Abstract
Objective
Schizophrenia is a complex mental disorder with high heritability. The hypothalamic–pituitary–adrenal (HPA) axis, which is the stress system of the neuroendocrine system, is considered to impact psychotic disorders. We hypothesized that polymorphisms of HPA axis genes might be involved in the development of schizophrenia.
Methods
A case–control study comprising 234 patients with schizophrenia and 399 matched healthy controls was conducted to investigate the association between the human melanocortin 2 receptor (MC2R) gene and schizophrenia risk. Seven tag single nucleotide polymorphisms (SNPs) (rs16941303, rs16941314, rs2186944, rs28926188, rs7230126, rs948322, and rs948331) of MC2R were genotyped by direct sequencing.
Results
No significant associations were observed between any of the alleles, genotypes, or haplotypes examined within the MC2R gene and the risk of schizophrenia in the total group or in subgroups stratified by smoking or alcoholism. However, a subgroup analysis stratified by sex revealed that under the additive model, the C allele of the MC2R rs948331 SNP significantly decreased the risk of schizophrenia in females (odds ratio=0.18).
Conclusion
The C allele of the MC2R rs948331 locus may be a protective factor, reducing the risk of schizophrenia in the female Han Chinese population.
Keywords
Introduction
Schizophrenia, a complex disorder typified by delusions, hallucinations and thought disorders, has both genetic and environmental triggers. 1 Stress sensitivity and activity of the hypothalamic–pituitary–adrenocortical (HPA) axis have been implicated in the development of schizophrenia. 2 When exposed to stress, the HPA axis is activated by binding of corticotropin-releasing hormone (CRH) receptors, which stimulates adrenocorticotrophic hormone (ACTH) release 3 and aids in inhibiting CRH expression in amygdala neurons via negative feedback. ACTH can induce the formation of myelin sheaths and dendrites by increasing the circulating sugar content, thus promoting brain maturation and shortening the vulnerable period. It can also act as a neuroregulator and neurotransmitter that can affect nervous system development, whereby it plays roles in preventing convulsion, improving learning and memory functions, and reducing attention deficits.4–6 ACTH can have pharmacological effects by binding to the melanocortin 2 receptor (MC2R, also known as the ACTH receptor). 7 Recently, five MCR genes (MC1R, MC2R, MC3R, MC4R, and MC5R) were identified. MC2R is an important member of this family that is mainly distributed in the adrenal cortex and binds ACTH as a ligand. 8
Thus, it is plausible that variants of this receptor gene may be involved in disease pathogenesis and drug resistance. Single nucleotide polymorphisms (SNPs) in the coding region of the MC2R gene could influence the molecular conformation of the ATCH receptor. Such structural differences could influence the efficiency of ligand binding and the strength of the transduction of signals sent through intracellular pathways, leading to diseases such as infantile spasm, 9 alopecia areata, 10 heroin addiction, 11 and familial glucocorticoid deficiency.12,13
Few studies have examined the link between MC2R gene polymorphisms and schizophrenia susceptibility. Using a case–control method, we investigated MC2R gene polymorphisms to determine whether they contributed to the development of schizophrenia in a Chinese population.
Materials and methods
Patients
Patients with schizophrenia were recruited from the First Affiliated Hospital of Changsha Medical College; their diagnoses were confirmed by at least two independent experienced psychiatrists according to the criteria in the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision. Individuals without schizophrenia were recruited for the control group. This study followed the ethical standards of the 1964 Helsinki Declaration and its later amendments, as well as national and international guidelines. The research procedures were approved by the Ethics Committee of the Changsha Medical College, China (Ethical Committee of Changsha Medical College (CMCEC-1801022)). Written informed consent for genetic analyses was obtained from all subjects.
