Abstract
Hepatic splenosis is an uncommon condition that occurs following traumatic splenic rupture or splenectomy. The case of a 41-year-old male patient with multiple isolated liver masses indistinguishable from primary and metastatic liver tumours is reported. Following laparotomy, the liver lesions were resected and histopathology confirmed a diagnosis of hepatic splenosis. At an 18-month follow-up examination, no abnormalities in routine blood test, liver function, and liver computed tomography (CT) scanning were observed. After review of the literature, the following diagnostic criteria for hepatic splenosis are proposed: (1) a history of splenic trauma or splenectomy; (2) lesion(s) with a surrounding rim, particularly near the liver capsule identified by CT scanning; (3) findings on superparamagnetic iron oxide-enhanced magnetic resonance imaging or technetium-99m heat-damaged red cell scanning; and (4) histopathological findings (needle biopsy or surgical pathology). The following diagnostic process is also proposed: suspect diagnosis when criteria 1 and 2 are met; make diagnosis when criterion 3 is met; confirm diagnosis when criterion 4 is met. Laparotomy is recommended for either diagnosis or treatment when invasive procedures are necessary.
Introduction
Splenosis is a benign condition commonly resulting from traumatic splenic rupture or splenectomy.1,2 Hepatic splenosis is rare and is usually diagnosed accidentally. 3 Due to its low prevalence, hepatic splenosis is difficult to diagnose by non-invasive methods, particularly when the mass presents as a malignant disease on imaging or the patient has a risk of liver tumour or a history of cancer. Thus, the diagnosis of hepatic splenosis remains elusive and requires further investigation. Here, the case of a patient with hepatic splenosis mimicking liver metastases is reported. In addition, the PubMed database was searched between January 1993 and December 2016 for literature relating to splenosis, and the resultant literature was analysed in order to provide information on the diagnosis and treatment of this disease.
Case report
This work was conducted following approval by the ethics committees at Chongqing University Cancer Hospital. The patient provided written informed consent.
The patient was a 41-year-old male who was referred to Chongqing Cancer Hospital in December 2015 with liver tumours of unknown origin. He had no history of weight loss, abdominal pain or jaundice. His medical history included an urgent splenectomy due to traumatic rupture of the spleen as the result of a traffic accident in July 1994. There was no self-reported history of smoking or alcohol misuse. There was no positive sign on physical examination, except for a previous surgical scar. Routine blood analysis showed a platelet count of 521 × 109/l and haemoglobin of 76 g/l. His liver function was normal and graded as A (score, 5) according to the Child-Turcotte-Pugh classification. 4 His α-fetoprotein (AFP) level was 1.3 ng/ml (normal range, 0–8.1 ng/ml), carcinoembryonic antigen was 1.27 ng/ml (normal range, 1–5 ng/ml), and carbohydrate antigen 19-9 was 10.76 U/ml (normal range, 0–30.9 U/ml). Chest radiography was normal. Ultrasonography revealed a mass in the right liver region and an abdominal computed tomography (CT) scan showed multiple lesions in the liver, abdominal wall and splenectomy bed (Figure 1). A metastatic process was initially suspected due to anaemia and multiple lesions. No tumours were found by oesophagogastroduodenoscopy and colonoscopy, however, a duodenal ulcer was found. Since the liver lesions had an unknown aetiology, a biopsy was recommended. The patient refused a transcutaneous biopsy, and thus, an exploratory laparoscopy was indicated following a multidisciplinary consultation.
