Abstract
Objective
In 2018, colorectal cancer (CRC) was the second most frequent malignancy in Romania after lung cancer. Although CRC is typically encountered in patients >50 years old, CRC's global incidence among younger adults has been increasing. We aimed to compare the disease characteristics of patients with CRC aged ≤50 years with those >50 years old.
Methods
We retrospectively evaluated data from patients with CRC who underwent standard surgery at “Pius Brinzeu” Emergency County Hospital, Timisoara, Romania. Patients were divided into two groups: Group 1 (patients ≤50 years old) and Group 2 (patients >50 years old). Six parameters were analyzed (sex, residence location, age, tumor localization, microscopic findings, pathological staging).
Results
Data on age-related CRC were available for 1380 patients treated from January 2012 to December 2018. Group 1 included 120 patients while group 2 included 1260 patients. Significantly more Group 1 patients presented with advanced CRC compared with Group 2 patients (94.2% vs. 87.4%). Furthermore, CRC in younger adults was more likely to be diagnosed at an advanced stage.
Conclusions
Monitoring the CRC incidence in younger adults is essential to assess whether screening practices require changes and to raise awareness among clinicians of the increasing CRC incidence among younger patients.
Introduction
Colorectal cancer (CRC) is among the most prevalent gastrointestinal malignancies in both women and men, particularly in western countries. 1 In 2018, CRC was the third most common malignancy (10.2% of all new cases of cancer) and the second leading cause of cancer mortality (9.2% of all cancer deaths) worldwide. In Romania, a country with a population of 19,580,628, 11,076 new cases of CRC (13.3% of all new cases of cancer) were diagnosed in 2018, representing the second most frequent malignancy after lung cancer. 2 A significant proportion of CRC cases can be prevented by colonoscopy screening and removal of adenomas before malignant transformation, or by non-invasive stool-based methods such as fecal occult blood testing or fecal immunochemical testing.3,4
Although the incidence of CRC has decreased following the introduction of screening programs, the literature points to an increasing rate of CRC in young adults aged below 50 years, who are typically not included in such programs. 5 Although the prevalence of CRC in adults under 50 years of age without risk factors is low, there has been increasing interest in this patient group in the medical literature, with most researchers reporting more advanced stages of the disease at diagnosis and worse prognosis in younger patients. 6
Given the scarcity of such studies from Romania in the literature, we aimed to evaluate the clinical, sociodemographic, and histological characteristics of young-onset CRC patients (adults <50 years of age) and, importantly, to raise awareness of the need for introducing government-funded screening for CRC in Romania. Although CRC is typically considered a cancer encountered in adults aged above 50 years, clinicians should not rule out CRC as a diagnosis when evaluating younger adults. 7 Our study aimed to determine whether there are different characteristics between young-onset patients with CRC (age ≤50 years) and older patients (age >50 years) in Romania.
Materials and methods
This study was an analysis of retrospectively collected data from patients with a diagnosis of CRC who underwent standard surgery (partial or radical resection with regional lymphadenectomy) between January 2012 and December 2018 at the Emergency County Hospital “Pius Brinzeu” in Timisoara, Romania. We created a database of all consecutive patients with histologically confirmed CRC after surgical intestinal resection. The study was approved by the institutional review board of our hospital. Patient informed consent was not required for this retrospective analysis.
The study assessment included patients with a confirmed diagnosis of adenocarcinoma, mucinous carcinoma, or signet ring cell carcinoma. Patients with inflammatory bowel disease, recurrent CRC, or synchronous tumors were excluded from the analysis. The characteristics of each patient were collected from archives, specimen submission documents, and pathology reports. Patients were divided into two groups according to age: Group 1 included patients ≤50 years old and Group 2 included patients >50 years old. The following patient demographics and clinical parameters were analyzed: sex, location of residence, age, tumor localization, pathological staging, and lymph node ratio (LNR).
