Abstract
Müllerian duct cyst is a congenital anomaly that originates from an abnormality in the degradation of the Müllerian system and remnants of the Müllerian duct. Müllerian cyst is rarely reported in male adolescents younger than 18 years of age. In this case report, a 17-year-old male adolescent presented with a scrotal mass of 6 years’ duration. Ultrasonography and computed tomography revealed a left scrotal mass, and the patient underwent open surgical resection of the cyst. Histologically, the cyst consisted of tall columnar cells and was consistent with a Müllerian duct cyst. We discuss the presentation and imaging findings in this case, drawing attention to the diagnosis and treatment.
Introduction
Müllerian duct cyst is an uncommon congenital anomaly in male patients, as a type of urogenital cyst. 1 Anatomically, the cyst is derived from the embryonic Müllerian ducts, extending above the prostate gland medially and posteriorly. Symptoms may occasionally develop; however, early clinical symptoms are not obvious. The clinical signs and symptoms of Müllerian duct cyst depend on the size of the cyst and the presence or absence of infection. The clinical presentation is diverse and includes urinary frequency, urgency, dysuria, urinary obstruction and haematuria. Most patients present with a single cystic lesion, but lesions can be multifocal. A mass effect from the cyst may occur when the cyst is sufficiently large, and if infection is present, the condition is often accompanied by abdominal pain, fever, nausea and scrotal oedema. The diagnosis of Müllerian duct cyst may be difficult before pathology is performed. 2 To our knowledge, no previous studies have reported a Müllerian duct cyst in a male patient younger than 18 years of age. We describe the ultrasonographic appearance of a Müllerian duct cyst in a male adolescent, with the pathological presentation and clinical symptoms and their relationship.
Case report
A 17-year-old male adolescent who presented with a left scrotal mass was admitted to our hospital. He had discovered the scrotal mass 6 years earlier, but did not seek medical treatment. A swollen left scrotum was identified on scrotal examination, and ultrasonography revealed a 4.7-cm × 1.7-cm left scrotal mass filled with cystic fluid. Ultrasonography revealed a scrotal mass with regular morphology, clear borders and thin walls with detailed hyperechoic areas and poor ultrasound penetration of the cyst’s interior (Figure 1a and Figure 1b). A preoperative diagnosis of funicular hydrocele was made, and with the patient's full informed consent, he underwent surgery. Intraoperatively, we identified a 5.0-cm left scrotal cyst with intact cyst wall, and the cyst was fused to the testicular appendix. However, the cyst had no attachments to the left scrotum, and the cyst was successfully removed (Figure 1c and Figure 1d).

Ultrasonographic, gross, and histopathological findings. Ultrasonographic images showing a scrotal mass with clear borders (a, arrow) and thin walls with obvious hyperechoic areas (b); intraoperative view of the cyst (c, arrow) with no attachments to the left side of the scrotum (c, double arrows), and the left scrotal cyst with intact cyst walls (d); histological findings showing a cystic lesion lined with tall columnar cells consistent with a Müllerian duct cyst (e, f).
Intraoperatively, we considered a diagnosis of congenital cyst of the distal urogenital tract. The cyst had a thin wall with no septa and no solid component, on histopathology. The final pathologic diagnosis was a simple serous cyst. However, further histologic examination of the cyst revealed mucin-secreting tall columnar cells, which was consistent with a diagnosis of Müllerian cyst (Figure 1e and Figure 1f). The patient recovered well and was discharged on the second postoperative day. One month postoperatively, the patient had no further complaints.