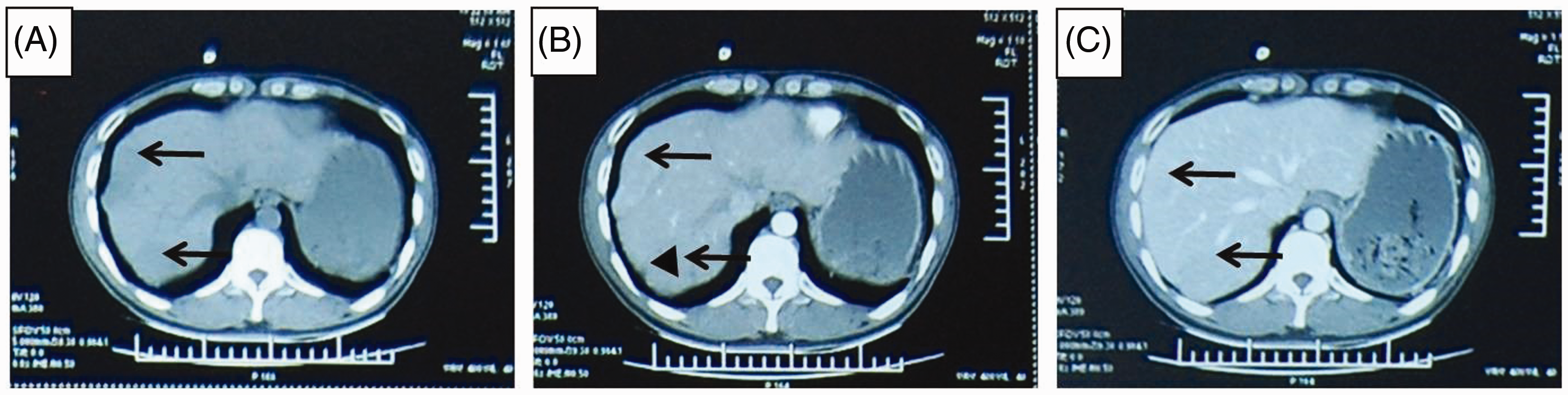
Abdominal computed tomography scan showing lesions in the liver located near the liver capsule (arrows) with a rim surrounding the lesion (triangle): (A) low density lesions located near the capsule in unenhanced phase; (B) lesions washed-in and enhanced during hepatic arterial phase revealing a rim surrounding the lesion (triangle); and (C) lesions washed-out in portal venous phase
The patient underwent an exploratory laparoscopy. General anaesthesia was induced using sufentanil (15 µg total dose), midazolam (2 mg total dose), propofol (100 mg total dose) and rocuronium bromide (50 mg total dose), and anaesthesia was maintained using remifentanil (2 mg total dose), propofol (1400 mg total dose) and sevoflurane (50 ml total dose). Sufentanil (45 µg) and atracurium (15 mg) were administered according the patient’s condition during surgery. A 1.3-cm incision was made along the superior umbilical fold. A pneumoperitoneum was created with a Veress needle, and a 10-mm trocar was placed as a camera port. Two 12-mm trocars and two 5-mm trocars were then introduced with direct visual observation at the right upper quadrant, subxiphoid, right midclavicular, and anterior axillary lines, respectively. Operative findings showed an intrahepatic lesion of 4 × 2 cm in size located on the surface of liver segment VIII and a nodule of 1.5 × 1.3 cm on the abdominal wall. A lesion in segment VII adhered to the diaphragm and dented the surface of the liver. All lesions had clear boundaries with the liver, and were rapidly removed without hepatic resection. Subsequent histopathological results confirmed that the lesions consisted of spleen tissue (Figure 2).
