Abstract
Objective
To evaluate the performance of an in vitro fertilization (IVF) laboratory using a new set of key performance indicators (KPIs) when the main treatment of IVF patients had been changed.
Methods
Patients who underwent fresh embryo transfer and the freeze-all strategy in August, September, and October 2017 were retrospectively studied to evaluate the performance of an IVF laboratory in September when implantation rate of fresh embryo transfer decreased. KPIs associated with blastocyst culture and the first frozen embryo transfer (FET) cycle in patients with the freeze-all strategy were compared over 3 months.
Results
Day 5 usable blastocyst and good quality blastocyst rates, and day 3 usable/good quality embryo rates were not different among the three periods. The implantation rate and KPIs associated with morphological changes in warmed blastocysts in the first FET cycle in patients with the freeze-all strategy were also not different among the periods.
Conclusions
KPIs associated with embryo quality, blastocyst culture, and the pregnancy outcome of the first FET cycle in patients with the freeze-all strategy suggested that performance was unaffected in our IVF laboratory in September. These KPIs might be useful for internal quality control analysis of IVF laboratories.
Keywords
Introduction
Providing evidence of an unaffected/affected performance of in vitro fertilization (IVF) laboratories with a complete set of performance indicators is essential for quality control (QC) analysis in these laboratories.1–5 In 2017, the Vienna consensus meeting presented 12 key performance indicators (KPIs) to overview some crucial steps and the overall efficiency of IVF laboratories. 1 This consensus meeting aimed to establish KPIs for use in assisted reproductive technology laboratories by monitoring fresh IVF cycles. However, the freeze-all strategy has been increasingly used in the most recent 10 years,6–11 especially since the use of antagonist protocols in China. The proportion of freeze-all cycles has dramatically increased in our center. Nearly half of our patients have had all embryos cryopreserved without fresh embryo transfer since 2017. Some IVF centers even canceled all fresh embryo transfers. Therefore, a new set of KPIs derived from freeze-all IVF cycles is required to evaluate the performance of IVF laboratories.
Some KPIs on cryopreservation were suggested by the Alpha consensus meeting in 2012. 12 However, scientists at this consensus meeting mainly focused on the cryosurvival of oocytes and embryos, possibly because cryopreservation methods were not optimal at that time. With the widespread use of the vitrification method, the cryosurvival rate has significantly increased. Therefore, new KPIs are required to further evaluate the quality and developmental potential of frozen–thawed embryos. The Vienna consensus meeting recommended that IVF laboratory indicators and KPIs should be further extended and/or revised in the future. 1
Blastocyst culture and single blastocyst transfer have also been increasingly used in recent years to minimize multiple pregnancy rates while maintaining a high pregnancy rate. Most of our patients have their cleavage stage embryos cultured to blastocyst stage and cryopreserved on day 5/day 6, except for some poor responders. Therefore, KPIs should focus more on the efficiency of blastocyst culture and frozen–thawed blastocyst transfer. Hammond and Morbeck recently reported that the day 5 usable blastocyst rate was a more sensible KPI to detect adverse clinical outcomes than the day 5 blastocyst development rate. 13
Some unique sets of KPIs have been reported for QC analysis of IVF laboratories and IVF devices. Franco et al. designed a KPI score system to establish benchmarks for internal QC. 14 A previous study used KPIs in a special group of patients to evaluate the performance of incubators. 15 In the present study, KPIs that are commonly used in the routine practice of IVF laboratories in China (although the Vienna consensus meeting did not include them) and KPIs especially associated with blastocyst culture and freeze-all cycles in patients who were treated in September 2017 were compared with those in patients who were treated in August and October in 2017. We aimed to combine these KPIs to evaluate the performance of our IVF laboratory for internal QC analysis in September 2017, when the implantation rate of fresh transfer cycles suddenly decreased.
