Abstract
Objective
To analyze serum levels of inhibitory costimulatory molecules and their correlations with innate immune cytokine levels in patients with pulmonary tuberculosis (PTB).
Methods
Data for 280 PTB patients and 280 healthy individuals were collected. Serum levels of immune molecules were measured using ELISA. Univariate, multivariate, subgroup, matrix correlation, and receiver operating characteristic curve analyses were performed.
Results
Host, environment, lifestyle, clinical features, and medical history all influenced PTB. Serum levels of soluble programmed death ligand 1 (sPD-L1), soluble T-cell immunoglobulin- and mucin-domain–containing molecule 3 (sTim-3), soluble galectin-9 (sGal-9), interleukin (IL)-4, and IL-33 were significantly higher in patients with PTB, while levels of IL-12, IL-23, IL-18, and interferon (IFN)-γ were significantly lower. Serum levels of sTim-3 were higher in alcohol users. Levels of sTim-3 were negatively correlated with those of IL-12. Levels of IL-12, IL-23, and IL-18 were positively correlated with those of IFN-γ, while levels of IL-12 were negatively correlated with those of IL-4. The areas under the curve of sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 for identifying PTB were all >0.77.
Conclusions
Inhibitory costimulatory molecules may be targets for controlling PTB. Immune molecules may be helpful for diagnosis of PTB.
Keywords
Introduction
Millions of people die from tuberculosis every year, and pulmonary tuberculosis (PTB) is a top 10 cause of death worldwide. 1 When Mycobacterium tuberculosis (MTB) enters the body, it first elicits an innate immune response. Cytokines produced by innate immune cells can trigger differentiation and proliferation of T cells and regulate their function. 2 The relationships between secreted cytokines are complex. These molecules interact with one another in cytokine networks, affecting the development and outcome of PTB.
Some of the common inhibitory receptors are programmed cell death protein 1 (PD-1), T-cell immunoglobulin- and mucin-domain-containing molecule 3 (Tim-3), lymphocyte activation gene-3, and cytotoxic T lymphocyte-associated antigen 4. 3 Expression of PD-1 and its ligands, PD-L1 and PD-L2, is increased in patients with active pulmonary tuberculosis. Blocking the PD-1/PD-L interaction can improve protective immunity mediated by MTB-specific CD4+ T cells as well as the phagocytic function of macrophages. 4 , 5 It has been reported that soluble PD-1 (sPD-1) can compete with membrane PD-1 (mPD-1) and block negative regulatory signals mediated by the mPD-1/membrane PD-L1 (mPD-L1) interaction. sPD-L1 is derived from mPD-L1 by enzymatic cleavage, so soluble PD-L1 (sPD-L1) and mPD-L1 have the same immune function. 6 , 7
Research on infectious diseases 8 has shown that soluble Tim-3 (sTim-3) levels are increased in patients with acquired immune deficiency syndrome (AIDS) and exert immunosuppressive effects mainly through inhibition of T cells responses and production of related cytokines. sTim-3 may also be associated with immune imbalances and disease progression in patients with AIDS. However, the precise roles of inhibitory costimulatory molecules in the pathogenesis of tuberculosis are still unclear. In addition, galectin-9 (Gal-9) is associated with severity of tuberculosis, and higher plasma Gal-9 levels are found in patients with active tuberculosis. 9 In MTB infection, the Tim-3/Gal-9 pathway plays an important role in antibacterial immunity by regulating innate immune inflammatory networks. 10
In recent years, studies on the roles of inhibitory costimulatory molecules and their ligands in PTB have mainly focused on regulation of effector T cell function after pathogen infection. 11 , 12 Nevertheless, both innate and adaptive immunity play a vital role in clearing MTB. 13 Interferon (IFN)-γ secreted by Th1 cells plays an important role in the immune response against tuberculosis. 14 Development of CD4+ T cells, which produce IFN-γ, depends on IL-12 and IL-23. Hence, IL-12 and IL-23 are required for IFN-γ– dependent immunity against MTB, both alone and in combination. 15 IL-12, IL-23, and IL-18 are secreted by innate immune cells, and IL-12 performs important functions in the immune response to MTB infection. 16 IL-23 can promote immunity against MTB. 17 IL-18 also plays an important role in immune responses against tuberculosis infection. 18
IL-4 can inhibit T-cell responses against tuberculosis, resulting in decreased bactericidal and bacteriolytic activity of macrophages. 19 IL-33, which activates and promotes Th2 cells to release inflammatory mediators such as IL-4 and IL-5, 16 has potential therapeutic effects on established MTB infections and may represent a novel therapy for PTB. 20
Most studies of PTB have focused on membrane-bound PD-1/PD-L1 and Tim-3/Gal-9, while the roles of sPD-1, sPD-L1, sTim-3, and soluble galectin-9 (sGal-9) in the pathogenesis of PTB remain unclear. The relationships between sPD-1/sPD-L1, sTim-3/sGal-9, and innate immune-related cytokines are also unclear.
The development of tuberculosis after MTB infection also depends on nonbiological factors such as sex, age, socioeconomic status, 21 living environment, 22 and lifestyle factors such as smoking 23 and drinking alcohol. 24 In view of this, we analyzed the factors influencing PTB and levels of immune molecules in this study. We also explored correlations between levels of costimulatory molecules and innate immune-related cytokines in PTB patients. Our overall goal was to determine whether immune molecules may be effective biomarkers of PTB.
Materials and methods
Selection of patients
This was a retrospective case–control study. Based on the diagnostic criteria for tuberculosis (ws288-2017) of the health industry standard of the People’s Republic of China, 25 patients with active PTB were randomly selected from the First and Fifth Affiliated Hospitals of Xinjiang Medical University.