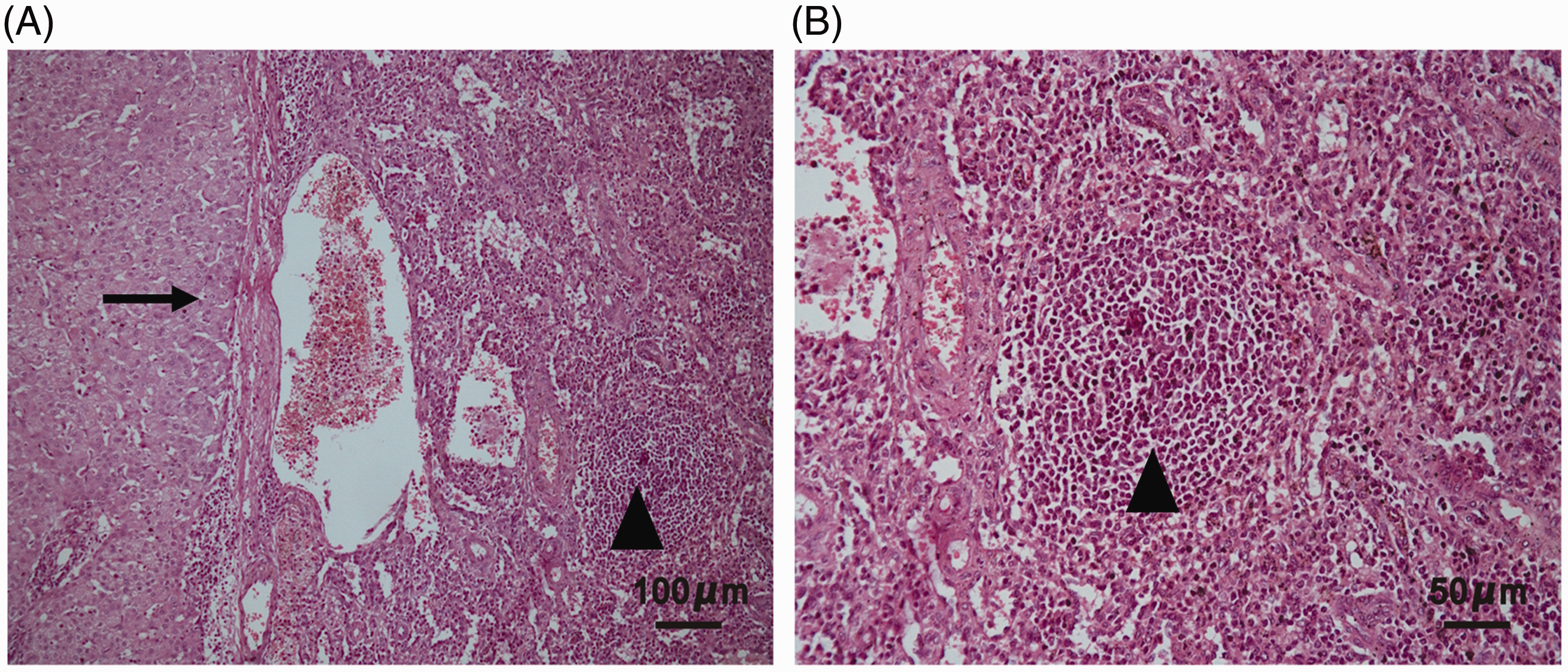
Representative photomicrographs of a liver lesion tissue section stained with haematoxylin and eosin showing hepatic splenosis and normal liver tissue: (A) fibrous tissue isolates the liver and spleen parenchyma (arrow) with liver tissue on the left and spleen tissue on the right including lymphoid follicular tissue (triangle), original magnification × 100; and (B) lymphoid follicular tissue (triangle), original magnification × 200.
The diagnosis of hepatic splenosis was thus established. Follow-up examination including routine blood analysis, liver function, and liver CT scanning at 18 months did not show any abnormalities. No adverse or unanticipated events occurred.
Discussion
The first case of splenosis was described by Albrecht in 1896 5 and the condition was named by Buchbinder and Lipkoff in 1939. 6 Splenosis is mainly found in patients who have undergone splenectomy 1 and although is considered a rare event, its incidence can be up to 67% in cases of traumatic splenic rupture. 2 Splenosis can be found in any part of the body, but common sites include the serosal surface of the small bowel, greater omentum, and parietal peritoneum, and the serosal surface of the large intestine and mesentery. 3
Hepatic splenosis is frequently found in the left lobe of the liver and is usually located near the liver capsule. The most likely mechanism underlying splenosis is the seeding of splenic fragments onto serosal surfaces during splenic trauma or splenectomy 7 and cell proliferation, promoted by local hypoxia of the liver. 8 Hepatic splenosis is rarely identified in the clinic, with no more than 41 cases published between 1993 and 2016 (Table 1),1,8–44 and due to its low prevalence, knowledge of the clinical features and diagnosis of splenosis remains limited. Hepatic splenosis has no specific symptoms, although abdominal pain may arise due to heterotopic splenic infarction or compression, resulting in missed diagnosis.10,11 Hepatic splenosis is difficult to distinguish from adenoma and hepatocellular carcinoma, and can occur as single or multiple lesions at the surface or within the liver,1,12,13 which may be misinterpreted as metastatic lesions in patients with malignant tumours.
