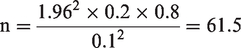Abstract
Objective
The prevalence of Mycoplasma hominis and Ureaplasma urealyticum (genital mycoplasma) amongst Indonesian women is poorly understood because of limited availability of diagnostic techniques. We sought to compare the diagnostic parameters of the AF Genital System® with those of culture methods and PCR as the gold standard for identification of M. hominis and U. urealyticum in vaginal swab specimens.
Methods
This was an observational diagnostic study. Eighty-eight specimens were collected from patients with abnormal vaginal discharge. Detection of M. hominis and U. urealyticum was performed using the AF Genital System®, culture methods, and PCR.
Results
Compared with PCR and culture methods, respectively, the AF Genital System® had sensitivities of 66.6% and 57% (M. hominis) and 55.5% and 77.8% (U. urealyticum). Compared with PCR and culture methods, respectively, the AF Genital System® had specificities of 82.9% and 86.5% (M. hominis) and 82.3% and 84.8% (U. urealyticum).
Conclusion
The sensitivity of the AF Genital System® for detection of M. hominis and U. urealyticum from vaginal swab samples was lower than that of PCR, but specificity was reasonably good (82% to 83%).
Keywords
Introduction
Genital mycoplasmas (Mycoplasma hominis and Ureaplasma urealyticum) are commonly found in the urogenital tracts of healthy, asymptomatic, and symptomatic individuals (both men and women). 1 Symptomatic patients may present with severe urogenital complications such as pelvic inflammatory disease, prostatitis, bacterial vaginosis, amnionitis, postpartum infections, non-gonorrheal urethritis, and even infertility.2–6 However, the presence of any other sexually transmitted infection-causing pathogens must be excluded before proceeding to genital mycoplasma-specific diagnosis and treatment in asymptomatic or symptomatic individuals.7,8 Data regarding the prevalence of M. hominis and U. urealyticum among Indonesian women with abnormal vaginal discharge are lacking, 9 potentially because of limited availability of diagnostics for these infections. Routine diagnosis largely relies on culture methods, polymerase chain reaction (PCR), and rapid tests.10–14 Conventional culture methods for microbiological detection are time-consuming, laborious, cumbersome, and less sensitive while PCR-based methods are costly and require considerable expertise.10–13 Here, we compared the diagnostic parameters of the AF Genital System®, a biochemical test-based diagnostic tool, with those of culture methods and PCR as the gold standard for identification of M. hominis and U. urealyticum in vaginal swab specimens.
Materials and methods
Study protocol and specimens
This observational and diagnostic study was performed to compare the diagnostic parameters of the AF Genital System® (https: //www.liofilchem.com; LIOFILCHEM® s.r.l., Roseto, Italy) with those of culture methods and PCR as the gold standard for identification of M. hominis and U. urealyticum in vaginal swab specimens. Clinical specimens (vaginal and urine samples) were procured from patients who complained of abnormal vaginal discharge and presented at the obstetrics and gynecology and/or genital clinics at The Atma Jaya Hospital and The Royal Taruma Hospital, Jakarta. The inclusion criteria were age 21 to 38 years, no children after 2 years of marriage or longer, and suspected pelvic inflammatory disease. Patients who fulfilled the inclusion criteria but were unwilling to participate or did not sign informed consent forms were excluded from the study. The reporting of this study conformed to STROBE guidelines. 15
Ethics and informed consent
Clinical specimens were obtained with prior written informed consent of the patient. The study was performed according to the standards and guidelines outlined in the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of the School of Medicine and Health Sciences, Atma Jaya Catholic University of Indonesia (approval number: 11/05/KEP-FKUAJ/2017).
Microbiology work-up and identification
The collected samples were subjected to Gram staining3–5 and analysis using the AF Genital System®, culture methods,3–5 and PCR 6 at the microbiology laboratory of The Atma Jaya Hospital. In this study, PCR was used as the gold standard as it is a highly sensitive molecular biology technique that aids in the rapid diagnosis of genital mycoplasmas (e.g., M. hominis and U. urealyticum) using organism-specific primers.