Material and methods
Study population
Patients who had undergone fresh transfer and the freeze-all strategy in August, September, and October 2017 at the IVF Center of the Third Affiliated Hospital of Sun Yat-sen University were retrospectively studied. All patients’ details had been de-identified. KPIs associated with the pregnancy outcome of the first frozen embryo transfer (FET) cycle were retrospectively collected (within 6 months) (Table 1). IVF and ICSI cycles were included. The reporting of this study conforms to the STROBE guidelines. 16 This study was approved by the Institutional Review Board of the Third Affiliated Hospital of Sun Yat-sen University (approval number: [2019] 02-515-01). Written or verbal informed consent of patients was not required because this was a retrospective study on routine treatment of patients with IVF.
Evaluation of an in vitro fertilization laboratory using performance indicators that were not recommended by a consensus meeting1

FET, frozen embryo transfer.
Freeze-all cycle and IVF procedures
We routinely transferred embryos on day 3 or 5, and excess day 3 embryos were further cultured to the blastocyst stage and cryopreserved. Patients with the freeze-all strategy underwent IVF, and all embryos were cryopreserved in accordance with standard protocols.17–21
Ovarian stimulation was initiated on day 2 or 3 of the menstrual cycle with either recombinant follicle-stimulating hormone or highly purified human menopausal gonadotropin. The starting dose was selected according to the women’s baseline characteristics, including age, body mass index (BMI), and antral follicle count. Cycles were monitored by serial vaginal ultrasound scans and serum estradiol and progesterone measurements. Additionally, whenever necessary, dose adjustments of recombinant follicle-stimulating hormone/human menopausal gonadotropin were performed according to the ovarian response. Pituitary downregulation was performed with daily administration of either cetrorelix or ganirelix starting from days 5 to 7 of the menstrual cycle. As soon as three follicles with a mean diameter of ≥17 mm were observed, final oocyte maturation and ovulation were triggered using urine purified human chorionic gonadotropin or recombinant human chorionic gonadotropin. Cumulus–oocyte complexes were collected by transvaginal aspiration 36 hours after triggering.
Insemination of the collected oocytes was performed via either conventional IVF or ICSI. Indications for insemination by ICSI included <1% sperm with a normal morphology, isolated sperm with <85% motility, the presence of anti-sperm antibodies, and/or procedures using cryopreserved or surgically retrieved sperm. Fertilization was assessed approximately 18 hours after insemination. Embryo quality and development were assessed on days 3, 5, and 6.
Day 3 cleavage embryos were assigned quality grades as described previously.16–18 Briefly, this scoring system relies on morphological assessment of the cell number, fragmentation, and blastomere equality. Except for the day 2 four-cell rate, the day 3 eight-cell rate, day 3 usable embryo rate, and day 3 good embryo rate were analyzed. The day 3 usable embryo rate included embryos transferred/cryopreserved and further cultured to blastocyst staged embryos. Day 3 good embryos were defined as embryos with ≥six cells and they had even or slightly uneven blastomeres with <25% fragments (Table 1). For fresh embryo transfer, the best quality day 3 cleavage stage embryos (<3) were selected for fresh transfer, and supernumerary embryos were further cultured and cryopreserved on day 5/day 6, or day 3 embryos were cultured to the blastocyst stage, and the highest scored blastocyst was transferred on day 5 (Table 2). Cleavage embryos were labeled and vitrified in sequence for patients with the freeze-all strategy without blastocyst culture according to their morphological score. The best quality embryos were warmed first and transferred back to patients first. For patients with freeze-all cycles with an extended culture, some had two embryos with a medium score cryopreserved on day 3 to guarantee that they had embryos available for FET. Other patients chose to have all of their embryos further cultured to blastocysts and cryopreserved. If blastocysts were cryopreserved, the blastocyst with the best score was transferred first at the FET cycle. At our institution, every embryo was scored by two of five embryologists using live video images according to the Alpha Scientists Istanbul Consensus for day 3 embryos and to Gardner et al. for blastocysts.17,19,22,23
Baseline characteristics, performance indicators, key performance indicators, and additional indicators in patients with fresh embryo transfer

Data are mean ± standard deviation, number, or percentage.
*P<0.05 compared with September.
