Abstract
Objective
To investigate whether GSTA1, GSTO2, and GSTZ1 are relevant to an increased risk of amyotrophic lateral sclerosis (ALS) in a Chinese population.
Methods
In this study, 143 sporadic ALS (sALS) patients (83 men, 60 women) and 210 age- and sex-matched healthy subjects were enrolled. Blood samples were collected by venipuncture. Genomic DNA was isolated by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) according to the manufacturer’s instructions. The potential associations between ALS and GSTA1, GSTO2, and GSTZ1 polymorphisms were estimated using chi-squared analysis and unconditional logistic regression.
Results
The D allele and genotype frequencies of GSTO2 were increased in sALS patients compared with healthy subjects, indicating that the GSTO2 DD genotype was associated with an increased risk of sALS (odds ratio [OR] = 3.294, 95% confidence interval [CI] = 1.039–10.448). However, a significant association between the DD genotype and the risk of sALS was evident in men only (OR = 7.167, 95% CI = 1.381–37.202).
Conclusion
This study revealed that the D allele and genotype frequencies of GSTO2 were increased in sALS patients. The GSTO2 DD genotype was associated with an increased risk of sALS in men in a Chinese population.
Introduction
Amyotrophic lateral sclerosis (ALS) is an adult-onset degenerative disorder characterized by the death of motor neurons in the cortex, brain stem, and spinal cord. This leads to progressive muscle weakness, atrophy, and death from respiratory failure within 3 to 5 years of symptom onset. The cause of the disease and the mechanisms underlying the premature death of motor neurons remain unclear. 1 Most ALS cases occur in sporadic form (sporadic ALS [sALS]), while 10% to 15% of cases have a positive family history (familial ALS [fALS]).
Mutations in the Cu/Zn superoxide dismutase gene (SOD1) are responsible for approximately 20% of fALS. 2 A growing body of evidence indicates that the excessive production of reactive oxygen species and the insufficient activity of antioxidant defense mechanisms may play an important role in the pathogenesis of many neurodegenerative diseases, including ALS. 3 Glutathione S-transferases (GSTs) are a family of eukaryotic and prokaryotic phase II metabolic isozymes. GSTs regulate the detoxification of electrophilic compounds and organic peroxides. 4 The antioxidant activities of GSTs are believed to be neuroprotective by reducing oxidative stress. 5
GST isoforms are widely distributed in mammalian tissue, including in the central nervous system. Human cytosolic GSTs belong to different classes, known as alpha, zeta, theta, mu, pi, etc. GST Mu 1 (GSTM1), GST Theta 1 (GSTT1), and GST Pi 1 (GSTP1) have been identified as genetic polymorphisms of GSTs. 6 Single nucleotide polymorphisms (SNPs) in the promoter region of the GST Alpha 1 (GSTA1) gene have also been identified. 7 Furthermore, polymorphisms in GSTM1, GSTT1, and GSTP1 have been investigated for their oxidation and detoxification capactities. 8 , 9 The GSTA1*A haplotype (-567T, -69C, -52G) has more potent enzyme activity than the GSTA1*B haplotype (-567G, -69T, -52A). 10 The human GST Omega 2 (GSTO2) protein has 243 amino acids and its genetic polymorphism results from the replacement of N142D in the coding region. 11 It has been reported that the expression of the GSTO2 rs156697 (D142) variant is 20% lower than the wild type (N142) allozyme. 12 The GSTZ1 rs3177427 and GSTZ1 rs7972 polymorphisms are caused by an A→G transition and result in amino acid substitutions. 13 These amino acid alterations affect the activities of the resultant GSTZ1 enzyme for different substrates. 14
GSTA1 is the most abundant of the alpha-class GST isoenzymes, which can inactivate quinones. 15 Twenty SNPs in GSTA1 are located in the promoter region of this gene. Some studies have shown that GSTA1 variants have a pathogenic effect in many diseases. 16 , 17 There are three common nonsynonymous SNPs in the GST Zeta 1 (GSTZ1) gene, resulting in four common protein isomers 18 , 19 and one rare protein isomer. 20 Although many genetic polymorphisms are associated with sALS, the roles of GSTA1, GSTO2, and GSTZ1 in sALS have not yet been investigated. The present study aimed to investigate whether GSTA1, GSTO2, and GSTZ1 polymorphisms are associated with an increased risk of ALS in a Chinese population.
