Abstract
Objective
To examine sonication results in presumed aseptic conversion total hip arthroplasty (THA) after hip fracture fixation and to evaluate its implications on the treatment outcome.
Methods
This retrospective cohort study reviewed the data from presumed aseptic patients that underwent conversion of prior internal fixation of proximal femoral fractures to THA between 2012 and 2018. Microbiological analysis was performed using sonication of osteosynthesis material and tissue samples. Treatment outcome including the occurrence of periprosthetic joint infection (PJI) was recorded.
Results
A total of 32 patients were included in the study. Of these, five patients (15.6%) had positive intraoperative cultures. The mean follow-up following conversion THA was 43.0 months (range, 19.0–91.5 months). Sonication was positive in three patients (9.4%), all of whom were deemed contaminated and did not develop PJI. Tissue cultures were positive in two patients (6.3%). One patient with Enterococcus faecalis received antibiotic treatment and did not develop PJI. Another patient with growth of Cutibacterium acnes that was initially classified as a contaminant later developed acute PJI caused by the same pathogen. Overall, PJI occurred in two patients (6.3%) after conversion THA.
Conclusion
Sonication of internal fixation devices did not add diagnostic value in clinically aseptic conversion THA. Further studies are needed to better understand the relevance of unexpected positive cultures, and to develop diagnostic criteria for the management of these patients.
Keywords
Introduction
Hip fractures constitute an important medical challenge and the incidence is expected to rise even further due to the aging of populations. 1 Contemporary internal fixation techniques allow for most fractures to heal uneventfully. However, fixation failure occurs in 5–41% of patients and conversions to total hip arthroplasty (THA) for painful implants, osteonecrosis, non-union or post-traumatic osteoarthritis (OA) are increasingly common.2–4
Several studies have shown higher complication rates for conversion THA when compared with primary THA, including an increased risk of infection.5–7 Periprosthetic joint infections (PJI) are a particularly feared complication in THA, as they result in increased morbidity to affected patients and a considerable economic burden to the healthcare system. 8 As yet, it remains unclear whether the increased risk for PJI after conversion THA only applies to patients with signs of infection or also to clinically unremarkable patients. Patients with elevated inflammatory markers or clinical suspicion of infection should prompt further evaluation with preoperative diagnostic hip aspiration including white blood cell count, differential and culture prior to conversion THA. 7 In patients without suspicion of infection, conversion THA is typically performed in a standard single-stage procedure without a thorough preoperative work-up. To date, there are no guidelines or consensus on the diagnostic criteria and treatment algorithm of patients undergoing conversion THA.
Recently, a high percentage of microbial colonization of internal fixation implants has been demonstrated even in clinically unremarkable patients.9,10 Thus the possibility of occult implant-associated infections may be considered for any patient with prior internal fixation, even in the absence of clinical or laboratory signs of infection. Besides that, a paucity of literature exists for diagnosing infection in the setting of a conversion THA. Recent studies focused on serological screening and intraoperative tissue sampling,7,11 but at this time no study was investigating on the utility of implant sonication for diagnostic workup of patients undergoing THA after prior internal fixation of the hip.
The main objective of this study was to present the results of sonication of osteosynthesis material in patients undergoing conversion of prior hip fracture fixation to THA and to describe the natural course of treatment for a consecutive series of patients with unexpected positive intraoperative cultures during conversion THA.
Patients and methods
Study design and patient selection
This retrospective cohort study reviewed a prospectively maintained institutional database at the Centre for Musculoskeletal Surgery, Charité University Medicine Berlin, Berlin, Germany for the period between September 2012 and December 2018 to identify all consecutive patients that underwent conversion of prior internal fixation of the proximal femur to THA and whose osteosynthesis material had been sent for sonication. Exclusion criteria were as follows: (i) patient age < 18 years; (ii) administration of antibiotics within 4 weeks before implant removal; (iii) patients with a previously known infection prior to conversion THA; (iv) patients with acetabular osteosynthesis material. Medical records were reviewed for all details on demographics, comorbidities, indications for conversion THA, intraoperative samples and postoperative follow-up. The STROBE guidelines were followed for the preparation of the manuscript and all patient details were de-identified. 12
The study was approved by the institutional review board of Charité University Medicine Berlin, Berlin, Germany (Ethikkommission der Charité – Universitätsmedizin Berlin, Germany; no. EA2/026/19) on 1 March 2019. All patients provided written informed consent to treatment.