Tumors were classified according to the 2010 World Health Organization (WHO) Classification of Tumors of the Digestive System. 8 We evaluated the following parameters: histologic type (conventional adenocarcinoma, mucinous carcinoma, conventional adenocarcinoma with areas of mucinous differentiation, signet-ring cell carcinoma), degree of differentiation (G), presence of lymphatic and venous invasion (LV), tumor extension (pT), and presence of lymph node metastases (pN). Staging was according to the TNM classification of the American Joint Committee of Cancer (AJCC), seventh edition. 9 Tumor localization was defined as right-sided (cecum, ascending colon, hepatic flexure, transverse colon) or left-sided (splenic flexure, descending colon, sigmoid colon and rectum). 6 LNR was defined as the ratio of positive lymph nodes to the total number of removed lymph nodes. 10
All statistical analyses were performed with GraphPad Prism software, v8.2 (GraphPad Software Inc., San Diego, CA, USA). To compare the disease characteristics of patients aged ≤50 years with those of patients aged >50 years, we used the Pearson’s chi-squared test (χ2), Student’s t-test, and Fisher’s test. Results were considered statistically significant for p-values <0.05.
Results
Among 1380 patients with a confirmed diagnosis of adenocarcinoma, mucinous carcinoma, or signet ring cell carcinoma, 120 (8.7%) patients with young-onset CRC and a median age of 44.6 years (range 24–50 years; Group 1) and 1260 patients with late-onset CRC and a median age of 67.8 years (range 51–92 years; Group 2) were included in our study. Among the excluded patients were 53 cases of synchronous colorectal tumors (5 cases in patients aged <50 years).
Out of 183 patients diagnosed with CRC in 2012, 12 (6.5%) were young-onset, whereas in 2018, 16 out of 229 patients (5.2%) were young-onset cases of CRC. The incidence of CRC in young adults (≤50 years) comprised 8.7% of all cases. Among the 120 young adults included in Group 1, 99 (82.5%) were aged between 41 and 50 years at diagnosis. The characteristics of the patients included in our study are presented in Table 1.
Clinico-pathological characteristics of patients included in the study.
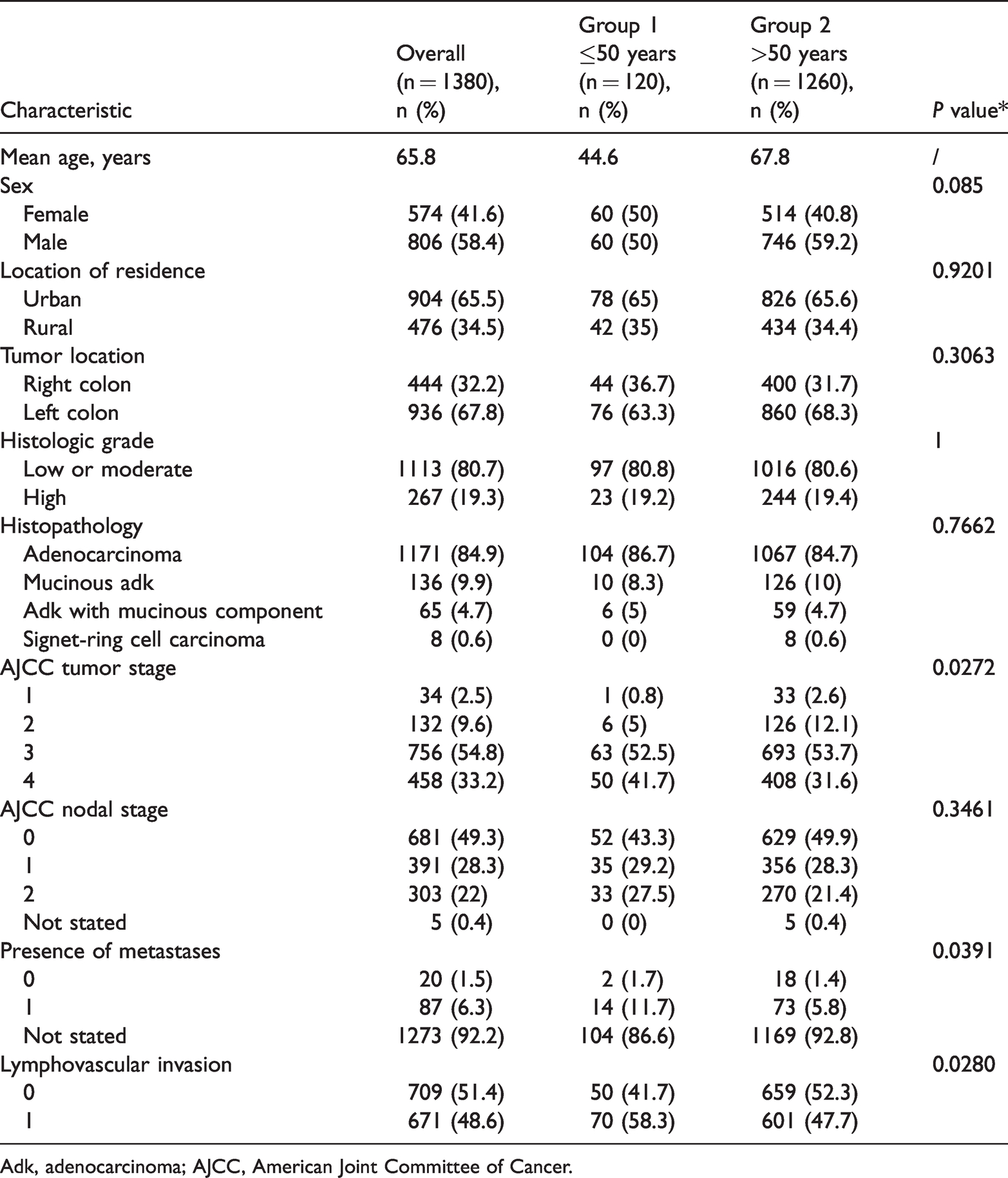
Adk, adenocarcinoma; AJCC, American Joint Committee of Cancer.
