Abstract
Objective
Major depressive disorder is associated with abnormal functioning of the hypothalamic–pituitary–adrenal (HPA) axis. Studies using hair cortisol to measure the effect of antidepressants on the HPA axis are lacking. The aim of this study was to explore the long-term effects of antidepressants on hair cortisol concentration (HCC).
Methods
Participants were 42 patients and 36 healthy individuals. The patients took antidepressants for 4 weeks. Patient HCC was measured pre-treatment and post-treatment. The HCC of healthy controls was also measured.
Results
Patient post-treatment HCC (mean ± standard deviation: 34.40 ± 32.57 pmol/mg) was significantly higher than patient pre-treatment HCC (17.42 ± 12.40 pmol/mg) and healthy control HCC (10.22 ± 7.99 pmol/mg). No significant correlation was found between Hamilton Depression Rating Scale scores and HCC at pre-treatment or post-treatment.
Conclusions
Hair cortisol concentration analysis could be used to monitor the dynamics of the effects of antidepressants on the HPA axis.
Keywords
Introduction
Major depressive disorder (MDD) is a serious mental health problem characterized by emotional distress, hopelessness and low activity, which greatly reduce quality of life. 1 MDD may cause serious economic and health problems and even increase the risk of suicide.
The activity of the hypothalamic–pituitary–adrenal (HPA) axis is a key pathophysiological correlate of depression; patients with depression show excessive cortisol,2–4 a measure of HPA axis activity. 5 Traditionally, cortisol concentrations have been measured in serum, saliva and urine, and show elevated levels in depressed patients.6–9 After antidepressant treatment, HPA axis function is normal and plasma cortisol levels decrease.10–11 However, serum, salivary and urinary measures of long-term cortisol levels can be unreliable owing to circadian rhythm variation, sample collection at different time points, food intake and exercise habits.12–15
More recently, hair has been used to measure long-term cortisol levels;16–18 this measure can establish the average cortisol secretion level between 1 month and 12 months.19–21 In addition, hair cortisol concentration (HCC) is not affected by confounding factors, such as age, tobacco use, hair curvature, washed and unwashed hair, and frequency of hair washing.22–24 A previous study comparing cortisol levels in hair to levels obtained with other measures suggests that HCC is very sensitive to HPA axis activity. 25
Previous studies of patients with depression have shown that serum cortisol levels are substantially reduced after antidepressant treatment.11,26–30 Dettenborn et al. point out the lack of studies using hair cortisol to measure the effects of antidepressants on the HPA axis. 6 The aim of this study was to compare HCC in male and female patients with depression to HCC in healthy controls, and to explore the long-term effects of antidepressants on HCC.
Methods
Participants
Forty-two patients (aged 18–55 years) were recruited from the Mental Health Center of West China Hospital of Sichuan University and from the Mental Health Center of Hebei Province between February 2013 and February 2014. All patients were assessed by trained psychiatrists using the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV). The inclusion criteria were (1) duration of depression 1 to 2 months, including first-episodic and recurrent (defined as having depression again after being cured) depression and (2) antidepressant-naive or antidepressant-free for at least 6 months. The exclusion criteria were (1) obvious psychosocial reasons for depression, such as marriage failure, relationship problems, job failure, economic problems and traffic accidents; (2) use of steroid medication; (3) physical illness (e.g. encephalitis, brain injury, epilepsy and endocrine diseases); (4) other axis I psychiatric disorders (e.g. dementia, generalized anxiety disorder, schizophrenia, obsessive–compulsive disorder, bipolar disorder, and alcohol or substance abuse; and (5) pregnant or breast feeding.
We also recruited 36 pairwise and age-matched healthy controls who met the following criteria: (1) normal social function; (2) no psychiatric disease; and (3) no physical illness. Any healthy controls or patients with dyed hair were excluded, as bleaching and dyeing can affect HCC. 19 All participants provided written informed consent before participating in the research, and the research was approved by the Institutional Ethics Committee of Sichuan University.
Because some individuals’ hair was too short to provide an adequate sample, we did not analyse data from all patients taking antidepressants. The patients took medication for 4 weeks. During the treatment period, 15 patients were temporarily treated for sleep disorders with benzodiazepines or sleep medications, and were not treated with other types of antipsychotic drugs. The control group received no intervention.