Materials and methods
Study population
In this study, sALS patients and age- and sex-matched healthy subjects were enrolled. All patients and healthy subjects were from the Chinese Han population. Patients were diagnosed according to the El Escorial criteria. Patient diagnosis was confirmed by two board-certified neurologists and stratified as definite, probable, or possible. No family history of ALS was evident in any patients, and the healthy subjects had no known hereditary disorders. The study was reviewed and approved by the ethics committee of the Second Hospital of Hebei Medical University (approval number 2016-R026). Written informed consent was obtained from all participants. The reporting of this study conforms to the STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) statement. 21
Genomic DNA extraction
Blood samples were collected by venipuncture. Genomic DNA was isolated using High Pure polymerase chain reaction (PCR) Template Preparation Kits (Roche Diagnostics GmbH, Mannheim, Germany) according to the manufacturer’s instructions.
Genotyping
Analysis of the GSTA1 rs3957357 polymorphism: The GSTA1 rs3957357 polymorphism was determined using PCR-restriction fragment length polymorphism (PCR-RFLP). The primers for GSTA1 rs3957357 were 5′-CCCTACATGGTATAGGTGAAAT-3′ and 5′-GTGCTAAGGACACATATTAGCA-3′. PCR was performed on 50-µL samples, each containing genomic DNA (0.1 mg), PCR primers (0.5 mM), deoxyribonucleoside triphosphates (0.2 mM), magnesium chloride (2 mM), and TaKaRa Ex Taq DNA polymerase (1 U; Takara Bio Inc., Shiga, Japan). The amplification was performed with 95°C initial denaturation, followed by 35 cycles of 96°C denaturation for 1 minute, 63°C annealing for 1 minute, and 72°C extension for 1 minute, and finally 72°C extension for 6 minutes before the reaction was cooled to 4°C. After adding Hinf I (Takara Bio Inc.), the restriction enzyme digestion was performed at 37°C for 2 hours. The -69C allele was cut into 525-bp, 254-bp, and 42-bp fragments. The samples containing the -69T allele produced 525-bp, 197-bp, 57-bp, and 42-bp fragments. The products were identified using ethidium bromide after separation in a 3% agarose gel.
Analysis of the GSTO2 rs156697 polymorphism: The GSTO2 rs156697 polymorphism was determined using the PCR-RFLP method described by Masoudi et al. 12 The primers for GSTO2 were 5′-AACCCTCCTAAAGCACCC-3′ and 5′-GCCTGTGAAAGCTG GTGTTAG-3′. The amplification was performed on 20-µL samples, each containing MgCl2 (1.5 mM), deoxynucleotide triphosphates (0.2 mM each), primers (0.5 pmol each), 10× PCR buffer (0.2 µL; CinnaGen Co., Tehran, Iran), and Taq DNA polymerase (1 U). The thermal conditions of PCR consisted of 5 minutes of initial denaturation at 94°C, followed by 35 cycles of 94°C for 1 minute, annealing at 62.5°C for 1 minute, and 72°C for 1 minute. The amplified segments were treated with MboI restriction enzyme and incubated overnight at 37°C. Following ethidium bromide staining after separation in a 2.5% agarose gel, 420 bp, 420/287/133 bp, and 287/133 bp bands were visualized for the NN, ND, and DD genotypes, respectively.
Analysis of the GSTZ1 polymorphisms: The GSTZ1 polymorphisms were determined using the PCR-RFLP method described by Menegon et al. 13 The GSTZ1 rs3177427 polymorphism is caused by an A→G transition. This transition was detected using PCR with the primers 5′-TGACCACCCAGAAGTGTTAG-3′ and 5′-AGTCCACAAGACACAGGTTC-3′, followed by digestion of the product with Alw26 I, and analysis of the digestion products after separation by 12% polyacrylamide gel electrophoresis. The GSTZ1 rs7972 polymorphism is also caused by an A→G transition. This transition was detected using PCR with the primers 5′-TGACCACCCAGAAGTGTTAG-3′ and 5′-AGTCCACAAGACACAGGTTC-3′, followed by digestion of the product with Fok I, and analysis of the digestion products after separation by 12% polyacrylamide gel electrophoresis. Genotypes were determined as AA (308 bp), GG (115/93 bp), or AG (308/115/93 bp) for the rs7972 polymorphism, and AA (186/125 bp), GG (159/125/26 bp), or AG (186/159/125/26 bp) for the rs3177427 polymorphism.
