Abstract
Pleomorphic rhabdomyosarcomas of the uterus (PRMSu) is a rare malignant tumor of the female genital tract. Accurate diagnosis and effective treatment of PRMSu are important. We report an 81-year-old woman who was diagnosed with PRMSu. She had an extremely unusual presentation of secondary dyspnea because of an extremely large uterus (26.0 cm). Pelvic magnetic resonance imaging showed rare severe enlargement and intrauterine filling with tumor tissue, and she was initially diagnosed with uterine leiomyosarcoma. The patient underwent hysterectomy, as well as bilateral salpingo-oophorectomy and omentectomy, and was finally confirmed as having PRMSu by histopathology combined with immunohistochemistry. We performed a systematic review of the literature between 1982 and 2020 and focused on different treatment strategies and prognosis of PRMSu. A retrospective review of 28 cases was conducted and survival analysis was estimated by using the Kaplan–Meier method. We found that the accuracy of diagnosis of PRMSu completely depends on histopathology and immunohistochemistry because of no special clinical symptoms, no sensitive tumor markers, and no special imaging findings. Although there is no standardized approach for treating this rare disease, the treatment strategy of a surgical operation combined with adjuvant chemotherapy appears to be the best choice.
Keywords
Introduction
Pleomorphic rhabdomyosarcoma of the uterus (PRMSs) in adults is rare and has a poor prognosis. 1 PRMSu mainly occurs in postmenopausal women, and its main symptoms are vaginal bleeding and abdominal pain. 2 Because of high malignancy of the tumor in PRMSu and the inefficiency of adjuvant therapy, the outcome remains poor in the majority of reported cases.3,4 We present a case of a patient with a rare, large-sized uterus, secondary dyspnea, and usual postmenopausal vaginal bleeding caused by PRMSu without any components of carcinosarcoma or a malignant mixed mesodermal tumor. Complete remission was achieved following surgery. This case report and literature review highlight a differentiated overview on how to diagnose and deal with this rare disease.
Case presentation