The inclusion criteria were as follows: (1) positive MTB in sputum; (2) clinical symptoms of PTB (cough, expectoration, chest tightness, and other symptoms); (3) X-ray or computed tomography imaging consistent with PTB (multiple morphological manifestations such as exudation, proliferation, fibrous and caseous lesions, and calcification and cavity formation); (4) sputum smear positive for acid-fast staining; (5) positive tuberculin test; (6) positive detection of T cell spots by T-SPOT.TB test; (7) positive serum anti-tuberculosis antibody; (8) effective anti-tuberculosis treatment; (9) elevated erythrocyte sedimentation rate; and (10) exclusion of other non-TB diseases. If the first criterion was met, a diagnosis of PTB was made; if the first criterion was not confirmed, diagnosis required three or more of the remaining inclusion criteria. The exclusion criteria were as follows: (1) immune diseases; (2) heart, liver, or kidney disease; or (3) other serious diseases.
Physical examinees were selected from the First and Fourth Affiliated Hospitals of Xinjiang Medical University as a control group. The two groups were matched in terms of age and sex.
This study was approved by the Ethics Committee of the Fourth Affiliated Hospital of Xinjiang Medical University (ethics audit number: 2020XE-YS0056). As a retrospective study, the requirement for informed consent was waived. We de-identified all participants’ details. The reporting of this study conformed to the STROBE statement. 26
Questionnaire
A unified questionnaire was used to collect demographic data and information on living and workplaces, diet, lifestyle, and medical history.
Sample collection
Before anti-tuberculosis treatment, fasting venous blood was collected in the early morning from all participants into red vacuum negative pressure tubes (without any anticoagulant). Within 2 hours, the samples were centrifuged at 1500 ×g for 10 minutes. The upper serum layer was obtained, aliquoted in microcentrifuge tubes, and stored at −80°C.
Enzyme-linked immunosorbent assay (ELISA)
ELISA kits (Dakewe Biotech Co., Ltd. Shenzhen, China) were used to assess levels of sPD-1, sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 in serum following the manufacturer’s instructions. Frozen serum aliquots were placed at room temperature for 20 minutes, and then 50 µL each of diluted standard, serum sample, and biotin-labeled antibody were added into each well.
After covering the plate, shaking, and mixing, the samples were incubated at 37°C for 1 hour. Each well was washed three times with detergent. Horseradish peroxidase-labeled streptavidin (80 µL) was added to each well. After shaking and mixing, the serum samples were incubated at 37°C for 30 minutes. After washing each well three times with detergent, 50 µL each of substrates A and B were added into each well. After shaking and mixing, the samples were incubated at 37°C for 10 minutes in the dark. Finally, 50 µL of stop solution was added.
Absorbance values were measured using a plate reader (Multiskan FC, Thermo Fisher, Waltham, MA, USA). The wavelength was 450 nm, and a blank well was adjusted to zero. The optical density values of the standard and samples were corrected by subtraction of the blank well absorbance value.
A standard curve was constructed using MS Excel. According to the standard curve equation, the absorbance values of samples were used to calculate concentrations of sPD-1, sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 in serum.
The detection ranges of each analyte were as follows: sPD-1, 15.18–48.61 ng/mL; sPD-L1, 1.16–6.36 ng/mL; sTim-3, 3.23–19.86 ng/mL; sGal-9, 350.25–1125.69 pg/mL; IL-12, 10.19–28.36 pg/mL; IL-23, 331.25–508.98 pg/mL; IL-18, 15.56–38.12 pg/mL; IFN-γ, 2.30–5.35 pg/mL; IL-4, 1.21–3.98 pg/mL; and IL-33, 219.43–464.25 pg/mL.
Statistical analysis
Statistical analysis was performed in SPSS version 13.0 (SPSS Inc., Chicago, IL, USA). Figures were prepared using GraphPad Prism (GraphPad Software, San Diego, CA, USA). Normality of distributions was analyzed using the Shapiro–Wilk test. Data were expressed as means ± standard deviations (SDs). Independent-sample t tests were used to assess differences between two groups. Differences between multiple groups were assessed using analysis of variance. Categorical data were expressed as n (%) and differences between groups were assessed using the chi-square test. Matrix correlation analysis (Pearson correlation analysis) was used to examine relationships among immune molecules. Values of P < 0.05 were considered statistically significant. Receiver operating characteristic (ROC) curve analysis was performed to assess the diagnostic value of immune molecules for identifying patients with PTB.
Results
Baseline characteristics of study participants
The average (SD) age of the 280 PTB patients enrolled in this study was 55.60 ± 15.93 years. Among these patients, 73 had mild PTB (no hospitalization/steroids/procedures), 118 had moderate PTB (invasive procedures/corticosteroid use), and 89 had advanced PTB (hospitalization/death). Fifty-two patients had newly diagnosed PTB, while 228 were undergoing retreatment for PTB. One hundred and six patients were sputum smear-positive, while 174 were sputum smear-negative. One hundred and seventeen patients had PTB cavity, while 163 showed no signs of PTB cavity.
A total of 280 physical examinee were selected as a control group. Their mean (SD) age was 53.39 ± 14.77 years. There were no significant differences in age and sex between the PTB and healthy control groups (Table 1). However, there were significant differences in education, occupation, and marital status between the PTB group and the healthy control group (P < 0.05). In the PTB group, the proportions of individuals with education less than senior high school, farmers, and married individuals were higher. There were no significant differences in registered residence and monthly income between PTB patients and healthy controls.
Baseline characteristics of study participants.

All data represent n (%).
PTB, pulmonary tuberculosis; HC, healthy control.
Comparison of diet and lifestyle between patients with PTB and healthy controls
There were significant differences in consumption of fresh fruit, drinking unboiled water, smoking, alcohol consumption, and exercise between patients with PTB and healthy controls (P < 0.05) (Table 2). The proportions of individuals who often or occasionally drank unboiled water was higher among patients with PTB compared with healthy controls. The proportions of patients with PTB who consumed fresh fruit two to three times per week, smoked, drank alcohol, and exercised rarely were higher than those of healthy controls.
Diet and lifestyle factors in patients with PTB and healthy controls.
