Abstract
Objective
To investigate the effects and potential mechanism of action of shikonin (SHK) on the development of ovarian follicles and female germline stem cells (FGSCs).
Methods
Female Kunming adult mice were administered SHK (0, 20 and 50 mg/kg) by oral gavage. Cultures of FGSCs were treated with SHK 32 μmol/l for 24 h. The ovarian index in mouse ovaries was calculated. Numbers of primordial, primary and atretic follicles were counted. Germline stem cell markers and apoptosis were examined. Levels of glutathione (GSH), superoxide dismutase (SOD) and reactive oxygen species (ROS) were measured.
Results
Both doses of SHK significantly decreased the ovarian index, the numbers of primordial follicles, primary follicles and antral follicles in mice. SHK significantly increased the numbers of atretic follicles and atretic corpora lutea. SHK promoted apoptosis
Conclusions
These current results suggested that follicular development and FGSCs were suppressed by SHK through the induction of apoptosis and oxidative stress might be involved in this pathological process.
Keywords
Introduction
Shikonin (SHK) is a naphthoquinone compound extracted from the dried roots of Lithospermum erythrorhizon, a commonly used Chinese herbal medicine.
1
SHK and its derivatives have various pharmacological effects, such as immune surveillance,
2
oxidative equilibrium regulation
3
,
4
and anti-inflammatory actions.
5
Recently, SHK was shown to have robust anti-tumorigenic effects on colon cancer,
6
lung cancer
7
and other tumours
In contrast, several studies reported some deleterious effects of SHK on the reproductive system.11–15 For example, in male mice, SHK damaged the spermatogenic epithelium, suppressed the process of spermatogenesis and impaired reproductive capacity through oestrogen receptor 1 (alpha) (
Female germline stem cells (FGSCs) maintain the female reproductive functions and FGSCs have been extensively studied during the stages of oogenesis. 16 The ability to isolate FGSCs and study their proliferation enables the understanding of neo-oogenesis and ovarian recession. 17 This current study explored whether SHK controls the proliferation, differentiation and apoptosis of FGSCs. The imbalance between FGSCs and the stem-cell niche caused by oxidative stress contributes to ovarian recession. 18 In addition, oxidative stress impairs the stem-cell niche and induces follicular atresia. 16 , 17 The bioactivity of superoxide dismutase (SOD) and glutathione (GSH) counteracts the oxidative cytotoxicity induced by reactive oxygen species (ROS).16–19
To investigate the possible mechanism underlying the effects of SHK on ovarian follicular development and FGSCs, this current study measured the GSH concentration and the activity of SOD in the peripheral blood of mice. The levels of ROS in FGSCs treated with SHK were also measured.
Materials and methods
Animals
This study was conducted in the Key Laboratory of Reproductive Physiology and Pathology of Jiangxi Province, Nanchang University, Nanchang, Jiangxi Province, China and conformed to the National Research Council Guide for Care and Use of Laboratory Animals. 20 All animal procedures followed the Institutional Animal Care and Use Committee rules and regulations. 20 All procedures involving mice were approved by the Animal and Ethical Committee of Nanchang University, Nanchang, Jiangxi Province, China.
Seven-week-old Kunming female mice were purchased from Jiangxi University of Traditional Chinese Medicine, Nanchang, Jiangxi Province, China and were housed under a 12-h light/12-h dark circle at 23°C with free access to food and water until they reached 2 months of age. Mice were randomly divided into three groups, five in each group. SHK (Meilunbio, Shanghai, China) powder was dissolved in a 10% sodium carboxymethylcellulose (CMC-Na) solution. The three groups were treated as follows: (i) the control group was administered 10% CMC-Na via oral gavage and the administered volume was lower than 2% of the total body weight; (ii) the low-dose group was administered 20 mg/kg SHK in CMC-Na via oral gavage; (iii) the high-dose group was administered 50 mg/kg SHK in CMC-Na via oral gavage. Two doses were given with an interval of 3 days between the two doses. Tissues and blood samples were collected 7 days after the last oral gavage.