Discussion
Müllerian cysts may originate from any part of the Müllerian ducts and may be associated with renal agenesis. Clinically, Müllerian cysts in female patients, also known as paraventricular cysts, are extremely rare. Paraovarian simple cysts originate from the embryologic remnants of the urogenital system. 3 The peak age for the clinical incidence of Müllerian duct cyst is from 20 to 40 years, and the reported incidence of asymptomatic cysts in male children is less than 1%. Müllerian duct cysts do not communicate with the urethra and usually occur as an isolated structure filled with cystic fluid. However, simple cysts can undergo multiple transformations, resulting in a neoplastic cyst or serous cyst. 4 Müllerian duct cysts causing ejaculatory duct obstruction have also been associated with subfertility. Persistent Müllerian duct syndrome, as a cause of male pseudohermaphroditism, might indicate a risk of aggressive non-testicular malignancies in normally virilised males, such as in the uterus and fallopian tubes. 5
Clinically, the early symptoms of Müllerian duct cyst are not obvious, and the cysts are often found when the duct cyst has achieved a certain volume that causes pressure on surrounding tissues and organs. The results of most preoperative laboratory tests are generally normal, and a swollen scrotum is identified on scrotal examination. Currently, several imaging techniques provide an extended field of view to related structures for preoperative planning and to rule out other urethral structures. Ultrasonography is valuable for initial cyst localisation, and computed tomography (CT) can determine the size, location and adjacent relationships of the duct cyst; however, the cyst’s characteristics cannot be judged. 6 Magnetic resonance imaging (MRI) is superior for demonstrating multiple cystic lesions of the epididymis and their surrounding related structures, and the signal intensity of the cyst is helpful in the diagnosis. 7 Müllerian duct cysts should be differentiated from ejaculatory duct cysts, and congenital or secondary prostate cysts. Ejaculatory duct cyst lesions point to the seminal caruncle of the prostate. The seminal vesicles on the ipsilateral side are often dilated, the cyst fluid is dark brown and laboratory examinations of the cystic fluid contain sperm, while the Müllerian duct cyst fluid is clear and does not contain sperm. Congenital prostate cysts may appear as single cysts with regular shapes and clear edges, and these are differentiated from Müllerian and ejaculatory duct cysts because of the more specific disease sites for the latter. Secondary cysts, accompanied by benign prostatic hyperplasia, are most often multiple, irregular in shape and small in size. MRI can clearly distinguish the relationships between the scrotal cysts and prostate and other tissues by three-dimensional arbitrary sector imaging. In addition, this technique has the advantages of good soft tissue contrast and some degree of evaluation of liquid properties; therefore, it has better diagnostic value in scrotal lesions.
In men, there is a long list of conditions to consider in the differential diagnosis of deep pelvic cysts, namely Müllerian duct cyst, seminal vesicle cyst, ejaculatory duct cyst, prostatic cyst or urachal cyst. The diagnosis is difficult because it is based on postoperative pathological results. Surgeons should consider excising Müllerian remnants where possible because the risk of malignancy is higher with cysts larger than 4.0 cm in diameter.
Microscopically, the final pathologic diagnosis in our patient was a simple serous cyst. Histologic examination revealed that the cystic lesion was lined with stratified columnar cells, and the epithelium stained positive with mucicarmine, 8 confirming the diagnosis of Müllerian cyst. The presence of malignancy cannot be ignored in these cases because primary adenocarcinoma may arise from a Müllerian cyst. 9 Immunohistochemical analysis showing positive expression for paired box gene 8 and oestrogen and progesterone receptors should always be considered in patients with a Müllerian cyst. 10
We reviewed seven previous cases of Müllerian cyst (Table 1)2,8,10–14 identified in the vaginal wall, scrotum, posterior mediastinum and retroperitoneum; more female patients than male patients were described. The Müllerian cysts varied in size from 1.0 to 9.0 cm in diameter. The imaging results in seven patients provided excellent anatomical views. Although the follow-up data were limited, most patients appeared to recover well, postoperatively. In previous studies, the Müllerian cysts grew large enough to cause symptoms or acute inflammation warranting excision. However, these data were obtained from only seven cases; therefore, literature bias might be present.
Literature review of the seven cases of Müllerian duct cyst published since 2006.