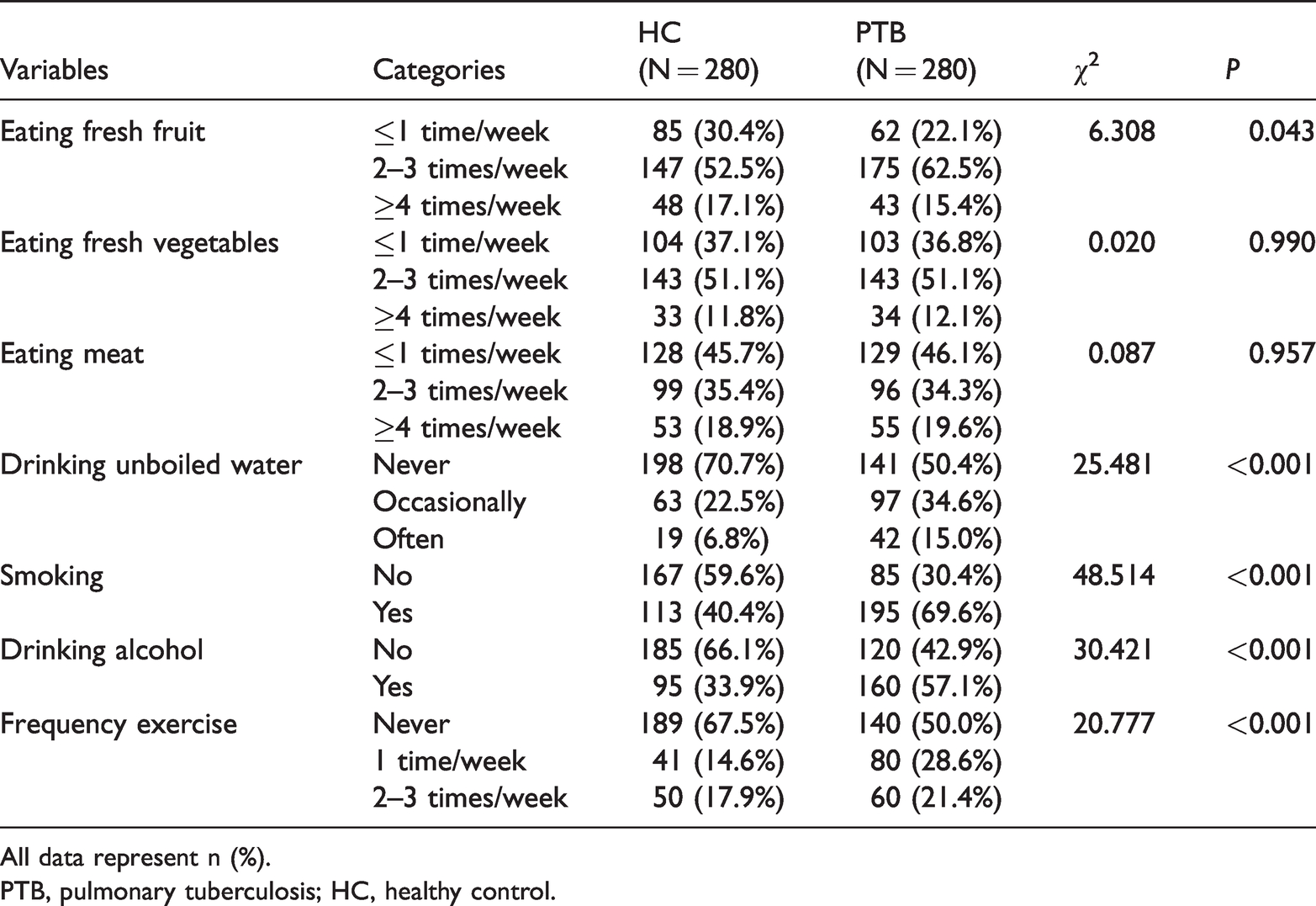
All data represent n (%).
PTB, pulmonary tuberculosis; HC, healthy control.
Comparison of living and workplace conditions between patients with PTB and healthy controls
There were significant differences in workplace dust, indoor pollution, and exposure to environmental microbes between patients with PTB and healthy controls (P < 0.05) (Table 3). The proportions of patients with PTB exposed to a medium level of workplace dust, indoor pollution, and environmental microbes were higher than those of healthy controls.
Conditions of living and workplaces of patients with PTB and healthy controls.

All data represent n (%).
PTB, pulmonary tuberculosis; HC, healthy control.
Comparison of clinical conditions and medical history between patients with PTB and healthy controls
The proportions of patients with PTB with lung cancer and diabetes as well as the proportion of patients having contact with other individuals with PTB were higher than those of healthy controls (P < 0.05). The proportion of patients with PTB who had received the Bacillus Calmette–Guérin vaccine was lower than that of healthy controls (P < 0.05). The proportion of patients with PTB who had received prior immunosuppressive therapy was higher than that of healthy controls (P < 0.05).
Identification of factors influencing PTB
Any factors mentioned above that were statistically significant were included in the multivariate analysis. Binary logistic regression was used to identify factors significantly associated with PTB (Table 5). Smoking, alcohol consumption, indoor pollution, exposure to environmental microbes, and diabetes were risk factors for PTB. Individuals who ate fresh fruit two to three times per week, drank unboiled water occasionally, and had contacts with PTB patients were at lower risk of PTB.
Clinical condition and medical history of patients with PTB and healthy controls

*Fisher’s exact test.
All data represent n (%).
PTB, pulmonary tuberculosis; HC, healthy control; HIV, human immunodeficiency virus; BCG, Bacillus Calmette–Guérin.