Culture of FGSCs in vitro
The isolation, culture and proliferation of FGSCs were performed as described previously. 20 , 21 The ingredients of the tissue culture medium are listed in Table 1. Sandos inbred mice embryo-derived thioguanine-resistant, ouabain-resistant cells were used as the feeder cell line.
The ingredients used in the tissue culture medium for the culture of female germline stem cells.

Ovarian weight and body weight
Ovaries were dissected under aseptic conditions and the ovaries were rinsed with 0.01 M phosphate-buffered saline (PBS, pH 7.4; Thermo Fisher, Shanghai, China). The organ coefficient was measured after complete drying of the ovaries as described previously.22,23 Weight was measured using a precision electronic balance (Shimadzu, Fujian, Japan). The following formula was used to calculate the ovarian index: ovarian index (%) = ovarian weight (g)/mouse body weight (g) × 100.
Ovarian follicle quantification
Mice were sacrificed by cervical dislocation after blood collection. Ovaries were fixed in 4% paraformaldehyde overnight and embedded in paraffin. 24 Tissues were sectioned into 5-μm sections and stained with haematoxylin and eosin. Images were obtained using an optical microscope (Olympus BX; Olympus, Tokyo, Japan). Quantification was performed as previously described. 25 , 26 The criteria for follicular classification for each structure was as follows: (i) primordial follicles were located in the superficial cortex, small in size and large in number. There was a layer of flat follicle cells around the oocyte; (ii) the primary follicle develops from the primordial follicle, which is larger than the primordial follicle and gradually penetrates into the deep cortex. The follicle cells surrounding the oocyte become multiple layers; (iii) the diameter of antral follicles was relatively large in clinical practice and the diameter can reach about 0.2 mm. Liquid is formed between granular cells to form a cavity; (iv) an atretic follicle is a type of degenerative follicles. The detachment of granulosa cells from the follicular basement membrane or the atrophy of granulosa cells will cause follicular atresia. After the follicle is atretic, fibrous connective tissue replaces the follicle; (v) atretic corpora lutea forms from atretic ovarian follicles and its granular cells undergo degeneration and destruction. The inner capsular cells undergo hypertrophy and change to form a luteal-like structure.
Immunohistochemistry and immunofluorescence
After dewaxing and rehydration, 5-μm thick paraffin sections were used for immunohistochemistry (IHC) and immunofluorescence (IF) according to a previous study. 27 For IF, paraffin sections were stained on a Dako Autostainer (Dako, Carpinteria, CA, USA) with a primary rabbit polyclonal antibody and an anti-rabbit secondary antibody (details below). For IHC, paraffin sections were stained on a Ventana Benchmark XT autostainer (Ventana Medical Systems, Tucson, AZ, USA). Antigen retrieval was performed with Ventana Cell Conditioner 1 (Ventana Medical Systems) for 30 min. Sections were then incubated with the primary rabbit polyclonal antibody at 37°C for 32 min. The primary antibodies used in this study were as follows: MVH (ab27591; Abcam, Shanghai, China) at a 1:100 dilution and OCT4 (11263-1-AP; Proteintech, Rosemont, IL, USA) at a 1:200 dilution. The proprietary Ventana anti-rabbit secondary antibody was then applied, followed by the diaminobenzidine solution (CAS91952; MERDK, Shanghai, China) used for the staining and chromogenic reactions. All of the images were examined under a NIKON Eclipse 80i microscope (Beijing Rongxing Guangheng Technology, Beijing, China).
TUNEL assay
The terminal deoxynucleotidyl transferase-mediated dUTP nick-end labelling (TUNEL) assay was performed using a TransDetect In Situ Fluorescein TUNEL Cell Apoptosis Detection Kit (TransGen Biotech, Beijing, China). 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (DAPI) was used to counterstain the tissue sections and FGSCs. After washing three-times with 0.01 M PBS (pH 7.4), images were obtained using a fluorescence microscope (XSP-BM-13C; Olympus).