Statistical analysis
Independent sample t-tests and chi-square tests were used to compare the basic characteristics of the patients and healthy subjects. The chi-square test was used to determine whether samples from healthy subjects and patients showed the Hardy–Weinberg equilibrium. The associations between ALS and the GSTA1, GSTO2, and GSTZ1 polymorphisms were estimated using the chi-square test. The odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using unconditional logistic regression.
Fisher's exact test was used to evaluate the allele associations. Logistic regression was used to analyze the codominant, dominant, recessive, overdominant and logarithmic genetic models, and to determine the ORs. Akaike information criterion and Bayesian information criterion were used to select the best genetic model. All statistical analyses were performed using SPSS for Windows, Version 13.0 (SPSS Inc., Chicago, IL, USA) and SNPStats software (https://www.snpstats.net/start.htm). All statistical tests were two-sided and P < 0.05 was considered to be statistically significant.
In the present study, we assumed that there was comparability between the experimental group and the control group when there was no significant difference in basic information. We also assumed that the effects of other genes were consistent and comparable.
Results
In this study, 143 sALS patients (83 men, 60 women) and 210 age- and sex-matched healthy subjects were enrolled. Patient diagnoses were classified as definite (n = 101; 70.6%), probable (n = 34; 23.8%), or possible (n = 8; 5.6%). The patients had a median age of 52.20 ± 10.25 years (range 27–79 years) at onset, and included 21 patients with early onset (≤40 years) and five patients with late onset (>70 years). The patients were further classified into subgroups by duration of symptoms of ≤36 months (n = 111) or >36 months (n = 32), and by bulbar onset (n = 22) or spinal onset (n = 121). The median age at examination of the 210 healthy subjects was 49.27 ± 9.34 years (range 22–72 years).
Analyses of the characteristics of the study participants revealed no differences in age and sex distribution between the ALS patients and healthy subjects. All samples from both the patients and healthy subjects were in the Hardy–Weinberg equilibrium. The GSTZ1 rs7972 variant was identified as monomorphic in our dataset.
The D allele and genotype frequencies of GSTO2 were significantly higher in ALS patients compared with healthy subjects (P = 0.001 and P = 0.001). Moreover, logistic regression analysis revealed that the D allele of the GSTO2 gene was significantly associated with an increased risk of sALS (OR = 1.881, 95% CI = 1.316–2.688, P = 0.001). The combined ND+DD genotype of GSTO2 was also correlated with an increased risk of sALS (OR = 2.206, 95% CI = 1.426–3.411, P < 0.001). When analyzed separately, the OR of the DD genotype (OR = 3.294, 95% CI = 1.039–10.448, P = 0.043) was significantly greater than that of the ND genotype (OR = 2.122, 95% CI = 1.356–3.320, P = 0.001). No significant changes were detected in the allele or genotype frequencies of GSTA1 or GSTZ1 in the sALS patients (Table 1).
Distribution of genotype and allele frequencies in ALS patients and control groups.