Identification of factors influencing PTB using binary logistic regression analysis
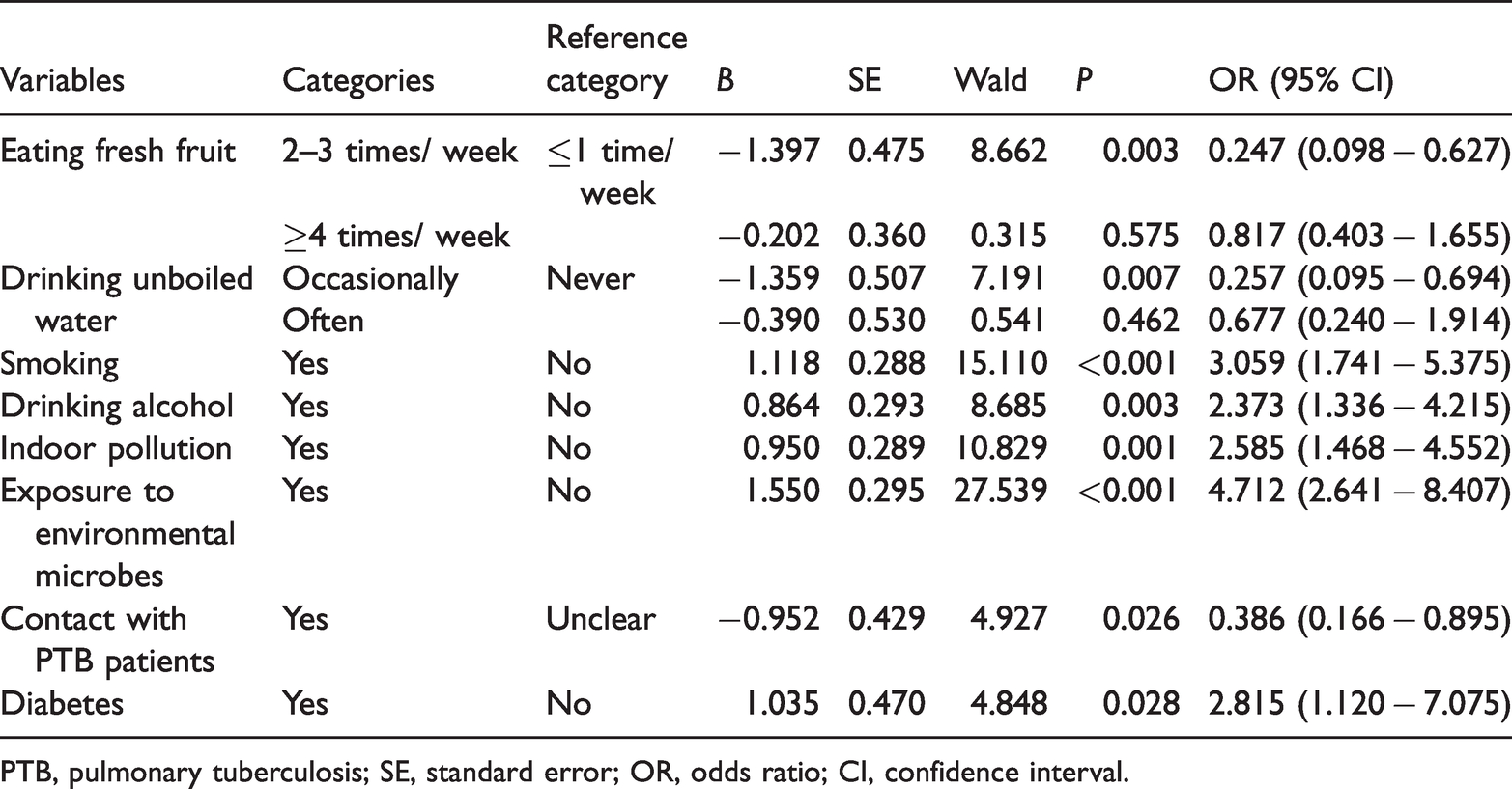
PTB, pulmonary tuberculosis; SE, standard error; OR, odds ratio; CI, confidence interval.
Serum levels of sPD-1, sPD-L1, sTim-3, and sGal-9
To explore whether levels of inhibitory costimulatory molecules and their ligands were related to PTB, levels of sPD-1, sPD-L1, sTim-3, and sGal-9 in the sera of patients with PTB and healthy controls were assessed (Figure 1). Compared with healthy controls, serum levels of sPD-L1, sTim-3, and sGal-9 were significantly higher in patients with PTB (P < 0.001). In contrast, there were no significant differences in serum levels of sPD-1 between patients with PTB and healthy controls. These data suggested that increased levels of sPD-L1, sTim-3, and sGal-9 may play important roles in the occurrence and development of PTB.

Serum levels of sPD-1, sPD-L1, sTim-3, and sGal-9 in patients with PTB and healthy controls measured using ELISA. Levels of sPD-L1 (b), sTim-3 (c), and sGal-9 (d) in patients with PTB were higher than those in healthy controls (P < 0.001). There was no significant difference in levels of sPD-1 between patients with PTB and healthy controls. Horizontal bars represent means ± SDs. Statistical analyses were performed using the independent-sample t test.
Serum levels of innate immune- and T-cell-related cytokines
Innate immune cells can affect the immune microenvironment through the production of cytokines, which can regulate immune activity. As shown in Figure 2a–c, serum levels of innate immune

Serum levels of IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 in patients with PTB and healthy controls. ELISA was used to measure serum levels of IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33. Compared with healthy controls, levels of IL-12, IL-23, IL-18, and IFN-γ were significantly decreased (P < 0.001), and levels of IL-4 and IL-33 were significantly increased in patients with PTB (P < 0.001). Horizontal bars represent means ± SDs. Statistical analyses were performed using the independent-sample t test.
Serum levels of immune molecules in patients with mild, moderate, and advanced PTB compared with healthy controls
To further assess the roles of sPD-1, sPD-L1, sTim-3, sGal-9, and innate immune- and T-cell-related cytokines in PTB, levels in multiple groups were compared using analysis of variance. We found significant differences in sPD-1, sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 among patients with mild, moderate, and advanced PTB compared with healthy controls (Table 6). Further intergroup comparisons showed that more severe PTB was associated with higher serum levels of some immune molecules (sPD-1, sPD-L1, sTim-3, sGal-9, IL-4, and IL-33) and lower serum levels of others (IL-12, IL-23, IL-18, and IFN-γ).
Serum levels of immune molecules in patients with advanced, moderate, and mild PTB and in healthy controls.