Hair collection and cortisol analysis
Hair samples were collected as previously described.31,32 Hair strands were cut using scissors from a posterior vertex position as proximal as possible to the scalp, wrapped in aluminium foil and stored in separate airtight plastic bags at room temperature until extraction. 33 For controls, a 1-cm segment close to the scalp was cut. For patients taking antidepressants, a 1-cm hair segment close to the scalp was cut and labelled as pre-treatment; after the treatment period, another 1-cm hair segment close to the scalp from the same region as the first cut was collected and labelled as post-treatment.
After sample collection, the samples were centralized to determine HCC and to ensure the consistency of the processing procedure. Hair cortisol levels were determined using an electrochemiluminescence immune assay with Elecsys analysers (Roche, Basel, Switzerland).
The hair cortisol extraction and analysis methods have been previously described.23,32–34 All samples were determined in duplicate, and the mean values of the two tests (pre-treatment and post-treatment) were analysed. The intra-assay and inter-assay coefficients of variation were below 8%.
Assessment of clinical symptoms
Patient depression severity (pre-treatment and post-treatment) was determined by trained psychiatrists using the 17-item Hamilton Depression Rating Scale (HAMD). 35
Statistical analysis
We conducted a test of normality of HCC variances using the K-S method and applied log transformations to ensure that the data conformed to the normal distribution. One-way analysis of variance (ANOVA) and χ2 tests were conducted to compare the characteristics of the three groups. We performed t-tests for independent samples to compare pre-treatment and post-treatment patient HAMD scores. One-way ANOVA was conducted to compare HCC means among patients at pre-treatment, patients at post-treatment, and healthy controls. If the ANOVA
Results
Sociodemographic characteristics
Forty-two patients and 36 healthy individuals completed the baseline HCC assessment. Of the 42 patients, 26 completed the post-treatment HCC assessment. Data were analysed from 26 patients: 10 patients taking escitalopram (mean = 16 mg/d); 7 patients taking paroxetine (mean = 31 mg/d); 3 patients taking sertraline (mean = 92 mg/d); 1 patient taking citalopram (mean = 10 mg/d); 3 patients taking venlafaxine (mean = 92 mg/d); 1 patient taking duloxetine (mean = 40 mg/d) and 1 patient taking mirtazapine (mean = 15 mg/d). There were no significant differences in age, sex or education years among patients at pre-treatment, patients at post-treatment, and healthy controls, but HAMD scores differed significantly between patients at pre-treatment and those at post-treatment (Table 1).
Comparison of sociodemographic characteristics between patients with depression and healthy controls.

SSRI: selective serotonin reuptake inhibitor; AD: antidepressant; Dual AD: venlafaxine or duloxetine; HAMD-17: Hamilton Depression Rating Scale.
Comparison of cortisol concentration
There was a significant difference in HCC among the three groups (F = 10.36,
Analysis of variance results comparing hair cortisol concentrations.
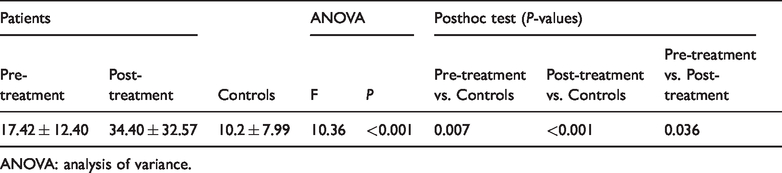
ANOVA: analysis of variance.
Correlation between HCC and HAMD scores
No significant correlation was found between HCC and HAMD scores at pre-treatment (r = 0.022) or post-treatment (r = 0.136).