Genotyping
SNP selection was performed using Haploview software (https://sourceforge.net/projects/haploview/) choosing only SNPs with a minor allele frequency higher than 0.05 and r2≥0.8 based on the HapMap database (CHB, Chinese Han population) (http://coriell.org/1/NHGRI/Collections/HapMap-Collections/HapMap-Project). After screening, seven tag SNPs (rs16941303, rs16941314, rs2186944, rs28926188, rs7230126, rs948322, and rs948331) in the MC2R gene were selected. Genomic DNA was extracted from peripheral blood leukocytes using the standard phenol-chloroform method. Multiplex polymerase chain reaction (PCR) assays were performed on a GeneAmp 9700 PCR thermocycler (Applied Biosystems, Foster City, California, USA). The reactions were performed in a total volume of 10 µL, including 1 µL of genomic DNA, 1 µL of primer mix (1 µM of each primer), and 1 U of Hotstar Taq polymerase (Qiagen Inc., Valencia, CA, USA). The cycling parameters were as follows: 95°C for 2 minutes; 10 cycles at 95°C for 20 s, 65°C for 40 s, and 72° for 30 s; 25 cycles at 95°C for 20 s, 55°C for 30 s, and 72°C for 1 minute; then the samples were held at 4°C. The genotypes of the MC2R polymorphisms were determined by Sanger sequencing. A 3730XL genetic analyzer (Applied Biosystems) was used for sequencing. GeneScan ™3.7 software (Applied Biosystems) was used for data analysis.
Statistical analyses
The Hardy–Weinberg equilibrium (HWE) of all polymorphisms under study was assessed using a χ2 test. The distributions of the genetic models (allelic, dominant, additive, and recessive models) were compared between the control and case groups using a t test. All calculations were performed with Plink 1.0 software (http://softwaretopic.informer.com/plink-software/). Corresponding odds ratios (ORs) and 95% confidence intervals (CIs) were evaluated by logistic regression analysis. A stringent Bonferroni correction was performed to adjust the p-value (padj) in all cases in which an initial significant relationship was found. Squared correlation coefficients (r2) for each pair of polymorphic sites were calculated with Haploview software. A value of p<0.05 indicated statistical significance. The allele data from patients stratified into subgroups according to alcoholism and smoking were also analyzed. A power analysis was conducted using G*Power software (www.gpower.hhu.de).
Results
A total of 234 patients with schizophrenia and 399 healthy controls were included in this study. The case group consisted of 234 unrelated patients including 130 males and 104 females (age 33.1±7.2 years, mean±standard deviation (SD)). Within the case group, 7.4% of the patients had a history of alcoholism, and 35.0% had a history of smoking. In addition, 399 unrelated healthy individuals (302 males and 97 females; age 27.25±8.4 years (mean±SD)) who did not have schizophrenia according to clinical examinations performed at The First Affiliated Hospital of Changsha Medical College were recruited for the control group. Within the control group, 62.2% of the participants had a history of alcoholism, and 46.8% had a history of smoking (Table 1). No significant deviations from HWE were observed for any of the seven tag SNPs (Table 2). Linkage disequilibrium analysis indicated that rs16941303 and rs2186944, rs16941303 and rs28926188, and rs2186944 and rs28926188 exhibited strong linkage (r2>0.85) (Figure 1). Moreover, our study obtained >71.4% power at the 5% significance level (two-tailed), which indicates that the sample size was sufficiently large to detect an association with an OR of 1.5 (Table 2).
Characteristics of the case and control groups.

SD, standard deviation; BMI, body mass index.
Allele distributions of MC2R gene polymorphisms in schizophrenia patients (case) and healthy controls (control).

aThe OR and 95% CI are calculated for the minor allele of each polymorphism.
bPower calculations assume α=0.05 and a small effect size (0.1) or OR=1.5.
A, major allele; B, minor allele; SNP, single nucleotide polymorphism; OR, odds ratio; 95% CI, 95% confidence interval; HWE, Hardy–Weinberg equilibrium.

Linkage disequilibrium analysis of seven single nucleotide polymorphisms of MC2R in the case and control groups.