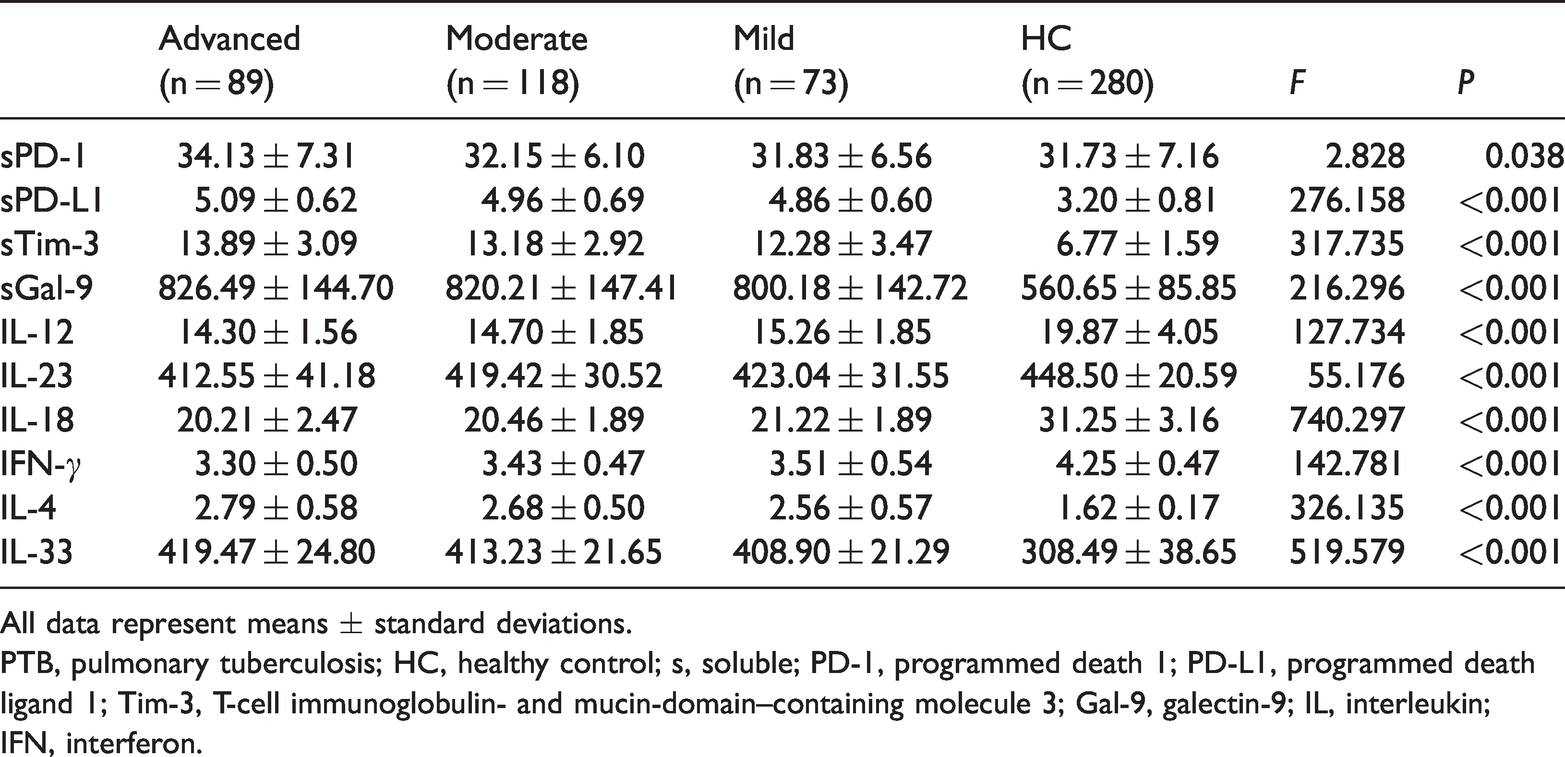
All data represent means ± standard deviations.
PTB, pulmonary tuberculosis; HC, healthy control; s, soluble; PD-1, programmed death 1; PD-L1, programmed death ligand 1; Tim-3, T-cell immunoglobulin- and mucin-domain–containing molecule 3; Gal-9, galectin-9; IL, interleukin; IFN, interferon.
We next stratified by serum levels of costimulatory molecules and innate immune-related cytokines levels and analyzed the distribution of PTB severity (Table 7). The proportion of patients with different stratified levels of sTim-3, IL-12, IL-23, IL-18, and IL-33 were significantly different among patients with mild, moderate, and advanced PTB. The proportions of patients with medium serum levels of sTim-3, IL-12, IL-23, IL-18, and IL-33 were the highest among patients with mild, moderate, and advanced PTB.
Stratification analysis of serum levels of costimulatory molecules and innate immune-related cytokines according to PTB severity.

All data represent n (%).
PTB, pulmonary tuberculosis; s, soluble; PD-1, programmed death 1; PD-L1, programmed death ligand 1; Tim-3, T-cell immunoglobulin- and mucin-domain–containing molecule 3; Gal-9, galectin-9; IL, interleukin; IFN, interferon.
Identification of factors influencing levels of immune molecules
To analyze whether demographic characteristics, diet, lifestyle, living and workplaces, clinical conditions, or medical history affected serum levels of immune molecules, multiple linear regression analysis was performed (Table 8). Demographic characteristics, living habits, environment, and medical history associated with serum levels of immune molecules in patients with PTB.
Identification of factors influencing levels of immune molecules using multiple linear regression analysis

PTB, pulmonary tuberculosis; SE, standard error; CI, confidence interval; s, soluble; PD-1, programmed death 1; PD-L1, programmed death ligand 1; Tim-3, T-cell immunoglobulin- and mucin-domain–containing molecule 3; Gal-9, galectin-9; IL, interleukin; IFN, interferon.