Detection of M. hominis and U. urealyticum using the AF Genital System®
The AF Genital System® comprises a panel of 24 wells, each containing a biochemical substrate (growth indicators) and antibiotics (selection markers). The purpose of the system is to detect, presumptively identify, and assess antimicrobial susceptibility in microorganisms from urogenital samples (urethral and vaginal swabs, urine, and seminal fluid).
M. hominis and U. urealyticum were detected using the AF Genital System® according to the manufacturer’s instructions. Accordingly, swabs (containing vaginal and urine samples) were immersed in physiological saline (provided with the kit) for 5 minutes. Next, 0.2 mL of clinical specimen suspensions were transferred into each well of the AF Genital System®. The inoculated wells were then overlaid with a drop of Vaseline oil and incubated at 36 ± 1°C for 18 to 24 hours. The test results were monitored via the change in color of the inoculated wells.
Detection of M. hominis and U. urealyticum by culture methods
Urogenital swabs were transported in Amies transport medium containing phosphate-buffered saline (Thermo Fisher Scientific, Waltham, MA, USA) to the fully equipped microbiology laboratory at the Microbiology Department, Atma Jaya Hospital, North Jakarta, Indonesia. Clinical specimens were inoculated in agar plates containing the following media (all from Oxoid Ltd., Hampshire, UK): mycoplasma agar base (CM0401), urea agar base (CM0053B), yeast extract solution (LP0021B), urea 40% solution (SR0020K), horse serum (SR0035C), and mycoplasma supplement-G (SR0059). The inoculated plates were incubated at 35 ± 2°C for 48 hours and monitored for up to 14 days under microaerophilic conditions using a candle jar. Finally, Gram staining was performed to identify characteristic mycoplasma colonies (M. hominis and U. urealyticum) by microscopic examination.
Detection of M. hominis and U. urealyticum by PCR
Clinical specimens (vaginal and urine samples) in cotton swabs (cotton was removed from the lid) were added to 400 µL of phosphate-buffered saline, vortexed, and centrifuged. Next, DNA extraction was performed using the QIAamp DNA Mini Kit column (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Purified DNA from these specimens was eluted in 150 µL of double distilled water and stored at −20°C until further use. This served as the template DNA for PCR.
PCR was performed according to the protocol of Sleha et al. with slight modifications.
6
Briefly, the reaction mix comprised 2× HotStarTaq® Master Mix (Qiagen) (2.5 units of HotStarTaq® DNA polymerase, 1× PCR buffer containing 1.5 mM MgCl2, and 0.2 mM of each deoxyribonucleotide triphosphate), 2 µM of each oligonucleotide/primer (10× concentrated), 1 µL of template DNA (added last), and double-distilled water to 25 μL. The primer sequences used in this study were as follows. M. hominis: forward primer 5′-
Sample size calculation
The sample size, ‘n’ was calculated using the formula:
In this equation, ‘Zα’ represents the conversion of the area under the normal curve at a given confidence level (when the accuracy interval applied was 95%); P is the estimated proportion of patients positive for mycoplasma and was assumed to be 20% or 0.2 (based on prior studies); Q equals 1-P (0.8); and d is the degree of precision desired (±10% or 0.1).
Thus,
Sixty-two participants were required based on this calculation.
Results and discussion
Eighty-eight clinical specimens were examined in this study. Detection of M. hominis and U. urealyticum in these specimens using the AF Genital System®, culture methods, and PCR methods is shown in Tables 1, 2, 3, and 4.
Results of M. hominis detection using the AF Genital System® and PCR.

PCR, polymerase chain reaction; PPV, positive predictive value; NPV, negative predictive value.
Results of M. hominis detection using the AF Genital System® and culture methods.

PPV, positive predictive value; NPV, negative predictive value.