We present here a case of an 81-year-old female patient who was admitted to the hospital owing to progressive abdominal enlargement for more than 1 year, accompanied by secondary dyspnea for 1 week. Her clinical manifestations included intermittent postmenopausal bleeding, which occurred three times 6 months previously, lasting for 3 to 5 days each time, and abdominal pain. Moreover, severe dyspnea occurred only in the supine position without a pillow. However, her medical history did not show smoking or respiratory diseases, and only mild hypertension. A physical examination showed an enlarged uterus that was similar in size to that at term pregnancy, with an irregular surface and a substantial abdominal mass that originated from the pelvic cavity (Figure 1). Additionally, her body mass index was 18.67 kg/m2 and score of World Health Organization Performance Status was 3.
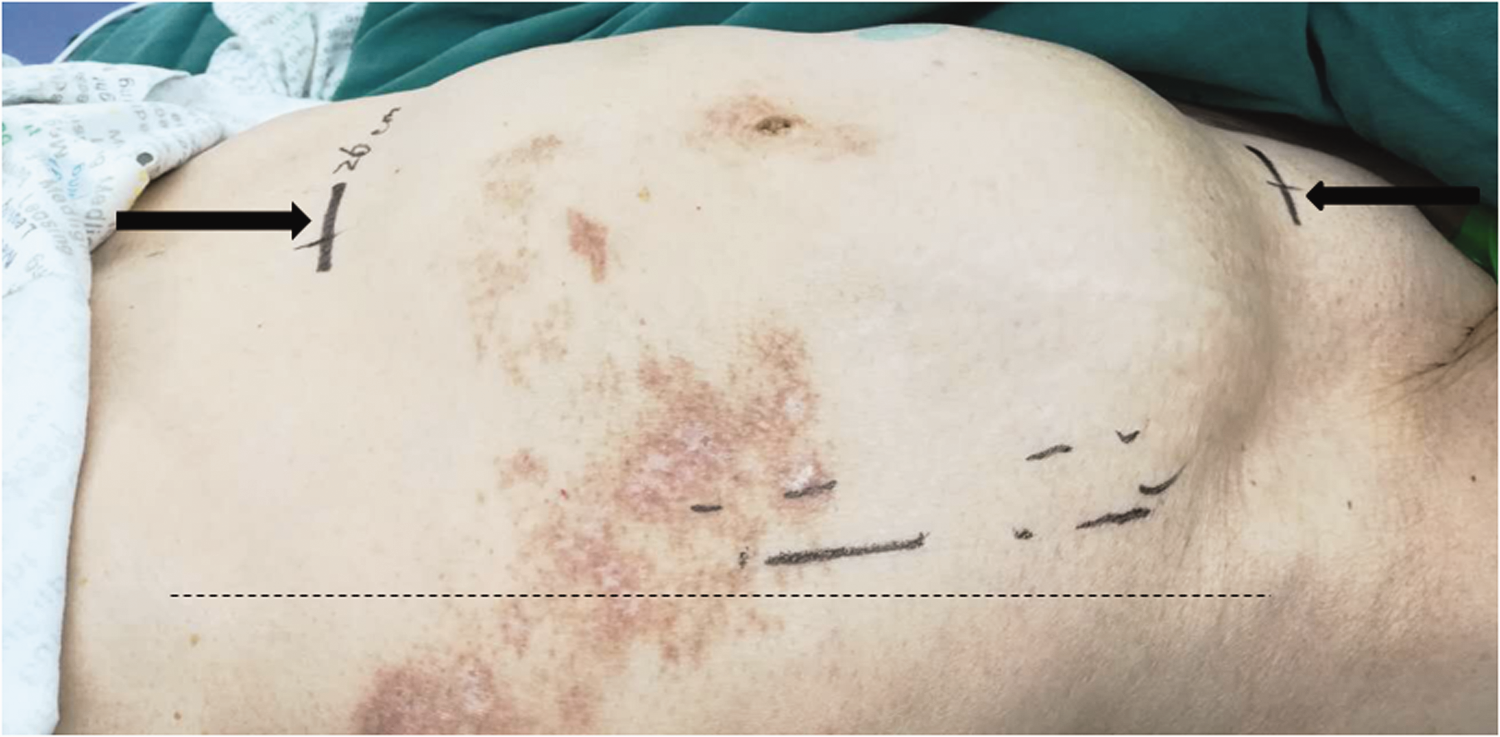
Photograph showing a large solid abdominal mass originating from the pelvic cavity up to the lower pubic symphysis (black arrows), on the left and right sides to the median axillary line (black dotted line). The mass is approximately 26 cm in maximum diameter between the superior border of the pubic symphysis (red arrow) and the bottom of the uterine body.
A blood test and liver function tests showed mild anemia (102 g/L) and hypoalbuminemia (24 g/L) after admission. Tumor markers were in the normal reference range, including cancer antigen (CA) 199, carcinoembryonic antigen, alpha fetoprotein, CA153, and neuron-specific enolase, and only CA125 was slightly elevated to 55 U/L. Chest computed tomography showed that both lungs were slightly thickened without nodules with enlarged lymph nodes. Magnetic resonance imaging of the abdomen and pelvis suggested uterine leiomyosarcoma (Figure 2). The patient was initially diagnosed with leiomyosarcoma stage I.

(a) T2-weighted fat-suppression sequence and (b) T1-weighted enhanced sequence show that the uterus is extremely enlarged, with a size of 267 × 180×122 mm3. There is a clear border and uneven internal signals, and irregular inner walls showing cauliflower and papillary changes. There is also liquefied and necrotic tumor tissue that fully fills the uterine cavity.
Under multidisciplinary cooperation and complete correction of hypoalbuminemia and anemia, the patient underwent exploratory laparotomy, total hysterectomy, and bilateral salpingooophorectomy and omentectomy. Intraoperatively, the peritoneum showed signs of further tumor spread. However, in a palpation examination of the liver and parietal peritoneum of the pelvis, no metastases were found. She recovered well postoperatively and was discharged home on day 8. Further chemotherapy or pelvic radiation was recommended, but not accepted. Follow-up physical examination and magnetic resonance imaging 4 months after surgery showed no abnormalities or signs of tumor recurrence.