Matrix correlation analysis of serum levels of sPD-L1, sTim-3, sGal-9, and innate immune-related cytokines in patients with PTB
It has been reported that upregulation of Tim-3 expression in macrophages induced by pathogen infection may be involved in regulation of macrophage activation and cytokine secretion. 27 To investigate correlations between increased levels of inhibitory costimulatory molecules and innate immune-related cytokines (IL-12, IL-23, and IL-18) in patients with PTB, matrix correlation analysis was performed (Figure 3). Serum sTim-3 levels of all patients were negatively correlated with levels of IL-12 (r = −0.160, P = 0.007). Levels of serum IL-12 and IL-18 were positively correlated (r = 0.194, P = 0.001), as were levels of IL-23 and IL-18 (r = 0.553, P < 0.001). These results suggested that sTim-3 may affect serum levels of innate immune-related cytokines in patients with PTB.
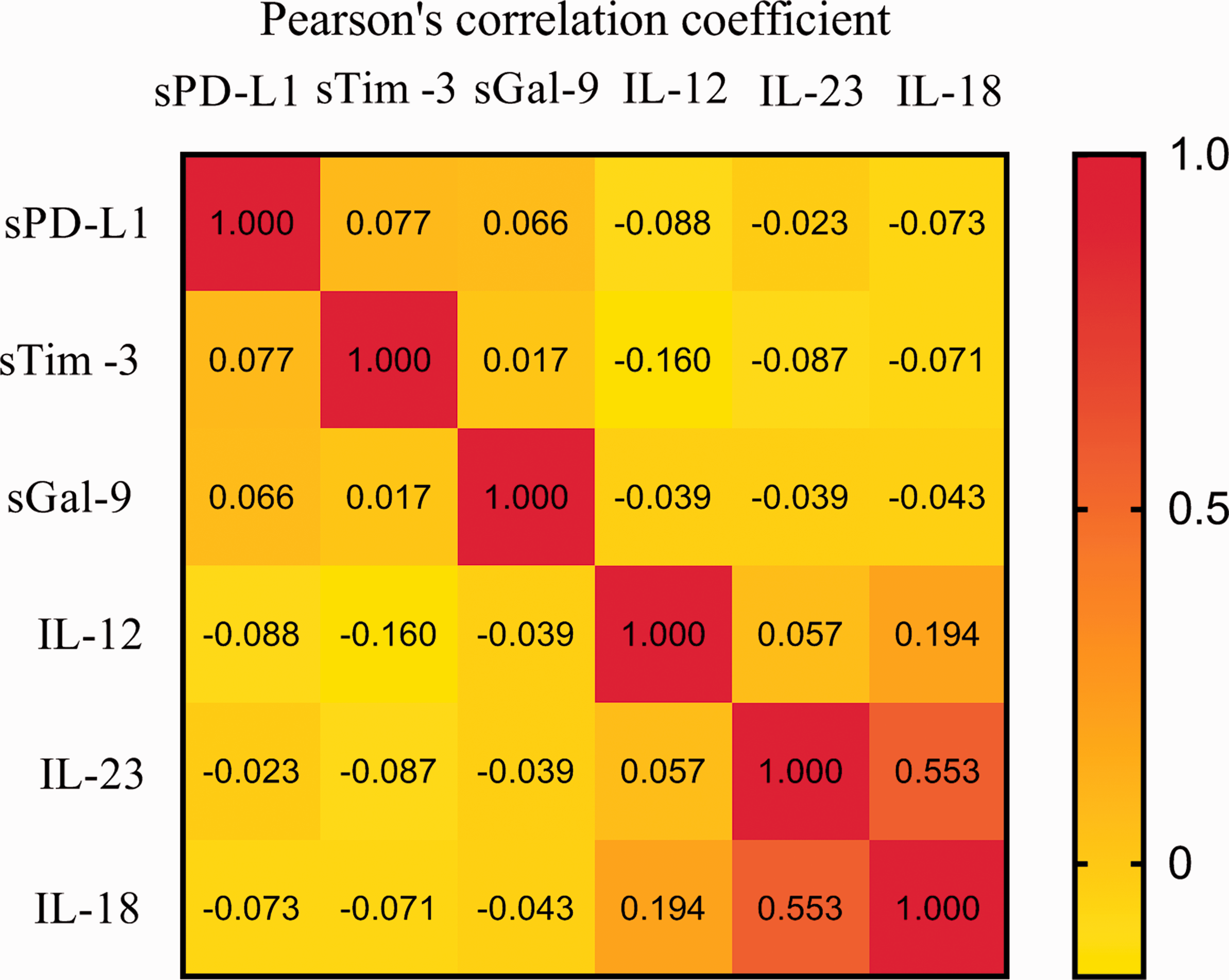
Among patients with PTB, correlations among serum levels of sPD-L1, sTim-3, sGal-9, and innate immune-related cytokines (IL-12, IL-23, and IL-18) were analyzed using the matrix correlation analysis (Pearson correlation analysis). The numbers in the squares represent correlation coefficients; the darker the color, the stronger the correlation.
Relationships between innate immune- and Th cell-related cytokines in patients with PTB
Innate immune cells can prime naïve T cells, stimulate Th cells, and promote T cell differentiation 28 to initiate immune responses. 29 We further investigated the relationships between innate immune- and Th cell-related cytokines in patients with PTB. As shown in Figure 4, serum levels of IL-12, IL-23, and IL-18 were all positively correlated with levels of IFN-γ (r = 0.215, P < 0.001; r = 0.226, P < 0.001; and r = 0.219, P < 0.001, respectively). Serum levels of IL-12 were inversely correlated with levels of IL-4 (r = −0.130, P = 0.029). Serum levels of IL-4 and IL-33 were positively correlated (r = 0.161, P = 0.007). Thus, when low levels of innate immune-related cytokines were present, levels of IFN-γ were also low while levels of IL-4 were increased, resulting in weakened anti-MTB immunity.

Among patients with PTB, correlations among serum levels of innate immune related-cytokines (IL-12, IL-23, and IL-18) and Th cell-related cytokines (IFN-γ, IL-4, and IL-33) were analyzed using matrix correlation analysis (Pearson correlation analysis). The numbers in the squares represent correlation coefficients; the darker the color, the stronger the correlation.