Microbiological methods
The removed osteosynthesis material was sent for sonication in sterile reusable polypropylene containers according to a previously published protocol utilizing a BactoSonic® 14.2 sonication unit (Bandelin Electronic GmbH, Berlin, Germany). 13 Additionally, synovial fluid (if present) was aspirated with a sterile syringe before incision of the pseudocapsule and inoculated in blood culture paediatric bottles (BacTec PedsPlus/F; Beckton Dickinson, Shannon, Ireland). Tissue samples were placed in sterile containers without medium or saline. All microbiological samples were processed according to a standard microbiological protocol in both aerobic and anaerobic cultures incubated at 35 °C for 14 days. 14 Identification and susceptibility testing of isolated microorganisms was performed using an automatic bacteriological analyser VITEK® 2 (bioMerieux, Marcy L'Etoile, France).
Definition of infection
As guidelines for infected fixation devices such as PJI are lacking,15,16 infection was defined in line with the institutional criteria for PJI, 16 which have been used in several clinical outcome studies for PJI.17–20 Accordingly, infection was diagnosed when at least one of the following criteria was present: confirmatory microbial growth in synovial fluid, tissue cultures (≥ 1 specimen in highly virulent organisms or ≥ 2 specimens in low virulent pathogens); or sonication fluid cultures of the retrieved fixation implant (> 50 colony-forming units [CFU]/ml sonication fluid). 21 Bacterial growth of < 50 CFU/ml in sonication fluid cultures and single specimen growth of a low-virulent pathogen in tissue cultures were deemed as contamination.
Surgical and antimicrobial treatment
All patients had undergone a standardized preoperative diagnostic work-up including physical examination, plain radiographs and laboratory tests, including serum C-reactive protein (CRP). The removal of osteosynthesis material was either performed in a single-stage procedure at the same time as the THA or preceding the THA as part of a two-stage procedure depending on the surgeon’s discretion. All fixation devices were removed completely and sent for sonication. Additionally, synovial fluid was aspirated (if present) and tissue samples were taken near the implant and/or from the synovium. Conversion THA was done through an anterolateral approach using cementless implants or cemented prostheses.
All patients received antibiotic prophylaxis with a single intravenous injection of 2 g cefazolin. Patients with positive intraoperative samples were discussed through an interdisciplinary team of orthopaedic surgeons, infectious diseases physicians and microbiologists. Patients that were classified as having an infection received targeted oral antibiotic treatment including a biofilm-active agent (i.e. rifampin) for 12 weeks following conversion THA.
Treatment outcome
Patients were monitored for the occurrence of PJI during routine follow-up examinations. PJI after conversion THA was defined according to institutional criteria, 16 obtained through a joint aspirate or revision surgery during the follow-up period.
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics are reported as n of patients (percentage) or mean (range) or mean ± SD, as appropriate. Continuous variables were compared using Student’s t-test and categorical variables using Pearson's χ2-test. A 2-sided P-value < 0.05 was considered significant.
Results
A total of 50 patients received removal of osteosynthesis material of the proximal femur during conversion THA. Of these, 32 patients (17 male, 15 female) matched the inclusion criteria. The indication for THA was fixation failure (including non-union) in 17 patients (53.1%) and post-traumatic OA in 15 patients (46.9%). The mean duration between hip fracture fixation and conversion THA was 35.7 months (range, 1–318 months). Conversion THA was done through an anterolateral approach using cementless implants in 29 patients (90.6%) (Figure 1) and cemented prostheses in three patients (9.4%). A total of 29 patients (90.6%) underwent a single-stage conversion and three patients (9.4%) a two-stage THA. For the latter, the mean duration between hardware removal and THA was 6.8 weeks (4.4–9.0 weeks). The removed osteosynthesis devices included intramedullary nails in 14 patients (43.8%), dynamic hip screws in 11 patients (34.4%) and cannulated screws in seven patients (21.9%). The mean follow-up following conversion THA was 43.0 months (range, 19.0–91.5 months).