Results summary from a literature review of hepatic splenosis showing 38 articles detailing 41 cases of hepatic splenosis published between 1993 and 2016.
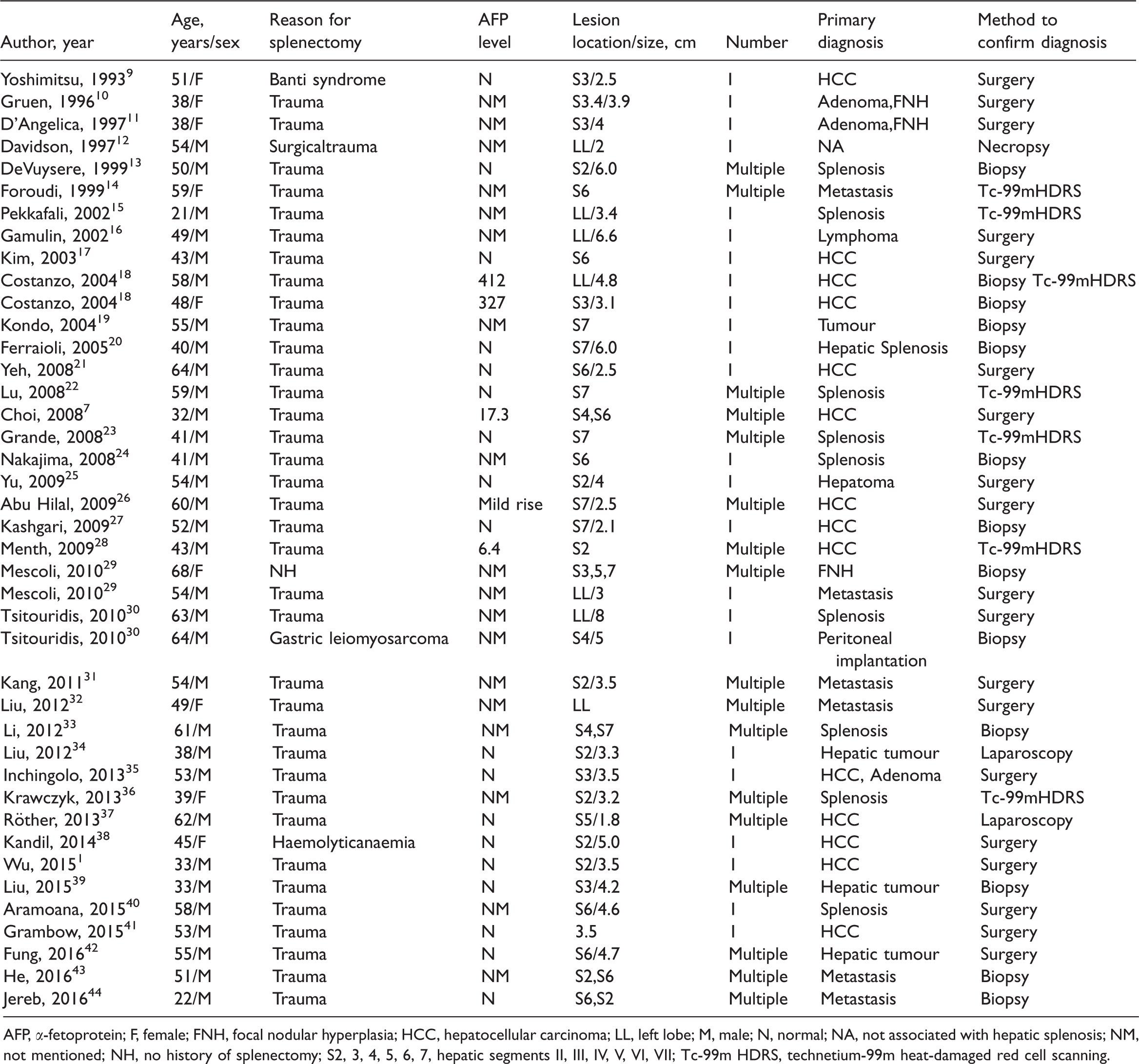
AFP, α-fetoprotein; F, female; FNH, focal nodular hyperplasia; HCC, hepatocellular carcinoma; LL, left lobe; M, male; N, normal; NA, not associated with hepatic splenosis; NM, not mentioned; NH, no history of splenectomy; S2, 3, 4, 5, 6, 7, hepatic segments II, III, IV, V, VI, VII; Tc-99m HDRS, technetium-99m heat-damaged red cell scanning.