BMI, body mass index; AMH, anti-Mullerian hormone; FSH, follicle-stimulating hormone; IU, international units; LH, luteinizing hormone; hCG, human chorionic gonadotropin; P, progesterone; Gn, gonadotropin; 2PN, two pronuclei; IR, implantation rate.
Day 3 usable embryo rate: percentage of embryos that were transferred, cryopreserved, and further cultured; day 3 good quality embryo rate: percentage of day 3 embryos with ≥six cells, with even or slightly uneven blastomeres with <25% fragments; day 5 usable blastocyst rate: percentage of transferred and vitrified blastocysts on day 5; day 5 good quality blastocyst rate: blastocysts with a score ≥4BB on day 5.
Indications for the freeze-all cycle were a high risk of ovarian hyperstimulation syndrome, late follicular progesterone levels >1.5 ng/mL, late follicular endometrium <7 mm, the patient’s preference, or other reasons 18 (Table 3).
Baseline characteristics, performance indicators, and key performance Indicators in patients with freeze-all cycles at three periods
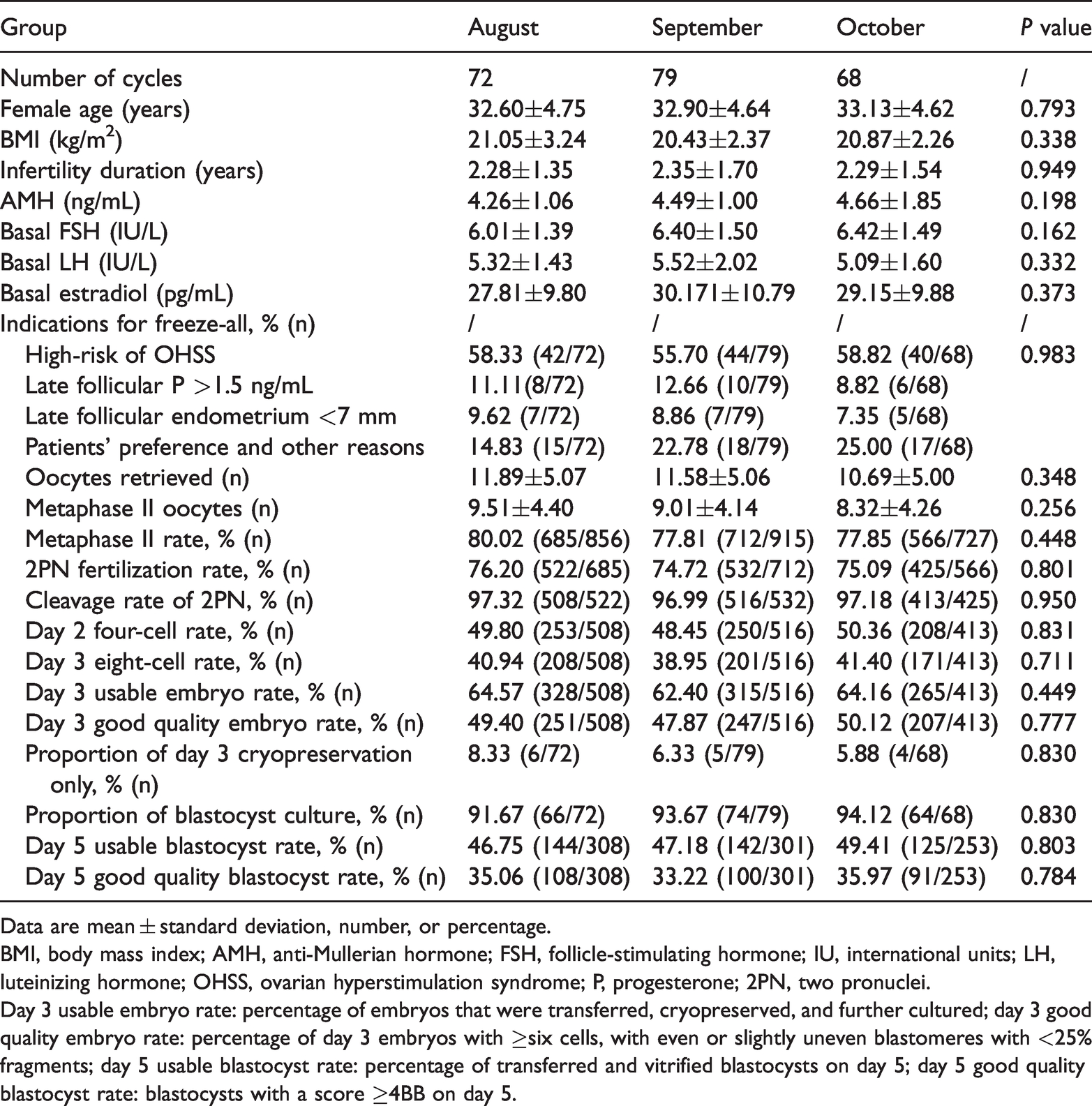
Data are mean ± standard deviation, number, or percentage.