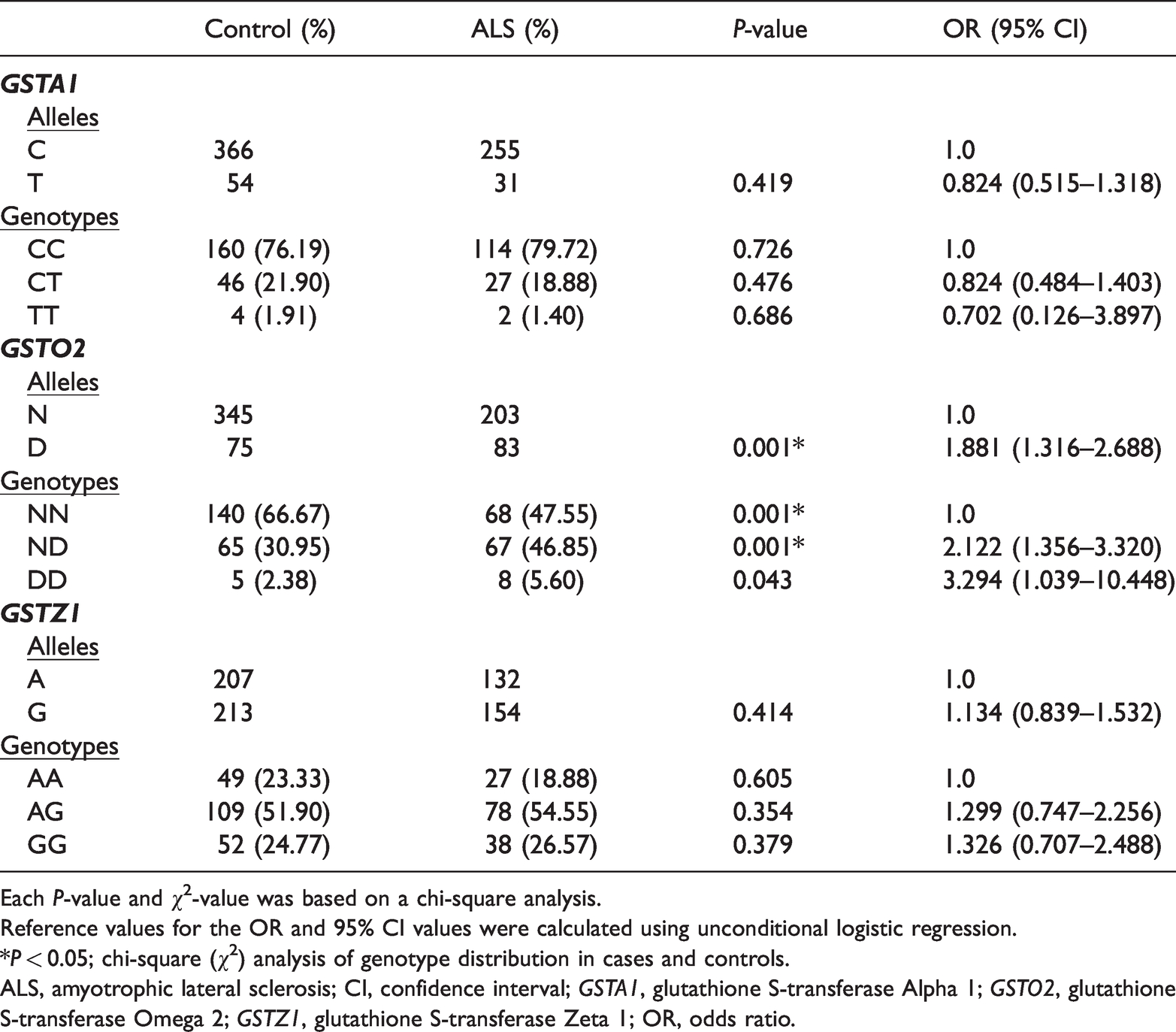
Each P-value and χ2-value was based on a chi-square analysis.
Reference values for the OR and 95% CI values were calculated using unconditional logistic regression.
*P < 0.05; chi-square (χ2) analysis of genotype distribution in cases and controls.
ALS, amyotrophic lateral sclerosis; CI, confidence interval; GSTA1, glutathione S-transferase Alpha 1; GSTO2, glutathione S-transferase Omega 2; GSTZ1, glutathione S-transferase Zeta 1; OR, odds ratio.
To further explore the relationship between the GSTO2 rs156697 polymorphism and ALS susceptibility, we conducted a genetic model analysis (Table 2). The results revealed significant differences between the case and control groups in the codominant, dominant and overdominant models (P < 0.05).
Comparison of the genetic inheritance models for the GSTO2 rs156697 polymorphism.