Representative pre- (a) and postoperative (b) radiographs of the conversion of prior failed hip fracture fixation to cementless total hip arthroplasty.
The baseline characteristics including a comparison between patients that received the conversion THA due to fixation failure and those for post-traumatic OA are shown in Table 1. Patients with fixation failure had a significantly shorter interval between osteosynthesis and conversion THA compared with those with post-traumatic OA (P = 0.015). Additionally, there were higher preoperative CRP values in patients with osteosynthesis failure compared with those with post-traumatic OA (11.2 versus 2.6 mg/l, respectively), but the difference did not reach statistical significance. The other baseline characteristics did not differ significantly between the two groups.
Demographic and clinical characteristics of patients (n = 32) that underwent conversion of prior hip fracture fixation to total hip arthroplasty (THA) stratified according to whether they underwent conversion due to fixation failure or post-traumatic osteoarthritis.
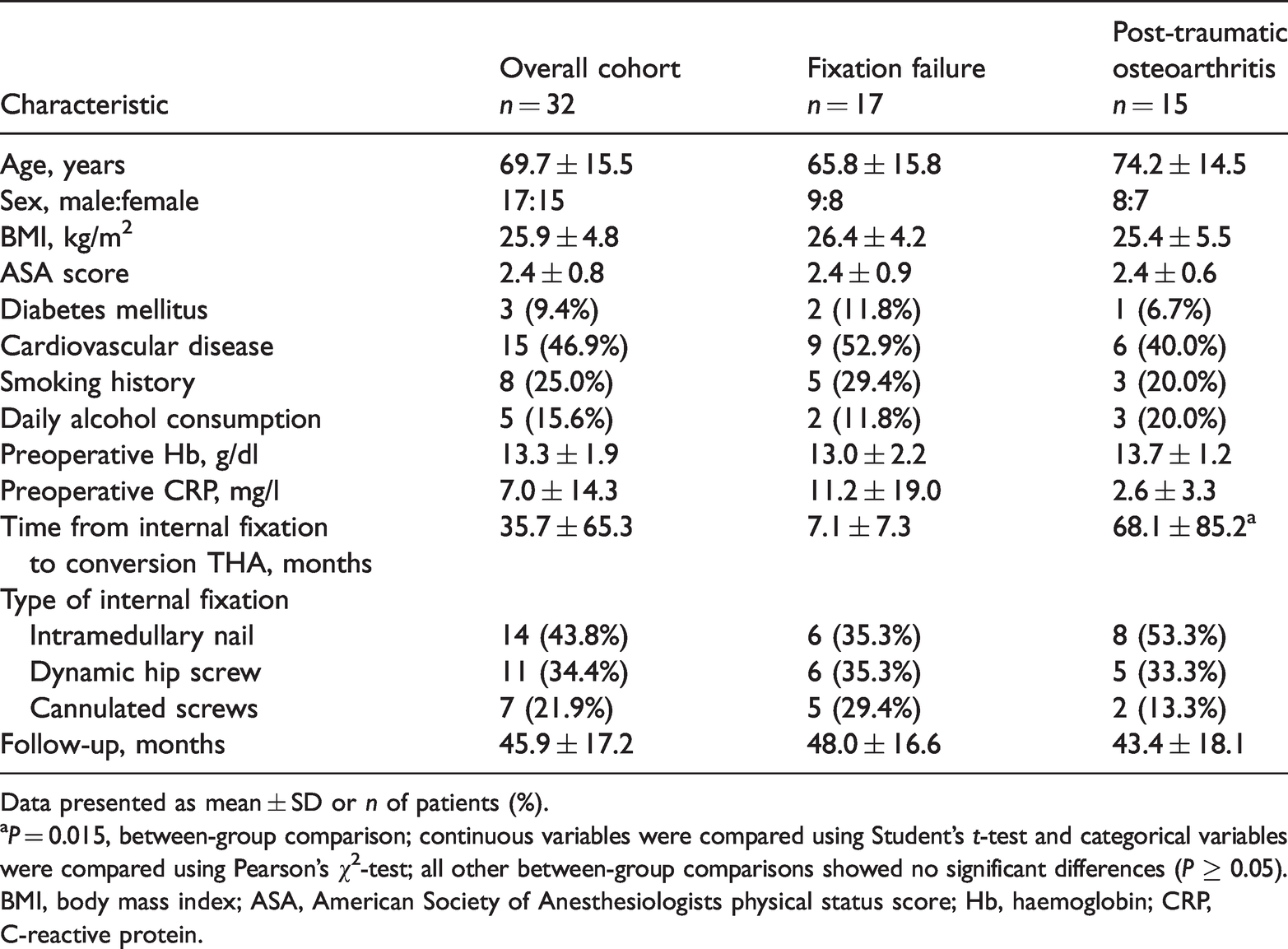
Data presented as mean ± SD or n of patients (%).
aP = 0.015, between-group comparison; continuous variables were compared using Student’s t-test and categorical variables were compared using Pearson's χ2-test; all other between-group comparisons showed no significant differences (P ≥ 0.05).
BMI, body mass index; ASA, American Society of Anesthesiologists physical status score; Hb, haemoglobin; CRP, C-reactive protein.
Overall, five of 32 patients (15.6%) had positive intraoperative cultures at the time of conversion THA, whereas one patient (3.1%) was classified as having an infection and received outpatient antibiotic therapy. Three patients had growth of a low-virulent pathogen in < 50 CFU/ml sonication fluid and were classified as contamination. Two patients had positive tissue cultures, of which one patient had multiple samples growing the same organism (Enterococcus faecalis). This patient received targeted antibiotic treatment including rifampin for 12 weeks postoperatively and did not develop PJI up