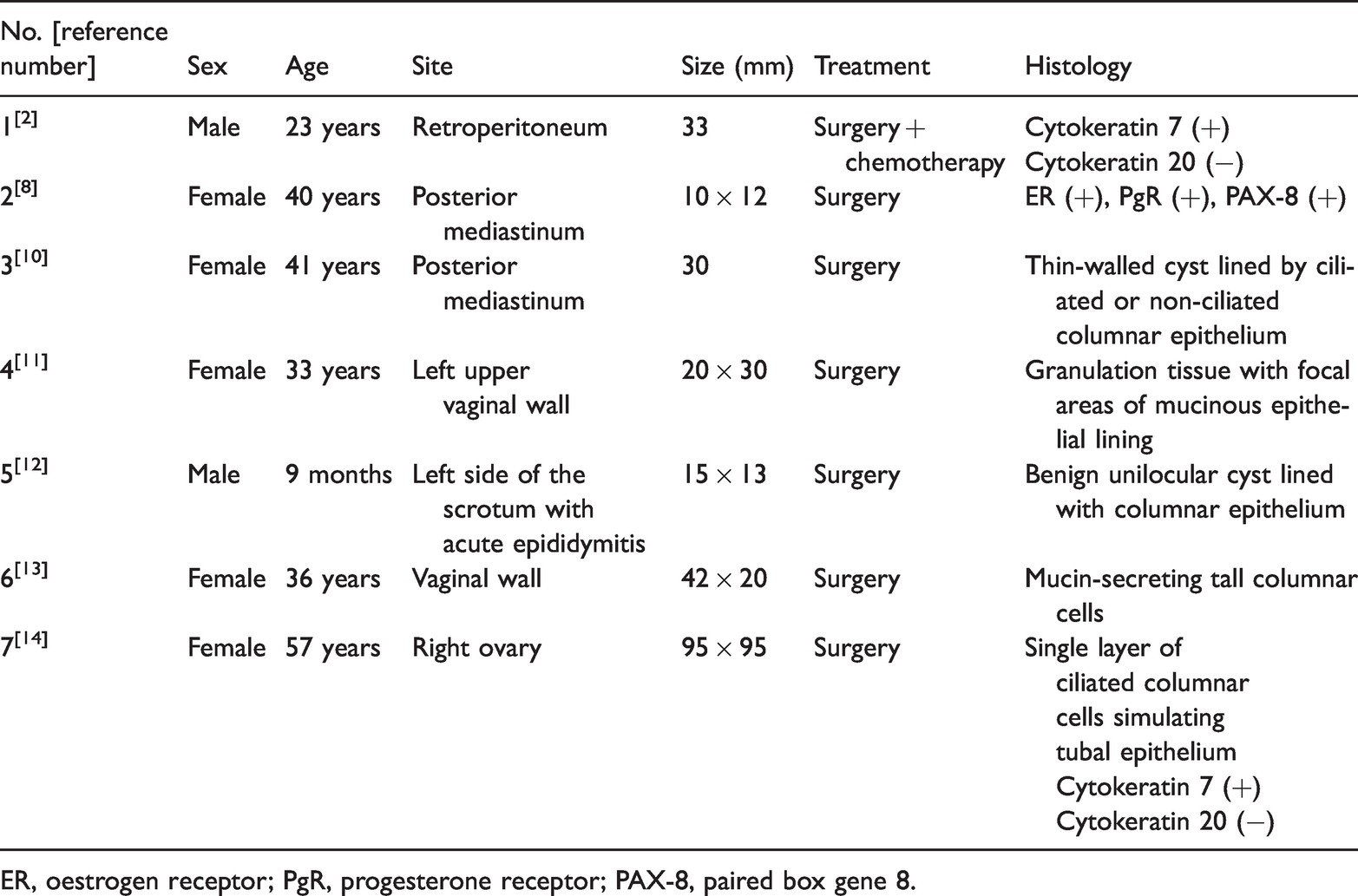
ER, oestrogen receptor; PgR, progesterone receptor; PAX-8, paired box gene 8.
Footnotes
Ethics statement
This study is based on a clinical case; therefore, ethical approval was not required by our institution. Written and verbal informed consent was obtained from the patient for publication of this case report and any accompanying images.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81660263).