BMI, body mass index; AMH, anti-Mullerian hormone; FSH, follicle-stimulating hormone; IU, international units; LH, luteinizing hormone; OHSS, ovarian hyperstimulation syndrome; P, progesterone; 2PN, two pronuclei.
Day 3 usable embryo rate: percentage of embryos that were transferred, cryopreserved, and further cultured; day 3 good quality embryo rate: percentage of day 3 embryos with ≥six cells, with even or slightly uneven blastomeres with <25% fragments; day 5 usable blastocyst rate: percentage of transferred and vitrified blastocysts on day 5; day 5 good quality blastocyst rate: blastocysts with a score ≥4BB on day 5.
Embryo vitrification and warming
Embryos were cryopreserved via vitrification using an open vitrification system with a Cryotop (Kitazato BioPharma Co., Ltd., Fuji, Shizuoka Japan). Vitrification and warming were carried out as previously described with minor adaptations. 17 The embryos were transferred from culture medium into equilibration solution and transferred into a new equilibration solution after being washed several times (Kitazato Corporation, Fuji, Shizuoka, Japan). The embryos spontaneously began shrinking, but they gradually returned to their original size, which suggested complete equilibration. This process took approximately 8 to 10 minutes. The embryos were immediately transferred to vitrification solution and placed on a Cryotop sheet with a minimal volume (<0.1 µL) of vitrification solution within 60 s. Excess volume was removed, and the Cryotop was quickly plunged into fresh liquid nitrogen. The warming procedure was performed by rapidly immersing the Cryotop sheet in 1 mL of warming solution 1 (Kitazato Corporation) for 1 minute to recover the embryos. The embryos were then transferred to warming solutions 2 and 3 and placed in a washing solution. Finally, embryos were transferred back to the transfer/blastocyst medium.
A blastocyst was considered vital after warming if it contained at least half of the intact cells grouped in a compact cell mass and if a blastocoel started to expand within 1 hour after in vitro culture.
Endometrial preparation for FET
The artificial endometrial preparation consisted of sequential administration of estradiol valerate and micronized vaginal progesterone as previously described.18,24,25 Briefly, 2 mg of estradiol valerate was administered at least twice daily for 10 to 14 days, and the dose was later adjusted according to the endometrial thickness measured by an ultrasound scan. If the endometrial thickness was ≥7 mm, vaginal progesterone supplementation was initiated. If the endometrial thickness was <7 mm, patients continued to take oral estradiol until the endometrium reached the necessary threshold, at which point progesterone supplementation was started (day −1). Progesterone was used for 2 weeks until a pregnancy test. If patients became pregnant, progesterone was continued until the 10th week.
Main outcome measures and statistical analysis
Descriptive statistics were used to summarize all continuous and categorical variables. Basic demographic characteristics were compared among women who had freeze-all cycles at the three periods and accepted their first FET in the following 6 months using analysis of variance (for continuous variables) or the χ2 test (for categorical variables). The implantation rate, which was defined as visualization of a heartbeat during a transvaginal ultrasound at 7 to 9 weeks’ gestational age, the cryosurvival rate, re-expansion, and the hatching rate of blastocysts 3 hours after warming were the main outcomes of our study (Table 1). A P value was considered statistically significant at <0.05. For all statistical analysis, we used IBM SPSS software version 19.0 (IBM Corp. Armonk, NY, USA).