to the latest follow-up. The other patient with a positive tissue culture had a single specimen growth of Cutibacterium acnes, which was classified as contamination. This patient required revision surgery due to persistent wound drainage. During irrigation and debridement (I&D) with head and liner exchange, implants were sent for sonication cultures, which showed growth again of C. acnes. After targeted antibiotic treatment including rifampin for 12 weeks, PJI was successfully controlled up to the latest follow-up. Overall, two of 32 patients (6.3%) undergoing conversion THA were diagnosed with PJI after a mean follow-up of 37 months (range, 19–74 months). In addition to the PJI caused by C. acnes, there was one patient with negative samples at the time of conversion THA that required I&D with head and liner exchange 1 week postoperatively due to local signs of inflammation and persistent wound drainage. During revision surgery, the synovial fluid culture showed growth of Staphylococcus aureus, whereas tissue and sonication fluid cultures remained negative. After targeted antibiotic treatment for a total duration of 12 weeks, PJI was controlled up to the last follow-up. Detailed data of all five patients with positive intraoperative cultures are shown in Table 2.
Demographic and clinical characteristics of patients (n = 5) with positive intraoperative cultures that underwent conversion of prior hip fracture fixation to total hip arthroplasty (THA).

BMI, body mass index; CRP, C-reactive protein; Hb, haemoglobin; PJI, periprosthetic joint infection; F, female; M, male.
Both patients that developed PJI underwent conversion THA due to early fixation failure (2.2 and 4.5 months after internal fixation). BMI was significantly higher (33.3 versus 25.4 kg/m2; P = 0.023) and preoperative haemoglobin values were lower (10.0 versus 13.5 g/dl, P = 0.007) in patients with PJI compared with those without PJI (Table 3).
Demographic and clinical characteristics of patients that did (n = 2) or did not (n = 30) develop periprosthetic joint infection (PJI) after conversion of prior hip fracture fixation to total hip arthroplasty (THA).