Quantitative polymerase chain reaction
Total RNA was extracted from ovaries using TRIzol® reagent (Thermo Fisher Scientific, Rockford, IL, USA) according to the manufacturer’s instructions. 28 For the ovaries from the control group, 2–3 ovaries were required to extract sufficient total RNA. For the ovaries from the two experimental groups, 4–6 ovaries were required to extract enough RNA. Approximately 2 µg RNA was used to synthesize cDNA using a PrimeScript® RT reagent Kit (TaKaRa Bio, Shiga, Japan). The amplification was performed on an ABI7000 PCR instrument (Applied Biosystems, Foster City, CA, USA). The cycling programme involved preliminary denaturation at 50°C for 2 min and at 95°C for 2 min, followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 60 s, followed by a final cooling step at 4°C. 29 The primers required for PCR are listed in Table 2. The reaction was carried out in a 20-µl volume containing 2 µl cDNA and other materials according to the manufacturer’s instructions. β-actin was used as the internal reference gene. This process was repeated independently at least in triplicate to achieve good reproducibility. The relative levels of MVH and OCT4 mRNA were compared with those of β-actin and calculated using the 2–ΔΔCT method. The CT value used for these calculations was the mean of the triplicate for each reaction.
The sequences of primers used for quantitative real-time polymerase chain reaction.

Cell proliferation assay
The viability of FGSCs
Measurement of GSH and SOD
The concentration of GSH in the peripheral blood of mice was measured using a GSH enzyme-linked immunosorbent assay (ELISA) (Elabscience, WuHan, China). The activity of SOD was detected using a Total Superoxide Dismutase (SOD) Colorimetric Assay Kit (WST-1 method; Elabscience) according to the manufacturer’s instructions. The minimum detectable concentrations were 0.94 µg/ml for GSH and 0.2 U/ml for SOD. Intra- and interassay coefficients of variation for all ELISAs were <6% and <5%, respectively.
ROS detection in cells
The levels of ROS were measured using a ROS Assay Kit according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). 2,7-Dichlorodi-hydrofluorescein diacetate was added to the culture medium at 10 μM/l. The FGSCs were incubated at 37°C for 1 h and were then digested using trypsin. Cells were collected after centrifugation at 1000
Statistical analyses
All statistical analyses were performed using GraphPad Prism 5 (Graphpad Software Inc., San Diego, CA, USA). All data were obtained from at least three separate experiments. Comparisons between experimental groups were analysed using unpaired Student’s
Results
After weighing each mouse and its ovaries in the control, 20 mg/kg and 50 mg/kg SHK treated groups, the ovarian indices of each group were calculated. The ovary-to-body weight ratio (ovarian index) is a straightforward method that is widely used to describe ovarian functions and the occurrence of ovarian atrophy.
23
SHK significantly decreased ovarian indices in both the 20 mg/kg (

The effects of shikonin (SHK) on ovarian structure and phenotype (
The levels of apoptosis in ovarian tissue sections were measured using TUNEL staining. The results showed that apoptosis occurred in the control group (Figure 2a). SHK administration significantly increased apoptosis at both concentrations compared with the control group (

Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labelling apoptosis assay of shikonin (SHK)-treated mouse ovaries. The cell nuclei are stained with 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (blue fluorescence) and the green fluorescent signals indicate apoptosis: (a) the intensity of apoptosis signals in the control, 20 mg/kg and 50 mg/kg SHK treated groups. Scale bar 100 μm; (b) The corresponding apoptotic rate in the control, 20 mg/kg and 50 mg/kg SHK treated groups. Data presented as mean ± SD. ##
Previous studies have indicated that FGSCs are mainly located in the ovarian surface epithelium (OSE).
29
,
30
MVH is specifically expressed in all germ cell lineages and serves as a specific marker of reproductive cells.
31
,
32
OCT4 is specifically expressed in pluripotent stem cells.
33
,
34
As a result, MVH and OCT4 were selected as the germline stem cell markers in the culture of FGSCs.