Group 1 included 60 women and 60 men (both representing 50% of the total) and Group 2 included more male than female patients (59.2%), with no statistical significance. Few studies to date have examined the effects of geographic factors on CRC incidence. We assessed the effects of rural versus urban residence on patients with CRC in our study. Although there was an overall significant difference between the proportion of patients with urban and rural residence (65.5% vs 34.5%, respectively; p < 0.001), no significant difference was observed between the two groups. In both groups, the most common tumor location was the left colon (>60% in both groups).
Regarding the presence of metastases in other organs, 11.7% of Group 1 patients had metastases, mostly in the liver, whereas metastases were observed in only 5.8% of cases (p = 0.0391) in Group 2. We also found that 56.7% and 49.7% of patients in Group 1 and Group 2, respectively, had nodal involvement at diagnosis.
Histological classification was examined in both groups. The most common histological type was adenocarcinoma not otherwise specified (NOS) in both groups (86.7% vs. 84.7%), followed by mucinous adenocarcinoma (8.3% vs. 10%) and adenocarcinoma with mucinous component (5% vs. 4.7%). Signet-ring cell carcinoma type was found only in Group 2, representing 0.6% (8) of cases. Regarding histological grade, G1 and G2 (well and moderately differentiated) were predominant in Group 1 and Group 2, occurring in 80.8% and 80.6% of cases, respectively.
Compared with patients with late-onset CRC (Group 2), younger patients (Group 1) were more likely to have advanced stage (pT3/pT4) disease (94.2% vs. 87.4%, p < 0.05), lymph node metastases (56.7% vs. 49.7%), and lymphovascular invasion (58.3% vs. 47.7%, p = 0.028). We also observed that, despite a low tumor histological grade, 94.2% of the Group 1 cases were diagnosed at an advanced stage of the disease (pT3/pT4).
A total of 24,537 lymph nodes were removed from all 1380 patients, with an average of 35.7 lymph nodes removed per patient. Overall, 2728 positive lymph nodes were detected. The average number of positive lymph nodes per patient was 3.7, and the average LNR was 22.1%. A positive correlation was found between the number of positive lymph nodes and the number of removed lymph nodes (p < 0.001). LNR also correlated with the number of removed lymph nodes (p < 0.001) and was positively correlated with the number of positive lymph nodes (p < 0.001), as shown in Table 2.
Relationship between lymph node ratio and pathologic factors.

LN, lymph node; LNR, lymph node ratio.
Discussion
CRC in younger adults represents a challenge for physicians as well as for patients and their families. Our findings show that CRC in this patient group tends to be diagnosed at a more advanced stage, with over 94% of patients presenting with pT3/pT4 tumors and 56.7% with regional metastatic disease. In comparison, 87.4% of patients older than 50 years presented with advanced-stage disease and 49.7% had regional metastatic disease.