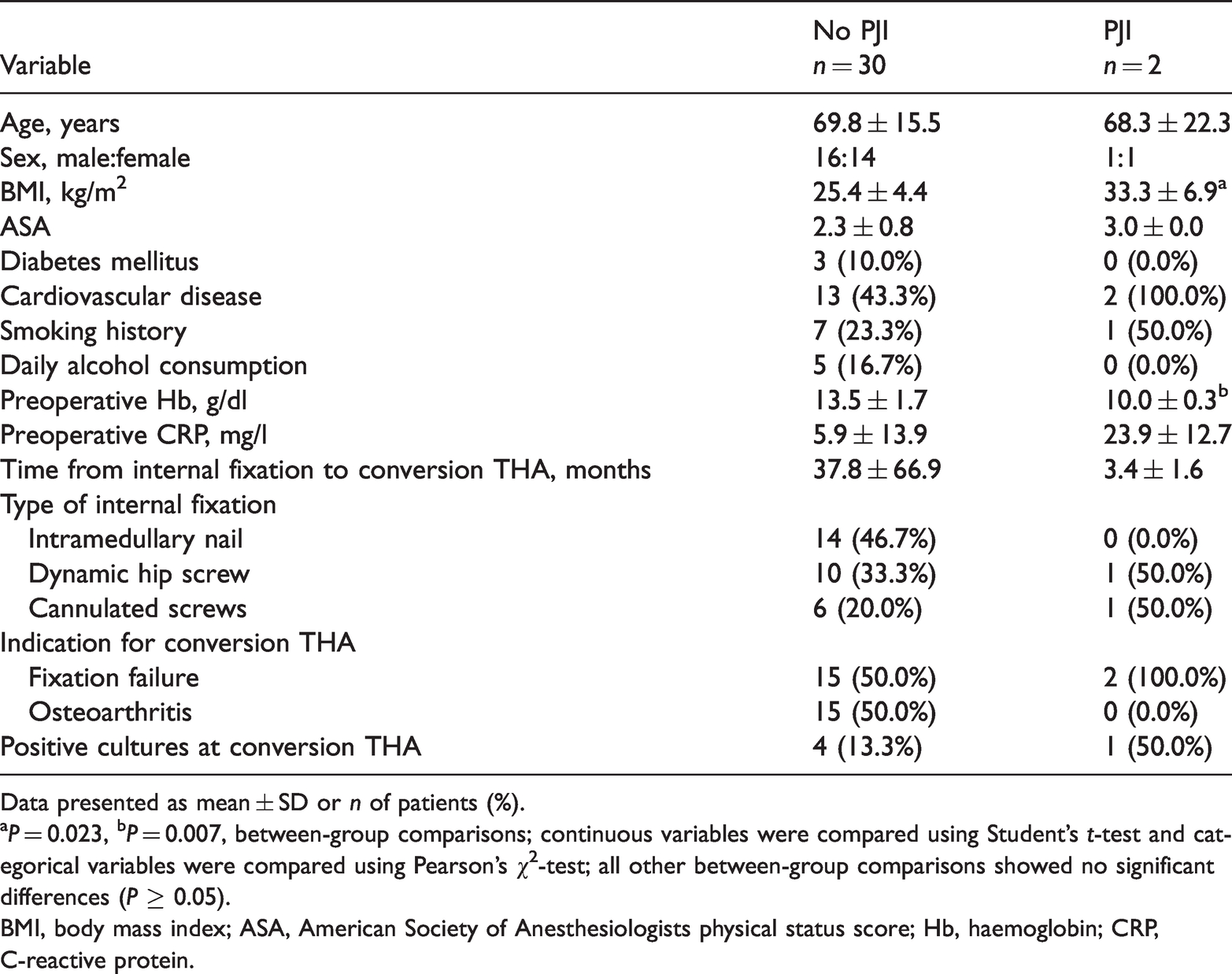
Data presented as mean ± SD or n of patients (%).
aP = 0.023, bP = 0.007, between-group comparisons; continuous variables were compared using Student’s t-test and categorical variables were compared using Pearson's χ2-test; all other between-group comparisons showed no significant differences (P ≥ 0.05).