AIC, Akaike information criterion; ALS, amyotrophic lateral sclerosis; BIC, Bayesian information criterion; CI, confidence interval; OR, odds ratio.
When the GSTO2 polymorphisms were stratified by sex, there was an increase in the D allele and genotype frequencies in men but not in women in the ALS group, indicating that the D allele of GSTO2 is only associated with an increased risk of sALS in men (OR = 2.543, 95% CI = 1.580–4.091, P < 0.001). Similarly, the frequencies of the ND (OR = 2.881, 95% CI = 1.584–5.240, P = 0.001), DD (OR = 7.167, 95% CI = 1.381–37.202, P = 0.019), and ND+DD (OR = 3.119, 95% CI = 1.741–5.587, P < 0.001) genotypes were associated with an increased risk of sALS in men but not women; of these, the OR for the DD genotype was the greatest (Table 3).
Distribution of genotypes and allele frequencies of GSTO2 in ALS patients and control groups stratified by sex.

Each P-value and χ2-value was based on chi-square analysis.
Reference values for the OR and 95% CI values were calculated using unconditional logistic regression.
*P < 0.05; chi-square analysis of genotype distribution in cases and controls.
ALS, amyotrophic lateral sclerosis; CI, confidence interval; OR, odds ratio.
There were no significant correlations between GSTA1, GSTO2, or GSTZ1 polymorphisms and the age of onset, site of onset, or survival time of sALS.
Discussion
GST plays an important role in the detoxification of reactive intermediates resulting from a diverse range of cytotoxic drugs. It is also vital for the neutralization of reactive oxygen species, and acts as a critical enzyme in the defense against oxidative stress in neurons. An association has also been reported between the age of onset of Alzheimer’s disease, Parkinson’s disease, and fALS and mutations in GSTO1 and GSTO2. 22 A reduction in GST activity may be attributable to motor neuron damage; a compromised protective barrier formed by GST in the blood of ALS patients may lead to an increased vulnerability to the toxic effects of electrophilic compounds and organic peroxides. 23
In the present study, GSTO2 D allele and genotype frequencies were significantly higher in sALS patients than in healthy subjects. An increased frequency of the D allele of GSTO2 was closely associated with an increased risk of sALS. In addition, the combined ND+DD genotype of the GSTO2 gene was associated with an increased risk of sALS. When analyzed separately, the DD genotype exhibited a greater OR for sALS than the ND genotype.
The male prevalence of ALS has been reported generally in the literature, 24 , 25 and the male/female ratio of ALS was reported as 1.6:1 in a study conducted in Minas Gerais (Brazil). 26 However, the underlying mechanisms of sex-based differences in ALS remain unclear. In the present study, GSTA1 and GSTZ1 polymorphisms did not correlate with sALS risk. However, the GSTO2 gene had significant differences in allele and genotype frequencies in sALS patients, and this was stratified by sex: the association between GSTO2 polymorphisms and the risk of sALS was significant in men but not women. Some previous studies have suggested that such differences may be determined by differing hormonal statuses between men and women. 27 , 28
Few studies have investigated GST polymorphisms and ALS, but there have been many studies on GST variants in other neurodegenerative diseases. Associations between GSTO1 and GSTO2 and the age of onset of Alzheimer's disease, 29 Parkinson's disease, 30 and fALS have been reported. 22 Furthermore, in a study evaluating 174 families with Parkinson's disease, it was reported that GSTO1 and GSTO2 polymorphisms can affect the age of onset of both Parkinson's and Alzheimer's diseases. 31 However, no association between GSTO1 and Parkinson’s disease was identified in an Australian case–control study. 32 In the current study, none of the investigated GST polymorphisms were correlated with the age of onset, site of onset, or survival time of sALS.
The major limitation of the present study was the small sample size, which may have reduced the statistical power for detecting changes, especially when the data were stratified by sex. In addition, some SNPs in GSTO2 (except rs156697) were not detected in the study. Therefore, future studies with larger sample sizes are warranted to confirm our findings of an association between GST polymorphisms and the risk of sALS.
Conclusion
Our results suggest that GSTO2 polymorphisms may play a role in the development of sALS. The GSTO2 DD genotype was associated with an increased risk of sALS in Chinese men.