29
There was a concentration-dependent decrease in MVH and OCT4 mRNA levels as measured by PCR (Figure 3a). Significantly lower levels of MVH and OCT4 mRNA occurred in the 50 mg/kg SHK treated group compared with the control (
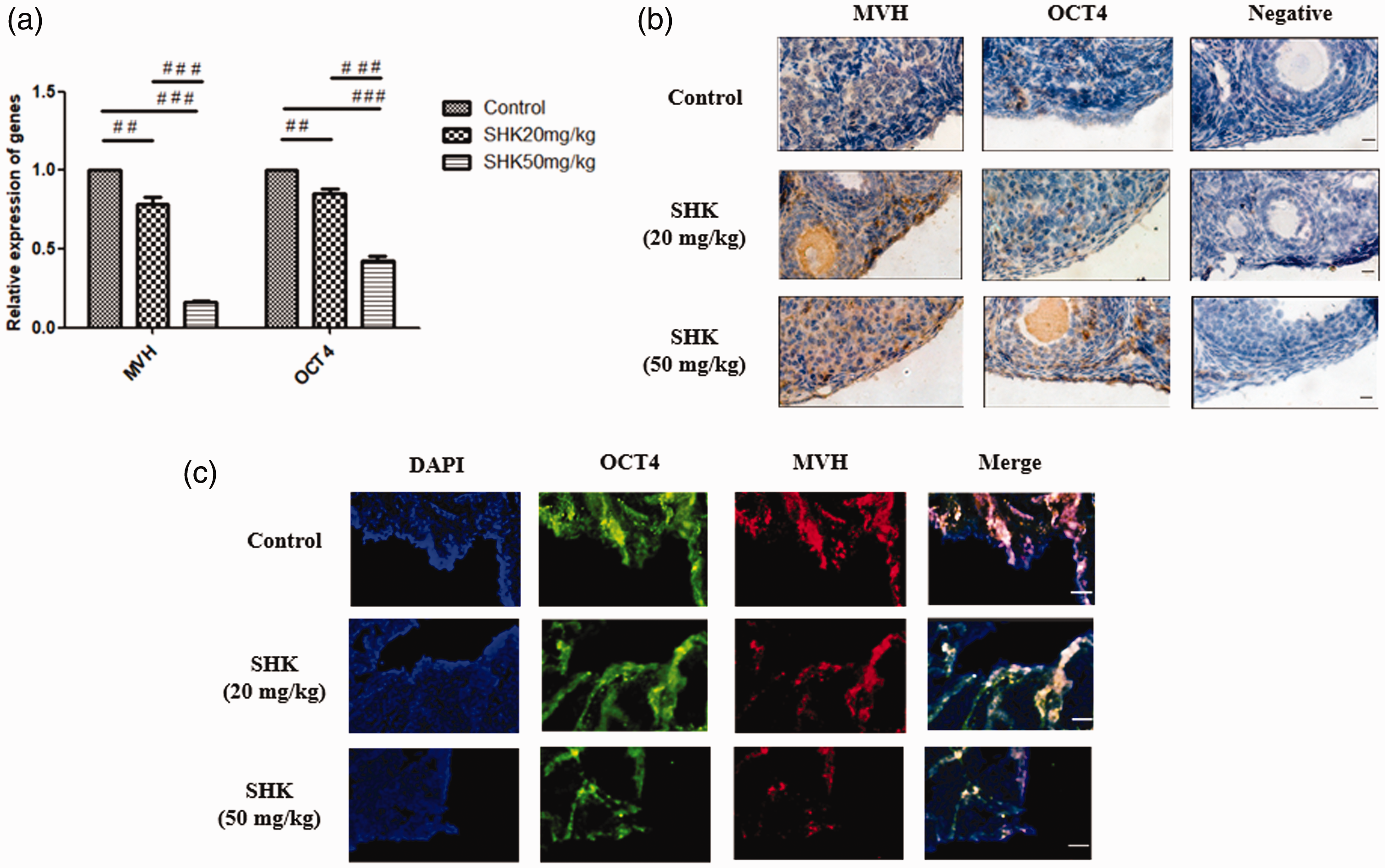
The levels of mouse vasa homolog (MVH) and octamer-blinding protein-4 (OCT4) in female germline stem cells: (a) quantitative real-time polymerase chain reaction analysis in the control, 20 mg/kg and 50 mg/kg shikonin (SHK) treated groups. Data presented as mean ± SD. ##
The effects of SHK (0, 4, 8, 16, 32 and 64 μmol/l SHK) at 0, 24, 48 and 72 h on the viability of FGSCs was measured using a CCK kit. The 32 μmol/l dose for 24 h significantly affected cell viability compared with the control group (

The effects of shikonin (SHK) treatment for 0–72 h at various concentrations on the viability of female germline stem cells

Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labelling apoptosis assay of shikonin (SHK)-treated female germline stem cells
Peripheral blood was collected from mice in order to measure the levels of GSH and SOD. The 50 mg/kg SHK treated group had significantly lower GSH content and suppressed SOD activity compared with the control and 20 mg/kg SHK treated groups (

The changes in glutathione (GSH) and superoxide dismutase (SOD) levels in the peripheral blood of shikonin (SHK)-treated mice: (A) concentration of GSH in the control, 20 mg/kg and 50 mg/kg SHK treated groups. Data presented as mean ± SD. ##
The accumulation of ROS is the leading cause of oxidate stress.