BMI, body mass index; ASA, American Society of Anesthesiologists physical status score; Hb, haemoglobin; CRP, C-reactive protein.
Discussion
Conversion THA following prior internal fixation of the hip has been associated with an increased risk of PJI.5,7 There are currently no guidelines or criteria regarding the diagnostic workup of patients undergoing conversion THA. Given the complexity of conversion THA, there is suspicion for occult infections similar to revision THA, especially given the number of studies that have shown high rates of microbial colonization of osteosynthesis material even in clinically unremarkable patients.9,10,22 To the best of our knowledge, there is no information on the utility of implant sonication for diagnostic workup of patients undergoing THA after prior internal fixation of the hip. Therefore, the purpose of this current study was to report the results of implant sonication of the proximal femur and its implications on the clinical course of patients undergoing conversion THA.
In the present study, none of the patients showed confirmatory bacterial growth in the sonication fluid. Overall, only one patient was judged to have subclinical infection, with growth of E. faecalis in three tissue samples. This patient received a targeted antibiotic therapy for a total duration of 12 weeks and did not show any signs of PJI after a follow-up of 36 months.
The rate of subclinical infection was 3.1% in this current cohort, while four additional patients had positive samples classified as contamination, resulting in a total rate of unexpected positive intraoperative cultures of 15.6%. Therefore, the colonization rate was much lower than the rate of 56.1% previously described for osteosynthesis material of long bones. 10 However, the authors only stated that the most common regions of fracture were the wrist/forearm without providing any detailed information. 10 It remained unclear if any of the pathogen detections reported in that study were observed around the hip. 10 Another study observed a very high colonization rate for the clavicle, but did not detect a single infection at the fibula. 9 The authors concluded that the colonization rate seems to be highly dependent on the site of the osteosynthesis. 9 This hypothesis is confirmed by the results of this current study, indicating that hip fracture fixation devices are less frequently colonized.
Overall, the rate of PJI was 6.3% in this current cohort. Both patients developed early PJI after a single-stage conversion THA. One patient had a single positive tissue sample of C. acnes, which was regarded as contamination during the conversion THA. During the revision surgery, C. acnes was found again in the sonication fluid cultures of the retrieved head and liner. Hence, single specimen growth of a low virulent pathogen was misinterpreted as contamination during conversion THA. The other patient had negative microbiological results during conversion THA, but later showed S. aureus in the intraoperative joint aspirate of the revision surgery. In this case, PJI might have represented a newly introduced infection.
Implant sonication has been shown to be effective for the dislodgement of adherent bacteria from implants and has a significantly high sensitivity for pathogen detection.23,24 However, the more culture methods are added, the more diagnostic difficulties may arise. As consensus on the definition criteria is lacking for this clinical setting, the interpretation of positive cultures leads to difficulties in choosing the correct diagnosis and treatment for the patient. The distinction between sample contamination and true infection is somewhat unfeasible, especially when searching for low-grade infections. This current study found only microbial growth in less than 50 CFU/ml sonication fluid in three patients, which were all regarded as contamination. None of these patients developed PJI. The challenges of positive sonication cultures in presumed aseptic patients have been described in several previous studies13,22,25 and whether colonized orthopaedic implants become clinically symptomatic at a later time-point or cause infection or implant loosening still remains unclear.