Results of U. urealyticum detection using the AF Genital System® and PCR.

PCR, polymerase chain reaction; PPV, positive predictive value; NPV, negative predictive value.
Results of U. urealyticum detection using the AF Genital System® and culture methods.
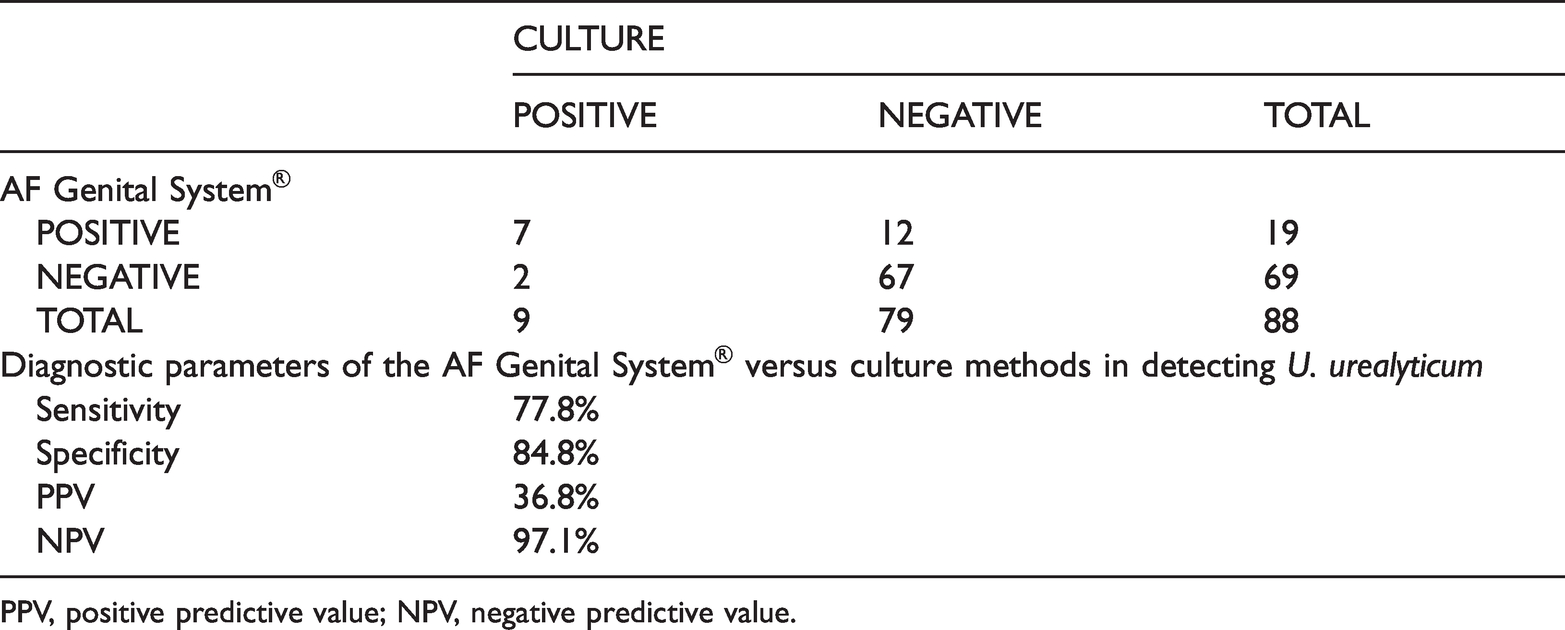
PPV, positive predictive value; NPV, negative predictive value.