No association was detected between the allelic, additive, dominant, and recessive models of any of the seven SNPs in the MC2R gene and schizophrenia in the Han Chinese population before or after Bonferroni correction (Table 2 and 3). Seven major haplotypes were identified among the rs16941303, rs16941314, rs2186944, rs28926188, rs7230126, rs948322, and rs948331 loci (lowest frequency threshold >0.01). HaplotypeACGGTGT was the most prevalent in both the case and control groups, and no significant difference in its frequency was observed between patients with schizophrenia and controls. The frequency of haplotypeACGATAC was significantly increased in the control group versus the case group (p=0.039, OR [95% CI]=0.153 [0.020–1.191]), but significance was not obtained after correction (p=0.273) (Table 4).
Genetic model distributions of MC2R gene polymorphisms in the case and control groups.

ap values were calculated using Fisher’s exact test.
bThe OR [95% CI] was estimated by logistic regression analysis.
A, major allele; B, minor allele; SNP, single nucleotide polymorphism; OR, odds ratio; 95% CI, 95% confidence interval.
Frequencies of haplotypes (>1%) of the seven single nucleotide polymorphisms of MC2R in patients with schizophrenia (case) and healthy controls (control).

aThe Plink program was used to estimate common (frequency >0.01) haplotypes of the seven SNPs of MC2R (rs16941303, rs16941314, rs2186944, rs28926188, rs7230126, rs948322, and rs948331).
bEach haplotype was compared with all other haplotypes combined.
cThe Bonferroni correction was applied to correct the p value.
SNP, single nucleotide polymorphism; OR, odds ratio; 95% CI, 95% confidence interval.
Analyses were performed on subgroups stratified by sex, smoking, and alcoholism. As shown in Table 5, the allele distributions under the allelic, additive, dominant, and recessive models for the MC2R rs16941303, rs16941314, rs2186944, rs28926188, rs7230126, and rs948322 loci did not significantly differ between the case and control groups. Interestingly, significant associations were observed under the allelic (p=0.046, OR [95% CI]=0.665 [0.445–0.992]) and additive models (p=0.0008, OR [95% CI]=0.18 [0.06–0.49]) for rs948331 and schizophrenia in female participants but not in male participants or in the smoking and alcoholism groups. The association under the allelic model disappeared after Bonferroni correction (p=0.362). A significant association remained under the additive model even after Bonferroni correction (p=0.0056).