35
The ROS concertation in FGSCs were measured after SHK treatment at 32 μmol/l dose for 24 h and compared with ROS concentration at baseline in the control group (Figure 7). Treatment with SHK 32 μmol/l for 24 h caused a significant increase ROS levels in FGSCs compared with the control group (

The concentration of reactive oxygen species (ROS) in shikonin (SHK)-treated female germline stem cells
Discussion
Shikonin is a commonly used anti-tumorigenic medicine because of its apoptosis enhancing effects. 6 SHK is a novel immunosuppressant that is effective in a variety of diseases and clinical transplantation. 36 , 37 Previous reports using histological approaches demonstrated that SHK negatively regulated metabolic rates, hormone levels and reproductive functions.11–15 However, the effects of SHK on ovarian follicular development and FGSCs and the underlying molecular mechanisms remain largely unclear.
The present study investigated the effects of SHK in oocyte development. These current results showed that SHK exhibited a dose-dependent effect on decreasing ovarian index, primordial follicles, primary follicles and antral follicles. SHK upregulated the numbers of atretic follicles and atretic corpora lutea. TUNEL methodology demonstrated that 50 mg/kg SHK significantly induced apoptosis, contributing to the blunted ovarian development and functional decline. In addition, SHK interrupts the endocrine system including the pituitary gland–ovary axis by interfering with the bioactivities of related hormones, such as follicle stimulating hormone, which subsequentially inhibits the maturation and ovulation of follicles in all grades. 14 This current study has identified a dose of SHK (50 mg/kg) that can effectively inhibit ovarian follicular development and reproductive functions in mice. This current study is the first comprehensive study on the biosafety of SHK during the development of ovarian follicles, providing an insight into drug safety in future female applications.
As a consequence of the apparent scarcity of FGSCs in ovaries,
38
it is difficult to access their response to drugs including SHK under regular laboratory conditions. In order to understand how SHK impacts on FGSCs and to provide future FGSC-related therapeutic approaches to restore ovarian function, this current study used the germline stem cell markers MVH and OCT4;28,29 which were suppressed in the presence of SHK. The decline of MVH and OCT4 may suggest that FGSCs were disordered
In order to study the mechanisms of SHK on ovaries and FGSCs, this current study measured the levels of molecules related to oxidative stress. The concentration of GSH and the activity of SOD were decreased by SHK in a dose-dependent manner
In conclusion, this present study uncovered a novel mechanism by which SHK might induce infertility during SHK anti-tumorigenic application, which should be considered when using this drug in the clinic.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211029461 - Supplemental material for Effects of shikonin on the development of ovarian follicles and female germline stem cells
Supplemental material, sj-pdf-1-imr-10.1177_03000605211029461 for Effects of shikonin on the development of ovarian follicles and female germline stem cells by Shu-Xin Ma, Li-Bo Tang, Zhi-Hang Chen, Min-Li Wei, Zi-Juan Tang, Yue-Hui Zheng, Guo Zong and Jia Li in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by grants from the Natural Science Foundation of Jiangxi, China (grant numbers: 2018ACB20018, 20192BAB215009, 20202ACB216003), the National Natural Science Foundation of China (grant number: 31460307,81671455,81771583) and the young teachers research and training foundation of Nanchang university medical department (grant number: PY201814).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