Finally, the patient was diagnosed with PMRSu by histopathology combined with immunohistochemistry and stage III C according to the International Federation of Gynecology and Obstetrics 2009. Macroscopically, the surface of the uterus was smooth and irregular, and weighed 5200 g, and its size was approximately 26.0 cm in maximum diameter (Figure 3a). Furthermore, in the uterus, the tumor tissue showed diffuse infiltrative growth, with a hard texture and a small amount of necrosis and liquefaction (Figure 3b).
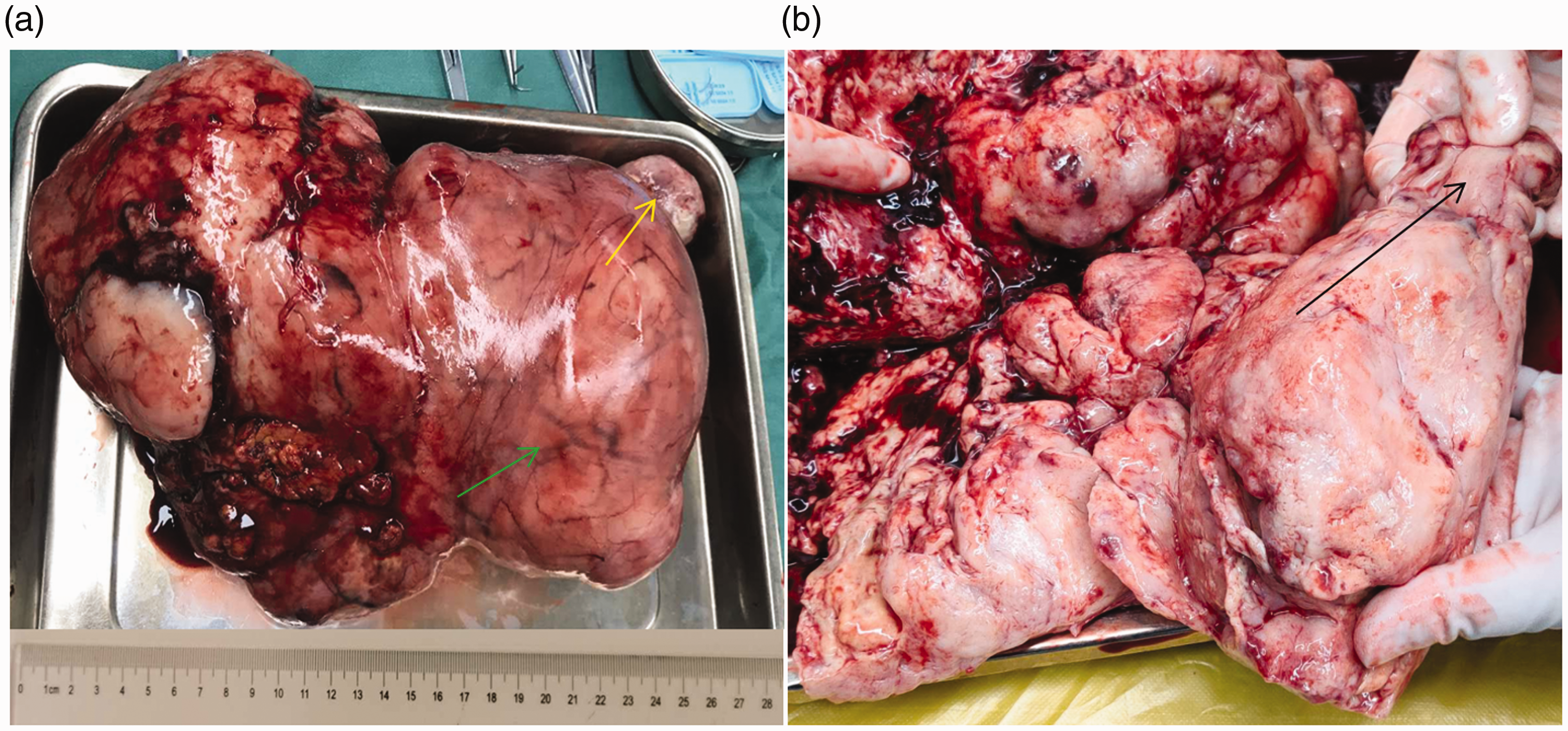
(a) Photograph showing the entire uterine body of approximately 26 cm in maximum diameter, with hyperplastic tumor blood vessels (blue arrow) and cervical atrophy (yellow arrow). (b) Photograph showing that the normal uterine cavity has completely disappeared and replaced by a diffuse fish-like appearance in growth of tumor tissue. Part of the tumor shows necrosis and liquefaction, with a hard and brittle texture, and infiltration of the deep muscle layer. There is atrophy of the cervical canal (green arrow).