The association between genetic models of MC2R single nucleotide polymorphisms and schizophrenia stratified by sex, smoking, and alcoholism.
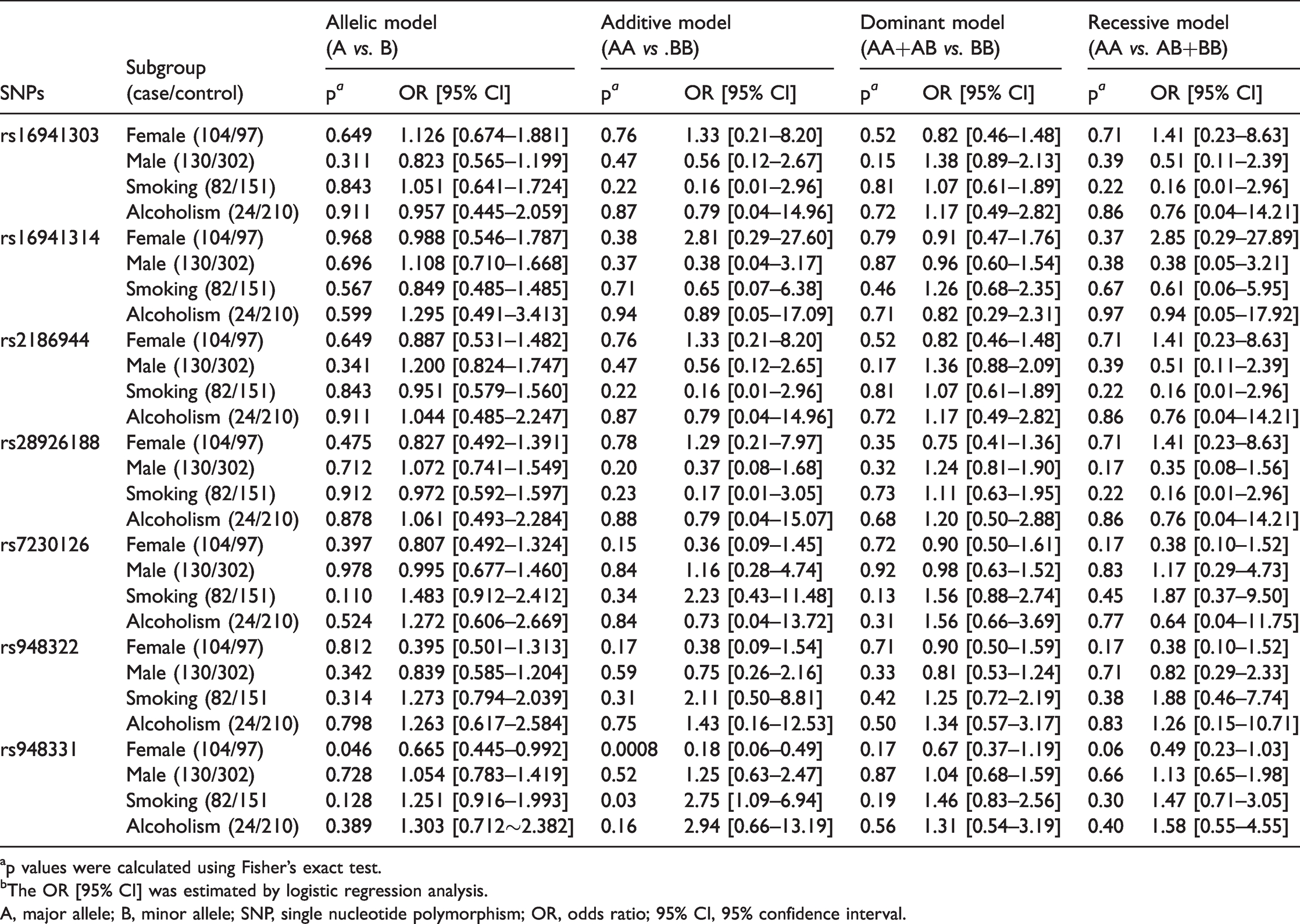
ap values were calculated using Fisher’s exact test.
bThe OR [95% CI] was estimated by logistic regression analysis.
A, major allele; B, minor allele; SNP, single nucleotide polymorphism; OR, odds ratio; 95% CI, 95% confidence interval.
The haplotypes were also analyzed based on sex, smoking, and alcoholism (Table 6), and the frequency of haplotypeACGGTGT in patients with schizophrenia was significantly lower than that in controls among the female subgroup (p=0.041, OR [95% CI]=1.508 [1.015–2.240]). The frequency of haplotypeACGGTGT in patients with schizophrenia was also significantly lower than that in the controls in the smoking subgroup (p=0.031, OR [95% CI]=0.659 [0.448–0.968]). However, these significant associations disappeared after Bonferroni correction (females: p=0.287; smoking: 0.217).
The association between haplotypes (>1%) of the seven single nucleotide polymorphisms of MC2R and schizophrenia risk stratified by sex, smoking, and alcoholism.

aThe Plink program was used to estimate common (frequency >0.01) haplotypes of the seven SNPs of MC2R (rs16941303, rs16941314, rs2186944, rs28926188, rs7230126, rs948322, and rs948331).
bEach haplotype was compared with all other haplotypes combined.
cThe Bonferroni correction was applied to correct the p value.
SNP, single nucleotide polymorphism; OR, odds ratio; 95% CI, 95% confidence interval.
Discussion
The HPA axis is an important aspect of the neuroendocrine system. 14 Under normal physiological conditions, the final products released by the HPA axis, corticosteroids (such as cortisol), regulate physiological and psychological stress through complex feedback mechanisms, helping the body maintain homeostasis.15,16 Continuous stress induces hyperactivity of the HPA axis, increasing corticosteroid secretion, and eventually disrupting the HPA axis function. 17 In the HPA axis, ACTH can promote adrenal glucocorticoid secretion by binding to ACTH receptor 1, which then increases plasma cortisol levels. 18 In contrast, there are inconsistencies among the reported results with regard to plasma ACTH levels in patients with schizophrenia. Duval et al. did not find any differences between their patient and control groups, 19 while Jiang et al. 20 reported that the plasma ACTH levels of patients with schizophrenia were higher than the normal level. These contradictory results may be caused by the different methods of ACTH determination used in these respective studies.