Results
There were 442 ovum pick-up cycles and 214 fresh embryo transfer cycles from August to October 2017. The baseline characteristics of all patients with fresh transfer were not significantly different among the three periods (Table 2). The implantation rate of day 3 cleavage stage embryos in September was significantly lower than that in the other 2 months (P=0.037). The implantation rate of blastocysts in September was significantly lower than that in the other 2 months (P=0.031). However, the fertilization rate, cleavage rate, and days 3 and 5 usable embryo rates were not significantly different between the three periods (Table 2). These embryonic KPIs showed a stable performance in our IVF laboratory. Moreover, more than half of our patients had no fresh embryo transfer.
A total of 228 patients had all of their embryos cryopreserved without fresh embryo transfer (freeze-all cycles). We recorded the pregnancy outcomes of 219 first FET cycles in the following 6 months (Table 3). The patients’ characteristics, routine performance indicators, and KPIs of these freeze-all cycles, and the pregnancy outcomes of the patients’ first FET cycle are shown in Tables 3 and 4. There were 79 freeze-all cycles in September 2017. The fertilization and cleavage rates in these patients were not different among the three periods. There were no significant differences in the day 3 usable embryo rate, day 3 good quality embryo rate, day 2 four-cell rate, and day 3 eight-cell rate among the three periods in patients with freeze-all cycles. The day 5 usable blastocyst and good quality blastocyst rates were not significantly different among the three periods (Table 3). The implantation rate in the first FET cycles in patients with the freeze-all strategy in September was as high as that in the other 2 months (Table 4). The blastocyst cryosurvival rate was 100% in all three periods. The blastocyst re-expansion rate and blastocyst hatching rate were not different among the three periods after warming (Table 4).
Comparison of the key performance indicators associated with the first frozen embryo transfer cycle in patients with the freeze-all strategy in 3 consecutive months

Data are mean ± standard deviation, number, or percentage.
IR, implantation rate.
aLess than 4BB blastocysts were transferred, including 4BC, 4CB, and 3BB.
Discussion
Theoretically, KPIs of blastocyst culture are more sensitive indicators than those of cleavage stage embryos because they reflect a more extended development of embryos in vitro. However, the Vienna consensus meeting recommended only one blastocyst KPI (day 5 blastocyst development rate). 1 Moreover, this KPI does not reflect the precise quality of cultured blastocysts because all staged blastocysts are included, even those not qualified for transfer or cryopreservation. Hammond and Morbeck proposed the day 5 usable blastocyst rate for QC analysis, which included only transferred and cryopreserved blastocysts. 13 The day 5 usable blastocyst rate appears to be more useful than the day 5 blastocyst development rate. However, the day 5 usable blastocyst rate also shows a wide variety of blastocysts (score ≥3BB on day 5). In the present study, the day 5 good quality blastocyst rate was used to further evaluate the efficiency of blastocyst culture (score ≥4BB on day 5) because these blastocysts had a high developmental potential. In the present study, the day 5 blastocyst development rate, day 5 usable blastocyst rate, and day 5 good quality blastocyst rate were used to indicate the efficiency of blastocyst culture in September 2017. Using these performance indicators to indicate the efficiency of blastocyst culture and the performance of IVF laboratories might be more accurate than using the day 5 blastocyst development rate alone. These three rates were not different between the 3 consecutive months in this study, which indicated a normal performance of our IVF lab in September 2017.