Diagnostic value of serum immune molecules for PTB
To further investigate the diagnostic value of serum levels of immune molecules for identifying PTB, ROC curve analysis was performed. The AUC values of sPD-L1 (Figure 5b), sTim-3 (Figure 5c), sGal-9 (Figure 5d), IL-12 (Figure 6a), IL-23 (Figure 6b), IL-18 (Figure 6c), IFN-γ (Figure 6d), IL-33 (Figure 6e), and IL-4 (Figure 6f) were all >0.77 (P < 0.0001). The AUC values of sPD-L1, sTim-3, sGal-9, and IL-18 were above 0.9.

ROC curve analysis of the diagnostic value of sPD-1 (a), sPD-L1 (b), sTim-3 (c), and sGal-9 (d) for identifying PTB.

ROC curve analysis of the diagnostic value of IL-12 (a), IL-23 (b), IL-18 (c), IFN-γ (d), IL-4 (e), and IL-33 (f) for identifying PTB.
Discussion
Patients infected with MTB eventually develop disease depending on variety of host, environment, and social factors. Our results showed that eating fresh fruit, drinking unboiled water, smoking, drinking alcohol, indoor pollution, exposure to environmental microbes, contact with PTB patients, and diabetes may be factors influencing the development of PTB (Table 5).
Studies have shown that eating fruit 30 and smoking 31 can impact tuberculosis. Fruit consumption may have immunoregulatory effects. Smoking may increase the incidence of tuberculosis by weakening immunity and enhancing nonspecific inflammatory responses. 32 Alcohol is also a risk factor for tuberculosis. 33 Drinking unboiled water can increase the risks of infectious diseases. 34 Workplace or home pollution and exposure to pathogenic microorganisms are risk factors for PTB. Higher levels of dust, sulfur dioxide, and other pollutants in the air may promote MTB aerosol formation and entry into the human respiratory system. For example, exposure to fine particles (PM2.5) may inhibit immune defense mechanisms and increase susceptibility to active tuberculosis and tuberculosis-related mortality. 35 Infection by pathogenic microorganisms may change sensitivity to MTB infection and affect growth of MTB in infected cells. 36
We found that compared with those who had no established contact with a PTB patient, those who ever had contact with PTB patients had a lower risk of PTB. However, the results of univariate analysis showed that the proportions of individuals in PTB patients were higher among patients with PTB (15.4%) compared with healthy controls (9.6%) (P < 0.05) (Table 4). One potential explanation for this discrepancy might be that individuals with such contacts knew that the risk of infection and occurrence of tuberculosis was high, so they paid more attention to prevention measures.
Diabetes and AIDS also increase susceptibility to tuberculosis. 37 , 38 The prevalence of PTB in diabetic patients with poor glycemic control was significantly higher than that in patients with good glycemic control. 39
Serum sPD-1 and sPD-L1 levels are closely related to the occurrence and development of disease. Some studies have confirmed the inhibitory effects of sPD-L1. 40 , 41 Our preliminary results showed that increased levels of sTim-3 may be associated with PTB and that sTim-3 may be involved in the pathogenesis of PTB by affecting cytokine secretion by CD4+ T-cell subsets. 42
The signals relayed by inhibitory costimulatory molecules have regulatory effects on both adaptive and innate immune cells. Cytokines produced by T-cell subsets play a role in immunity against tuberculosis, 43 while cytokines produced by activated innate immune cells may also play a role in progression of tuberculosis infection. We found that serum levels of sPD-L1, sTim-3, and sGal-9 were significantly higher in patients with PTB compared with healthy controls, while levels of IL-12, IL-23, and IL-18 levels were significantly lower.
Serum levels of sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 were significantly different among patients with mild, moderate, and advanced PTB compared with healthy controls. Levels of sPD-L1, sTim-3, and sGal-9 were lower in individuals with mild PTB, while levels of IL-12, IL-23, and IL-18 were higher. This finding suggested that increased levels of sPD-L1, sTim-3, and sGal-9 might participate in the pathogenesis of PTB (i.e., upon infection with MTB, innate immune cells might have restricted responsiveness).
Stratification analysis showed that strata of sTim-3, IL-12, IL-23, IL-18, and IL-33 differed significantly among patients with advanced, moderate, and mild PTB. The Cochran–Mantel–Haenszel test showed that different demographic characteristics (diet, lifestyle, living and workplaces, clinical conditions, and medical history) were associated with different serum levels of sPD-1, sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 in patients with PTB and healthy controls. Among individuals with nonlocal registered residence, exposure to a medium level of indoor ventilation, frequency of no prior immunosuppressive therapy, and serum levels of sPD-1 were significantly different between patients with PTB and healthy controls (data not shown).
Using multiple linear regression analysis, we found that demographic characteristics, living habits, environment, and medical history may affect serum levels of immune molecules in patients with PTB. Serum levels of sTim-3 among alcohol drinkers were particularly high. Drinking alcohol inhibits the immune system, particularly cellular immunity. 44 The chi-square test showed that the proportion of alcohol drinkers among patients with PTB (57.1%) was significantly higher than that among healthy controls (33.9%) (Table 2). These results suggested that demographic characteristics, living habits, environment, clinical condition, and medical history may affect the occurrence and development of PTB by altering the expression of immune molecules.
To further identify factors associated with decreased ability of innate immune cells to mediate anti-MTB immunity in PTB patients, we explored correlations of sPD-L1, sTim-3, and sGal-9 with innate immune-related cytokines. Matrix correlation analysis revealed that levels of sTim-3 were negatively correlated with levels of IL-12 (Figure 3). sTim-3 may interfere with the regulation of Tim-3/Gal-9 during MTB infection. High levels of sTim-3 in the sera of PTB patients may represent a mechanism of immune escape by MTB mediated by inhibiting the activation of innate immune cells and production of cytokines.