Histology of formalin-fixed, paraffin-embedded, hematoxylin–eosin-stained tumor sections showed sheets of large atypical cells and polygonal PMRSu with abundant acidophilic cytoplasms (Figure 4a–c). The tumor cells invaded adipocytes of the omentum and showed fat infiltration (Figure 4d).
Figure 4. (a) Hematoxylin–eosin-stained tumor sections show a pleomorphic atypical cell sheet, which is diffusely distributed. (b) Scattered large, polygonal, pleomorphic rhabdomyoblasts with an abundant acidophilic cytoplasm can be seen. (c) Bizarre giant cells are common. (d) Adipocytes infiltrating the omentum by tumor cells can be seen.

Immunohistochemical reactions for epithelial membrane antigen (CD34), Melan A (HMB45), α-smooth muscle actin, calmodulin (caldesmon), and S-100 were negative (Figure 5). In contrast, demsin was overexpressed in the cytoplasm of myogenic tumor cells, including pleomorphic rhabdomyoblasts and normal vascular smooth muscle cells (Figure 6a). MyoD1 and myogenin, which are specific markers of striated muscle cells, showed moderate nuclear staining in morphologically-appearing rhabdomyoblasts (Figure 6b–c), and Ki-67 positivity was approximately 20% (Figure 6d). The final diagnosis was PRMSu with metastasis to the great omentum. The pathological results were confirmed by Zhejiang Province Pathology Association.
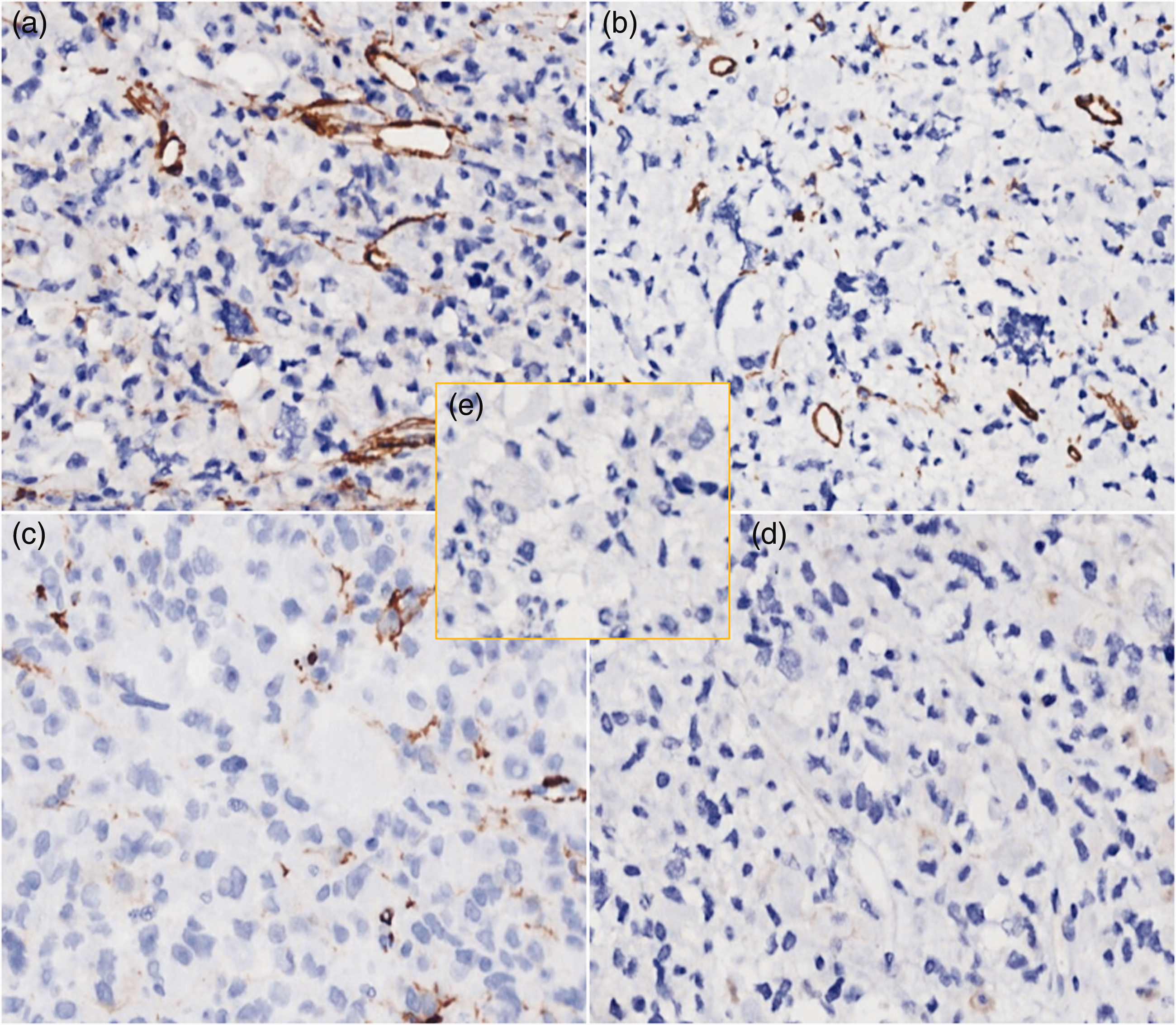
Immunohistochemical reactions for (a) CD34, (b) HMB45, (c) smooth muscle actin, (d) caldesmon, and (e) S-100 are negative.

(a–c) Demsin overexpression in the cytoplasm of myogenic tumor cells. (a) MyoD1 and (b) myogenin show moderate nuclear staining in morphologically-appearing rhabdomyoblasts. (c) Ki-67 staining was approximately 20% positive.