For cleavage stage embryo development, the day 3 usable embryo and good quality embryo rates were included in the present study to indicate development of the cleavage stage embryo, which is widely used in IVF laboratories in China.17,18 The day 3 good quality embryo rate includes embryos with ≥six cells, with even or slightly uneven blastomeres with <25% fragments in our center. Although the consensus meeting did not recommend the day 3 good quality embryo rate, it is the most commonly used indicator at our center and other IVF centers in China. The day 3 good quality embryo rate combines information on the cell number, the cell size, and fragments of day 3 cleavage stage embryos. Therefore, this rate includes more details than the day 2 four-cell rate and the day 3 eight-cell rate alone. Moreover, because embryos are cleaved at different speeds in different culture mediums (even different brands and different lots), the day 3 eight-cell rate alone does not fully represent good quality embryos on day 3. The day 3 usable embryo and good quality embryo rates (two KPIs) combined with the day 2 four-cell embryo rate and day 3 eight-cell embryo rate might be more comprehensive for showing development of the cleavage embryo than the day 2 four-cell embryo rate and the day 3 eight-cell embryo rate alone in vitro. In the present study, all of the four KPIs were not different over the 3 months studied, which indicated a normal performance of the IVF laboratory at the cleavage stage period in September 2017.
The combined use of blastocyst culture and the freeze-all strategy greatly increased vitrified/warmed blastocyst transfer in our center. In contrast to frozen–thawed cleavage stage embryo transfer, warmed blastocysts show visible morphological changes during a short time of in vitro culture before transfer. Warmed blastocysts gradually re-expand and started to hatch in 2 to 4 hours after warming (especially for blastocysts being collapsed by a laser before vitrification). These dynamic morphological changes are associated with the implantation potential of cryopreserved blastocysts.26–28 Therefore, the re-expansion rate and the hatching rate could reflect the quality of vitrified/warmed blastocysts of the first FET cycle in patients with the freeze-all strategy, without influence by the receptivity of the uterus. In the present study, the re-expansion rate and hatching rate of the first vitrified/warmed blastocyst transfer in patients with the freeze-all strategy were not significantly different among the three periods. This finding indicated the high developmental potential of cultured/vitrified blastocysts and a normal performance of the IVF laboratory in September 2017. Incubating warmed blastocysts in a time-lapse incubator for a while could lead to more objective KPIs for vitrified/warmed blastocysts. 27
Embryos with the highest score are traditionally freshly transferred and represent the quality of the whole cohort of embryos in patients who have IVF. With an increased proportion of freeze-all cycles, the first fresh embryo transfer cycle is gradually changed to the first FET cycle. Therefore, analyzing the pregnancy outcome of vitrified/warmed embryos as quickly as possible is vital to confirm the developmental potential of cultured embryos and thus the performance of IVF laboratories. Moreover, the endometrium of FET cycles is more “natural” and possibly has a higher receptivity without the influence of high hormone levels during fresh embryo transfer cycles.29–35 Therefore, the implantation rate of the first FET cycle might be a better indicator for reflecting the quality of cultured embryos and evaluating the performance of IVF laboratories than that of fresh embryo transfer cycles, especially for antagonist protocols. However, tracking the pregnancy outcomes of these embryos is time-consuming because they are warmed and transferred at different times. In the present study, the first FET cycle in patients with the freeze-all strategy was tracked within 6 months after oocyte retrieval. The implantation rate was not different over the 3 months in these patients. This finding further supports the quality of in vitro cultured embryos and a sufficient performance of our IVF laboratory in September 2017. However, this was an observational study, and not all cryopreserved embryos were transferred. Therefore, the performance of our IVF laboratory in September 2017 needs to be further evaluated.
In conclusion, the implantation rate of fresh embryo transfer was decreased in September 2017. However, other KPIs associated with embryo quality and blastocyst culture, and the pregnancy outcome of the first FET cycle in patients with the freeze-all strategy showed that the performance of our IVF laboratory was unaffected at this time. These KPIs might be useful for internal QC analysis of IVF laboratories because blastocyst culture and the freeze-all strategy are increasingly being used in IVF laboratories worldwide.
Footnotes
Acknowledgements
We thank all embryologists and the clinical staff in our IVF center for their assistance in IVF laboratory work and clinical work that made this study possible.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No: 82060176).
Author contributions
All authors were all involved in routine IVF work. HT Wang and PP Hong extracted data from the database. HT Wang and W Zhou analyzed the data. T Li wrote the manuscript with the assistance of HT Wang and PP Hong. All authors read and approved the final manuscript.
Availability of data and materials
Data are available only on request.