The MC2R gene maps to chromosome l8p l1.2. 21 Missense mutations and variants in the MC2R gene may result in the loss of signal transduction following ligand binding or may affect the affinity of ligand binding, but both types of defects would result in impaired receptor function. 22 Further studies are needed to investigate functional variants that are either regulated naturally or alternatively spliced upon ACTH release. Interestingly, one report described an association between polymorphisms of the MC2R gene in relation to other psychotic disorders including infantile spasm 9 and heroin addiction. 11 In this case, the heterogeneity of different diseases might explain the differences between their data and those of the present study. Follow-up studies with a larger population and other MC2R polymorphisms are required to determine the complex relationship of genetic and environmental factors in the development of schizophrenia.
Interestingly, we detected a significant association between the additive model of MC2R rs948331 and schizophrenia in a female Han Chinese population. To our knowledge, this is the first time that a significant relationship has been identified between any variant in the MC2R gene and schizophrenia in the Han Chinese population. This SNP is located in the 3ʹ-untranslated region of MC2R, but its physiological effect is unknown. We hypothesize that this polymorphism may decrease MC2R expression, resulting in a reduction of plasma cortisol, which would then increase the synthesis and secretion of CRH via a negative feedback loop to protect against schizophrenia. Based on the knowledge that autoimmune disorders can be triggered in women following menarche or childbirth, resulting in increased risk of a myriad of autoimmune diseases, it is likely that other factors that affect the autoimmune axis and the HPA axis may indirectly affect the cognitive functions affected by these axes. 23 Most neuroendocrine studies have focused on pharmacology, disease course, and symptoms without considering sex differences. 24 Our results suggest that sex might be an important factor in the genetic association between the MC2R rs948331 locus alleles and the pathogenesis of schizophrenia.
Several limitations of this study must be considered. First, the positive association found in this study may be a result of type I error; however, this limitation was addressed using the Bonferroni correction. Second, the sample size of this study is relatively small; however, a power analysis showed a 71.4% probability of detecting an effect size of 0.5 at the 0.05 significance level. Therefore, based on these caveats, we suggest that the association between the MC2R rs948331 locus and susceptibility to schizophrenia should be replicated in larger studies and among additional ethnic populations. Third, the definitions of types of alcoholism and smoking (chronic and acute) among patients in the study were not precise, which could have influenced the consistency of our results.
Conclusions
Our study showed for the first time that under the additive model, the C allele of the MC2R rs948331 locus might be a protective factor against schizophrenia among the female Chinese Han population.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211029504 - Supplemental material for A variant in the 3ʹ-untranslated region of the MC2R gene decreases the risk of schizophrenia in a female Han Chinese population
Supplemental material, sj-pdf-1-imr-10.1177_03000605211029504 for A variant in the 3ʹ-untranslated region of the MC2R gene decreases the risk of schizophrenia in a female Han Chinese population by Liang Tang, Qin Xiang, Ju Xiang, Jianming Li and Danna Chen in Journal of International Medical Research
Footnotes
Acknowledgment
We are grateful to all of the individuals who participated in this study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The present study was funded by the National Natural Science Foundation of China (Grant No. 81873780, 61702054); Hunan Natural Science Foundation Youth Program (2019JJ50697, 2018JJ3568, 2018JJ3573); Changsha Outstanding Innovative Young People Training Scheme (kq2009095, kq2009093, kq2004077, kq2009092); Foundation of the Education Department of Hunan Province (19A058, 19B072, 18A493); Foundation of the Health and Family Planning Commission of Hunan Province (20201918, 20201910); Application Characteristic Discipline of Hunan Province; Hunan Key Laboratory Cultivation Base of the Research and Development of Novel Pharmaceutical Preparations (No. 2016TP1029); and Project of Changsha Science and Technology (kq2004077).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