In the present case, secondary liver cancer was initially suspected due to ultrasonography and CT findings of multiple lesions in the liver, abdominal wall and splenectomy bed. Furthermore, low AFP levels and negative results for hepatitis increased the possibility of metastatic lesions. Due to negative findings on endoscopy, and patient refusal to undergo a transcutaneous liver biopsy, laparoscopic exploration was performed. The intrahepatic lesions were removed and diagnosed as hepatic splenosis by pathology.
False diagnoses not only have the potential to increase medical costs and the risk of adverse effects due to an invasive procedure, but also result in incorrect treatment. In the published case studies found in the present study, only 10 patients were appropriately diagnosed13,15,20,22–24,30,33,36,40 and the remaining patients were incorrectly diagnosed with liver tumours. Misdiagnosis of liver cancer due to chronic hepatitis and abnormal AFP usually results in patients receiving unnecessary transcatheter arterial chemoembolization 7 or surgeries, 26 or losing the opportunity of liver transplantation. 18 Such incorrect treatments have resulted in secondary damage in these patients.17,18 With the increased prevalence of abdominal trauma,45,46 hepatic splenosis may occur more often. Therefore, standard and effective diagnostic criteria need to be established for this disease.
In addition to a history of splenic trauma and splenectomy, alternative techniques have been used to distinguish hepatic splenosis from other hepatic masses, such as CT scans, 15 technetium-99m heat-damaged red cell scanning (Tc-99m HDRS) and superparamagnetic iron oxide-enhanced magnetic resonance imaging (SPIO-MRI).13,23,28 Low AFP level has been suggested as an index for diagnosing hepatic splenosis, however, in patients who also have hepatocellular carcinoma (HCC) or chronic hepatitis, the splenosis could be misinterpreted as a liver tumour, resulting in incorrect disease staging and loss of opportunity for optimal surgery/transplant or conservative treatments. Thus, pathological evidence was also required to confirm diagnosis according to the results of surgery or biopsy in 35 of the reported patients.1,7,9–11,13,16–21,24–27,29,35,37–44 Based on current knowledge, the present authors propose the following criteria and process for diagnosing hepatic splenosis. Criteria: (1) a history of splenic trauma or splenectomy; (2) lesion(s) with a surrounding rim, particularly near the liver capsule identified by CT scanning; (3) findings on SPIO-MRI or Tc-99m HDRS; and (4) histopathological findings (needle biopsy or surgical pathology). Diagnostic process: suspect diagnosis of hepatic splenosis when criteria 1 and 2 are met; make diagnosis when criterion 3 is met; confirm diagnosis when criterion 4 is met.
Splenosis treatment relies on the reason for splenectomy and complications of the lesion. For patients who have undergone splenectomy for splenic trauma, conservative treatment may be given in most cases without compressive symptoms. For patients who have undergone splenectomy for haematologic disorders, removing the splenosis may prevent disease recurrence. 47 If invasive procedures are necessary (for either diagnosis or treatment), laparotomy is recommended.
The present results may be limited by the retrospective nature of the case study and literature review. Thus, data on more cases should be continuously collected to further assess and verify the proposed diagnostic criteria, and a future observational study remains necessary to elucidate the specificity and sensitivity of each of the criteria.
In conclusion, due to the rareness and asymptomatic nature of hepatic splenosis, it remains difficult to diagnose, leading to missed and false diagnoses. Following a case study and review of the literature, the present authors have proposed criteria and a process for diagnosing hepatic splenosis. Laparotomy is recommended for either diagnosis or treatment when invasive procedures are necessary.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
This work was supported by grants to NQ Gong from the Clinical Research Physician Program of Tongji Medical college, HUST and to XF Sun from the Major State Basic Research Development Program of China (No. 2013CB530803, 973).