Discussion
We found a significantly higher HCC for patients with depression at pre-treatment than for healthy controls. This result is consistent with previous findings that HCC is higher in patients with depression than in comparable controls. 6 Our results also extend previous work by Wei et al., who examined female patients with depression.6,31
After 4 weeks of antidepressant treatment, patient HCC was significantly higher than patient pre-treatment HCC and control HCC. HCC reflects the average level of cortisol over a period of time, which may differ from cortisol levels in serum or urine samples. 31 That is, the cortisol concentration in 1 cm of hair represents the average level over 1 month. We found that if depression was not well controlled while the patient was taking antidepressant medication, the cortisol concentration continued to increase, and then decreased over time. However, as the margin of decrease was small, the average cortisol concentration was still higher than that before treatment. This assumption requires further verification. Dettenborn et al. found that medicated patients with depression had elevated HCC compared with controls, and medicated patients had higher HCC in the first hair segment (the scalp near the posterior vertex position) (mean ± standard deviation: 26.7 ± 20.8 pg/mg) than in the second (21.9 ± 23.7 pg/mg), which suggests that cortisol concentrations did not return to normal 6 months after antidepressant treatment. 6 We need to extend the follow-up time to further observe changes in hair cortisol. Dettenborn et al. also suggested that studies of antidepressant effects on the HPA system should examine both the clinical response to treatment and the type of HPA axis measure.36–38 The long-term effects of antidepressant drugs on the HPA axis require further investigation before firm conclusions can be drawn.
Previous research suggests that high serum cortisol concentrations in depressed patients may also be influenced by the potentially stressful effects of hospitalization and testing, at least for the most vulnerable patients. 39 Interaction with physicians, the hospital setting (and separation from their families), different diet and concerns about treatment options may affect changes in hair cortisol levels in depressed patients. One study showed that unsatisfactory dyadic relationships may influence normal HPA axis function because of external stress. 40 Persons who experience poor dyadic relationships also show elevated HPA axis responses to stress. 41 Conversely, the use of couple-coping strategies is associated with overall marital satisfaction and also with reduced HPA axis responses to laboratory stressors.42,43 When a partner becomes a patient, this may affect the relationship and cause changes in cortisol concentrations.
One study found that hair cortisol measures are reliable, valid and convenient measures of cortisol in patients and healthy individuals, suggesting that hair cortisol may be a potential biomarker for prolonged cortisol exposure and thus useful in the development and implementation of more effective depression treatments.25,39,44 However, it remains unclear whether hair cortisol levels reflect adrenal cortex secretion or intrafollicular production, or both. Human hair follicles possess their own HPA axis, commonly referred to as the ‘peripheral’ HPA axis.45–49 Hair cortisol is likely to reflect the activity of hair follicle HPA axis activity and therefore represents a ‘peripheral’ rather than a ‘central’ response to stress. Therefore, we cannot rule out the possibility that the elevated HCC found for patients with depression in this study reflects only follicle-based production. However, hair cortisol levels in rhesus monkeys and healthy humans correlate highly with salivary cortisol samples.50,51 Sauve has reported that HCC correlates strongly with 24-hour urinary cortisol. 33 D’Anna-Hernandez and van Holland found the same association in pregnant women and in construction workers, respectively.52,53 All these studies support the idea that HCC reflects stem cell activity in the adrenal glands. However, these studies provide only indirect evidence for adrenal sources of hair cortisol. Therefore, further research is needed to confirm the sources of HCC.
We found no significant correlation between HAMD scores and HCC in patients with depression either at pre-treatment or post-treatment, a finding inconsistent with previous studies using non-hair samples.54,55 Two factors may explain this inconsistency. First, the retrospective nature of hair analysis allows the measurement of average cortisol levels over a specific period of time, which may differ from cortisol concentrations in serum or urine samples. 31 Second, the effects of antidepressants should be considered in interpreting results. This study had several limitations. First, the sample was small, which may reduce the reliability of the results. Second, we found a significantly high HCC after 4 weeks antidepressant treatment, which may indicate that the antidepressant treatment did not result in optimum depression control within the 4 weeks. In fact, it can take 2 to 4 weeks (and sometimes 6 weeks) for antidepressants to take effect. 56 Third, antidepressant drugs should be selected based on their different specific mechanisms of action to enhance the clarity of results.
In summary, we found that the HCC of patients with depression increased significantly at post-treatment. HCC analysis could be used to monitor the dynamics of the effects of antidepressants on the HPA axis.
Footnotes
Acknowledgments
The authors thank Professor Dongtao Lin of Sichuan University for copyediting this manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was partly funded by a National Science and Technology Support Plan (2015BAI13B02) and State 863 Projects (2015AA020513).