In the current study, the only positive microbiological result during conversion THA that was classified as an infection and treated with outpatient antibiotics was obtained through tissue samples with growth of E. faecalis. The patient had initially received a dynamic hip screw and was converted to THA 10 months later due to cut-out, but did not show any signs of PJI after antibiotic treatment. A subclinical infection with E. faecalis might be possible as clinical presentation of enterococcal infections are similar to that caused by low-virulent pathogens such as C. acnes and coagulase-negative staphylococci. 26 However, other possible explanations might be either contamination or newly introduced pathogens. Therefore, microbiological sampling should always be accompanied by histopathological analysis, and possibly additional markers like synovial fluid leucocyte count or alpha-defensin, allowing for further assessment of possible infection. 23 However, as long as no robust tests to differentiate between true pathogens and contaminants are available, we recommend outpatient antimicrobial treatment for at least 6 weeks in any conversion THA with positive intraoperative cultures. 23
Detection of clinically silent infections prior to conversion THA may significantly affect preoperative planning. It should never be taken for granted that the patient’s pain is being caused solely from fixation failure or OA. A previous study compared the rate of early PJI after conversion THA between patients undergoing single-stage or two-stage surgery, but without taking into account preoperative signs of infection or other patient-specific factors. 27 This study had shown a significantly higher rate of PJI in patients treated with a single-stage surgery and therefore generally recommended a two-stage procedure. 27 In this current study, both patients that developed PJI had elevated CRP values preoperatively and both underwent conversion THA due to early fixation failure. These results are in keeping with a previous study that reported CRP to be a useful screening tool to identify patients that are at an increased risk of PJI after conversion THA. 11 Early fixation failure should also raise suspicion for infection and prompt careful diagnostic measures. In our opinion, a single-stage procedure is still justified for clinically unremarkable patients undergoing conversion THA for post-traumatic OA. Intraoperative cultures should be taken of any patient undergoing conversion THA5,7 in order to identify patients that might benefit from outpatient antibiotic treatment. In any patient with preoperative suspicion of infection, we recommend preoperative joint aspiration. Whether patients might benefit from implant removal with diagnostic biopsy at a first-stage and the THA procedure at a second-stage remains unclear.
The authors acknowledge the limitations of this study. First, it had a retrospective study design and a small patient cohort. Secondly, there are currently no guidelines for diagnosing infection in conversion THA as well as accepted cut-off values for sonication fluid cultures and/or intraoperative cultures, leaving an individual decision if further workup is required at the discretion of the treating interdisciplinary team. Another bias might be the heterogenous population prompting a conversion THA. However, the study tried to minimize this bias by excluding those patients with conversion THA using acetabular osteosynthesis material, which has been reported to be particularly susceptible to infection and osteosynthesis material is often retained. 28
In conclusion, this current study is the first to report sonication cultures in conversion THA and it found no benefit in diagnostic utility for unravelling infections. Patients with elevated CRP and conversion THA due to early fixation failure seem to have an increased risk to develop PJI and should strictly prompt a thorough preoperative work-up. Intraoperative cultures are mandatory in order to prevent a missed or untreated PJI. As long as there is no robust tool and definition criteria to rule out infection in this clinical scenario, any unexpected bacterial growth may have clinical importance and should not be considered merely contamination. Whether patients benefit from further use of fixation device sonication and biofilm-active antibiotics in cases with unexpected positive cultures is uncertain. Future larger-scale studies with standardized treatment protocols are needed to further investigate this issue.
Footnotes
Authors' contributions
Y.P. participated in the design of the work, the acquisition, analysis and interpretation of data, drafted the manuscript, approved the submitted version and agrees both to be personally accountable for the author's own contributions and to ensure that questions related to the accuracy or integrity of any part of the work. M.P. participated in the design of the work, substantially revised the work, approved the submitted version. C.P. also contributed to the critical revision and final approval of the manuscript. S.H. participated in the design of the work, approved the submitted version and agrees both to be personally accountable for the author's own contributions and to ensure that questions related to the accuracy or integrity of any part of the work. C.H. participated in the design of the work, the acquisition and interpretation of data, drafted the manuscript, approved the submitted version and agrees both to be personally accountable for the author's own contributions and to ensure that questions related to the accuracy or integrity of any part of the work.
Acknowledgement
Dr. Palmowski is participant in the BIH-Charité Junior Clinician Scientist Program funded by the Charité -Universitätsmedizin Berlin and the Berlin Institute of Health.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