The prevalence (positive detection rate) of M. hominis and U. urealyticum in vaginal swabs was 20.5% (18/88 specimens) and 21.6% (19/88 specimens), respectively, using the AF Genital System®. The prevalence (positive detection rate) of M. hominis and U. urealyticum in vaginal swabs was 6.8% (6/88 specimens) and 10.2% (9/88 specimens), respectively, using PCR (Table 1 and Table 3). The prevalence (positive detection rate) of M. hominis and U. urealyticum in vaginal swabs was 15.9% (14/88 specimens) and 10.2% (9/88 specimens), respectively, using culture methods (Table 2 and Table 4). These findings differed from the results of a comparative study by Amirmozafari et al. These authors reported that a higher prevalence of genital mycoplasmas was detected by PCR compared with culture methods, while a few other studies reported lower prevalence.12–14,16
Compared with PCR and culture methods, respectively, the sensitivity of the AF Genital System® in detecting M. hominis was 66.6% (Table 1) and 57% (Table 2) and the sensitivity of the AF Genital System® in detecting U. urealyticum was 55.5% (Table 3) and 77.8% (Table 4). Compared with PCR and culture methods, respectively, the specificity of the AF Genital System® in detecting M. hominis was reasonably good at 82.9% and 86.5% (Table 1 and 2) while the specificity of the AF Genital System® in detecting U. urealyticum was 82.3% and 84.8% (Table 3 and 4). Furthermore, compared with PCR and culture methods, respectively, the positive predictive value (PPV) of the AF Genital System® for detection of M. hominis was 22.3% and 44.4% (Table 1 and 2) and the PPV of the AF Genital System® for detection of U. urealyticum was 26.3% and 36.8% (Table 3 and 4). Compared with PCR and culture methods, respectively, the negative predictive value (NPV) of the AF Genital System® for detection of M. hominis was 97.1% and 91.4% (Table 1 and 2) and the NPV of the AF Genital System® for detection of U. urealyticum was 94.2% and 97.1% (Table 3 and 4). These findings contrast with the observations by Redelinghuys et al., who reported a PPV and NPV of 97.1% and 28.6%, respectively, for detection of genital mycoplasmas using a similar assay. 17
Although the sensitivity of the AF Genital System® in detecting M. hominis and U. urealyticum in vaginal swab specimens was below that of PCR (under 80%) (Table 1 and 3), specificity was reasonably good at 82% to 83% (Table 1 and 3). The high NPV of the AF Genital System® in detecting M. hominis and U. urealyticum indicates that the system is quite reliable in ruling out infection for patients whose specimens lack these bacteria. Thus, mycoplasma can be ruled out as a possible cause of genital infection in these patients with abnormal vaginal discharge. In developing countries like Indonesia, where limited data are available on mycoplasma-related genital infections in the female population, this method is recommended for routine diagnosis, especially when no other causes for vaginal discharge are identified. 6 Moreover, the AF Genital System® has the advantages of being safe, fast, and less expensive for routine microbiological investigations of mycoplasma compared with other methods; this is a particular advantage in developing countries like Indonesia. Although many other studies have examined similar diagnostic techniques, to the best of our knowledge this is the first report on detection of genital mycoplasmas (M. hominis and U. urealyticum) using the AF Genital System®.12–14,16,17 However, this observational study was performed using a limited number of clinical specimens, and the small sample size may have affected the statistical significance of the results. Therefore, further studies of a greater larger of samples from other populations should be conducted, including in patients (male and female) with infertility disorders. Such studies are necessary to establish the diagnostic value of the AF Genital System® versus other standard techniques.
Conclusion
For patients in whom no other causes for vaginal discharge can be identified, specimen examination using the AF Genital System® is recommended. Our findings support the use of the AF Genital System® for identification of mycoplasma-associated diseases in developing countries like Indonesia with scarce resources and limited facilities.
Footnotes
Author contributions
ET and LHM: Conceptualization, visualization, supervision, methodology, validation, and data curation. SJ: Formal analysis, data curation, original draft preparation, review, and editing. All authors contributed significantly to the study and have approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors thank the School of Medicine and Health Sciences, Atma Jaya Catholic University of Indonesia, Jakarta, Indonesia for providing financial support for this study (funding number: 07/PHK-PKPD-sekr/03-2013).
Data availability
All relevant data are present in the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.