The current guidelines and literature are not clear on the definition of “young adult CRC” or “young-onset CRC”; therefore, consensus on the age cut-off is needed because age groups are based on epidemiologic screening or clinical trial criteria. Similar to most global studies, we used 50 years as the cut-off age in our analysis because most national screening programs start at this age. However, different cut-off values have been used in other studies; for example, Teng et al. 11 reported that the incidence of CRC in young adults (aged 15–39 years) is rising in the United States. Screening colonoscopy was introduced in 2002 in Germany for patients aged 55 years and above, so the study by Ambe et al. 12 classified those aged below 55 years as young patients. In our study, out of 183 patients diagnosed with CRC in 2012, 12 were young-onset (6.5%), while in 2018, 16 out of 229 patients were young-onset CRC cases (5.2%) and 8.7% of all patients with CRC included in the study were diagnosed before the age of 50.
Population-based studies have shown that rates of CRC in young adults differ among countries, from low incidence in countries in Asia, 13 potentially attributable to a higher consumption of fruit and vegetables, 14 to a higher incidence in the United States 15 and Canada. 9 Bailey et al. 16 predicted a doubled incidence of CRC among younger adults by 2030. Studies from Europe indicate that the rate of CRC increased by 7.4% annually between 2008 and 2016. 17 We identified only one previous study from Romania, published in 2015, in which the incidence of young-onset CRC was 5.5%. 18 The reason for the increasing incidence of CRC remains unclear, but may be associated with dietary factors such as intake of processed meat, high rates of obesity, or sedentary lifestyle. 19
Screening for CRC is recommended from the age of 50 in current guidelines in most European countries. 17 From 2018, screening in the United States is recommended from age 45, 1 and the incidence of CRC in the general United States population has decreased as a result of screening measures. 20 A similar decrease has been observed in Germany, where screening colonoscopy, which is fully covered by health insurance, was introduced in 2002. 21 It is important to note that in Romania, as in the other European Union (EU) member states Bulgaria, Greece, and the Slovak Republic, there is no national screening program for CRC at present. 22 The unfavorable stage distribution observed among patients diagnosed after 50 years of age in our analysis highlights the need for a national screening among this age group in Romania. Furthermore, to ensure proper monitoring of such a screening program, a specialized colon cancer registry should be implemented. An EU-wide study 23 conducted between 2013 and 2016 in all EU countries found that only 6.3% of the Romanian population aged 50 to 74 years had undergone a fecal test within the preceding 2 years or a colonoscopy within 10 years, the lowest incidence among EU countries.
Regarding tumor localization, our findings are consistent with those from several retrospective studies conducted in other countries. For example, Quah et al., 24 in a study conducted at the Memorial Sloan Kettering Cancer Center in the United States, found that patients aged ≤40 years were more likely to have left-sided tumors.
Some studies report poor prognosis in young adults diagnosed with CRC attributable to tumor variables such as signet-ring cell or mucinous adenocarcinoma histological type 25 or to poor differentiation. 26 However, no significant difference in these tumor variables was observed between patients with young-onset CRC versus older patients with CRC in our study, in contrast to most published studies. Other studies have also reported a better prognosis among younger patients 27 when age groups were compared. Delayed diagnosis has been proposed as a potential explanation for the higher rate of advanced-stage disease at clinical presentation in younger adults. The incidence of advanced-stage disease (pT3 or pT4) at presentation in our analysis was significantly higher in younger patients, similar to other studies from Europe, 28 the United States, 29 and Asia. 30
Our study had several limitations, including a lack of information on the oncology treatment received and the subsequent progress of patients after surgery; thus the overall survival rate could not be established. Furthermore, this was a single-institution study, meaning that the results require confirmation in more extensive studies in Romania to investigate the incidence of CRC in younger adults in our country. Despite these limitations, the results of our study are comparable to the available scientific literature regarding different disease characteristics in younger adults with CRC.
Conclusions
In our study, which included a substantial number of patients with CRC diagnosed under the age of 50 over a 7-year period we report that young-onset CRC was more likely to be diagnosed at an advanced stage (pT3, pT4) with nodal or distant metastasis, indicative of more advanced disease and poorer prognosis. Our findings highlight the necessity of monitoring the incidence of CRC in the younger population to assess whether changes are needed in screening practices and to raise awareness among clinicians of the rising incidence of CRC in this patient population.
Footnotes
Author contributions
Adelina Gheju: Conceptualization, data curation, methodology, software, writing the original draft.
Aura Jurescu, Diana Al-Jobory: Supporting data curation, supporting the writing of the original draft, software.
Fulger Lazar: Supporting data curation, supporting the writing of the original draft.
Sorina Taban, Alis Dema: Reviewing microscopy data, supporting the methodology, supervision of the concept, validation.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