When the innate immune response cannot completely clear MTB, adaptive immune responses are elicited. As a component of adaptive immunity, CD4+ T cells play a significant role in responses against intracellular bacteria. 45 To investigate whether levels of innate immune-related cytokines (IL-12, IL-23, and IL-18) affected the secretion and release of Th cell-related cytokines (IFN-γ, IL-4, and IL-33) in patients with PTB, we further examined correlations among serum levels of Th cytokines and innate immune cell-related cytokines. Levels of IL-12, IL-23, IL-18, and IFN-γ were positively correlated, while levels of IL-12 were negatively correlated with those of IL-4.
Our results are consistent with other findings. IL-12, IL-23, and IL-18 have been shown to be upstream regulatory factors that promote the proliferation and differentiation of Th0 cells into Th1 cells, which can subsequently induce Th1 cells to secrete IFN-γ and other cytokines;16,46,47 IL-12 acts synergistically with IL-18 to promote secretion of IFN-γ by Th1 cells. 48 In addition, the negative correlation between levels of IL-12 and IL-4 is consistent with the finding that IL-12 can significantly inhibit the secretion of Th2 cytokines. 49 It has been further suggested that insufficient production of IL-12 may affect the differentiation of Th0 cells into Th1 cells 50 and increase Th2 cell reactivity.
In this study, levels of IFN-γ were significantly lower and levels of IL-4 and IL-33 were significantly higher in patients with PTB compared with healthy controls. The Th1/Th2 paradigm suggests that differentiation of CD4+ T cells into Th1 (cell-mediated) and Th2 (humoral) subsets is the core of appropriate induction of protective immunity. 51
To clarify whether serum levels of immune molecules can be used as diagnostic markers of PTB, we further performed ROC curve analysis. Serum levels of sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 may all have diagnostic value for PTB. Thus, these immune molecules may be helpful for diagnosis as well as monitoring the development and progression of PTB.
The following limitations of the study should be acknowledged. First, this was a retrospective case–control study, so it could not demonstrate causal relationships. The correlations between risk factors observed in our study and dose–effect relationships need to be further verified in large-scale studies in different regions. Second, we did not use a detailed classification of patients with PTB in this study. In future work, we plan to include patients with different disease progression (such as latent PTB) for more in-depth and detailed analysis. Third, this was only a descriptive study because of time, cost, and energy limitations. Further studies are required to confirm the expression of inhibitory costimulatory molecules and related cytokines by quantitative real–time PCR or western blotting. We hope that further studies will be able to fully elucidate the effects of environmental factors on immune molecules and to define the relationships between inhibitory receptors and immune cytokines, such as PD-1/PD-L1 and Tim-3/Gal-9, during tuberculosis infection.
Our study demonstrated that demographic characteristics, living habits, environment, clinical condition, and medical history may impact the occurrence of PTB. Drinking alcohol may affect serum levels of sTim-3. High levels of sTim-3 may play an important role in negative regulation of IL-12. IL-12 may downregulate serum levels of IFN-γ in patients with PTB. Furthermore, sPD-L1, sTim-3, sGal-9, IL-12, IL-23, IL-18, IFN-γ, IL-4, and IL-33 may represent valuable biomarkers for diagnosis of PTB. Therefore, in addition to improving lifestyle, workplace and living environments, and other factors, inhibitory costimulatory molecules may be targets for controlling MTB infection. This finding may provide new strategies for treatment of tuberculosis based on regulation of immune function.
Supplemental Material
sj-xlsx-1-imr-10.1177_03000605211036832 - Supplemental material for Serum levels of inhibitory costimulatory molecules and correlations with levels of innate immune cytokines in patients with pulmonary tuberculosis
Supplemental material, sj-xlsx-1-imr-10.1177_03000605211036832 for Serum levels of inhibitory costimulatory molecules and correlations with levels of innate immune cytokines in patients with pulmonary tuberculosis by Yunjuan Zhao, Jia Zhang, Bing Xue, Fengbo Zhang, Qian Xu, Haimei Ma, Tong Sha, Lei Peng, Fengsen Li and Jianbing Ding in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605211036832 - Supplemental material for Serum levels of inhibitory costimulatory molecules and correlations with levels of innate immune cytokines in patients with pulmonary tuberculosis
Supplemental material, sj-pdf-2-imr-10.1177_03000605211036832 for Serum levels of inhibitory costimulatory molecules and correlations with levels of innate immune cytokines in patients with pulmonary tuberculosis by Yunjuan Zhao, Jia Zhang, Bing Xue, Fengbo Zhang, Qian Xu, Haimei Ma, Tong Sha, Lei Peng, Fengsen Li and Jianbing Ding in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605211036832 - Supplemental material for Serum levels of inhibitory costimulatory molecules and correlations with levels of innate immune cytokines in patients with pulmonary tuberculosis
Supplemental material, sj-pdf-3-imr-10.1177_03000605211036832 for Serum levels of inhibitory costimulatory molecules and correlations with levels of innate immune cytokines in patients with pulmonary tuberculosis by Yunjuan Zhao, Jia Zhang, Bing Xue, Fengbo Zhang, Qian Xu, Haimei Ma, Tong Sha, Lei Peng, Fengsen Li and Jianbing Ding in Journal of International Medical Research
Footnotes
Acknowledgements
The authors are grateful to the First, Fourth, and Fifth Affiliated Hospitals of Xinjiang Medical University, China.
Authors’ contributions
Fengsen Li and Jianbing Ding contributed to study conception, design, and data validation, as well as review and revision of the manuscript; Yunjuan Zhao performed the experiments, analyzed the data, drafted the initial manuscript, and acquired funding. Jia Zhang, Bing Xue, Qian Xu, Haimei Ma, and Tong Sha were responsible for collecting blood samples and performing the experiments. Fengbo Zhang and Lei Peng contributed to data collection, analysis, and data curation. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Major Science and Technology Special Project of Xinjiang Uygur Autonomous Region (2020A03004 and 2020A03004-1).
Availability of data and materials
The datasets generated and analyzed during the present study are available from the corresponding author upon reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