We performed a systematic review of the literature that focused on different treatment strategies and prognosis. We found 28 cases of PRMSu described in the English language literature between 1982 and 2020.3–18 Recorded variables, including age, size of the uterus, vaginal bleeding, extrauterine spread, treatment methods, and follow-up are shown in Table 1. The Kaplan–Meier method was used to analyze and compare the overall mean survival rate and different mean survival rates under different treatment strategies. IBM SPSS statistical software version 19.0 (IBM SPSS, Armonk, NY, USA).
Literature overview on clinical features of pleomorphic rhabdomyosarcoma of the uterus.

NED, no evidence of disease; DOD, died of disease; AWD, alive with disease; HE, hysterectomy; BSO, bilateral salpingo-oophorectomy; LNE: lymphonodectomy; OE, omentectomy.
This systematic review of the literature showed that the mean age (± standard deviation) of patients was 67.50 ± 9.83 years and age ranged from 35 to 87 years. The mean size of the uterus was 12.00 ± 6.28 cm (range: 4.5–26 cm), and 16 of the 28 patients presented with extrauterine spread at the time of diagnosis. The overall mean survival was 16.95 ± 4.12 months (95% confidence interval [CI] = 8.87–25.03). The mean survival rate of patients without extrauterine spread at the time of diagnosis was 15.00 ± 2.72 months (95% CI = 9.68–20.33), whereas that with extrauterine spread was 16.24 ± 5.45 months (95% CI = 5.55–26.92). Patients with adjuvant chemotherapy showed the best outcome (95% CI = 8.35–41.15). Patients with only surgical treatment showed the shortest survival at the time of diagnosis (95% CI = 0.10–12.54) (Table 2).
Summary and analysis of 28 cases of pleomorphic rhabdomyosarcoma of the uterus.
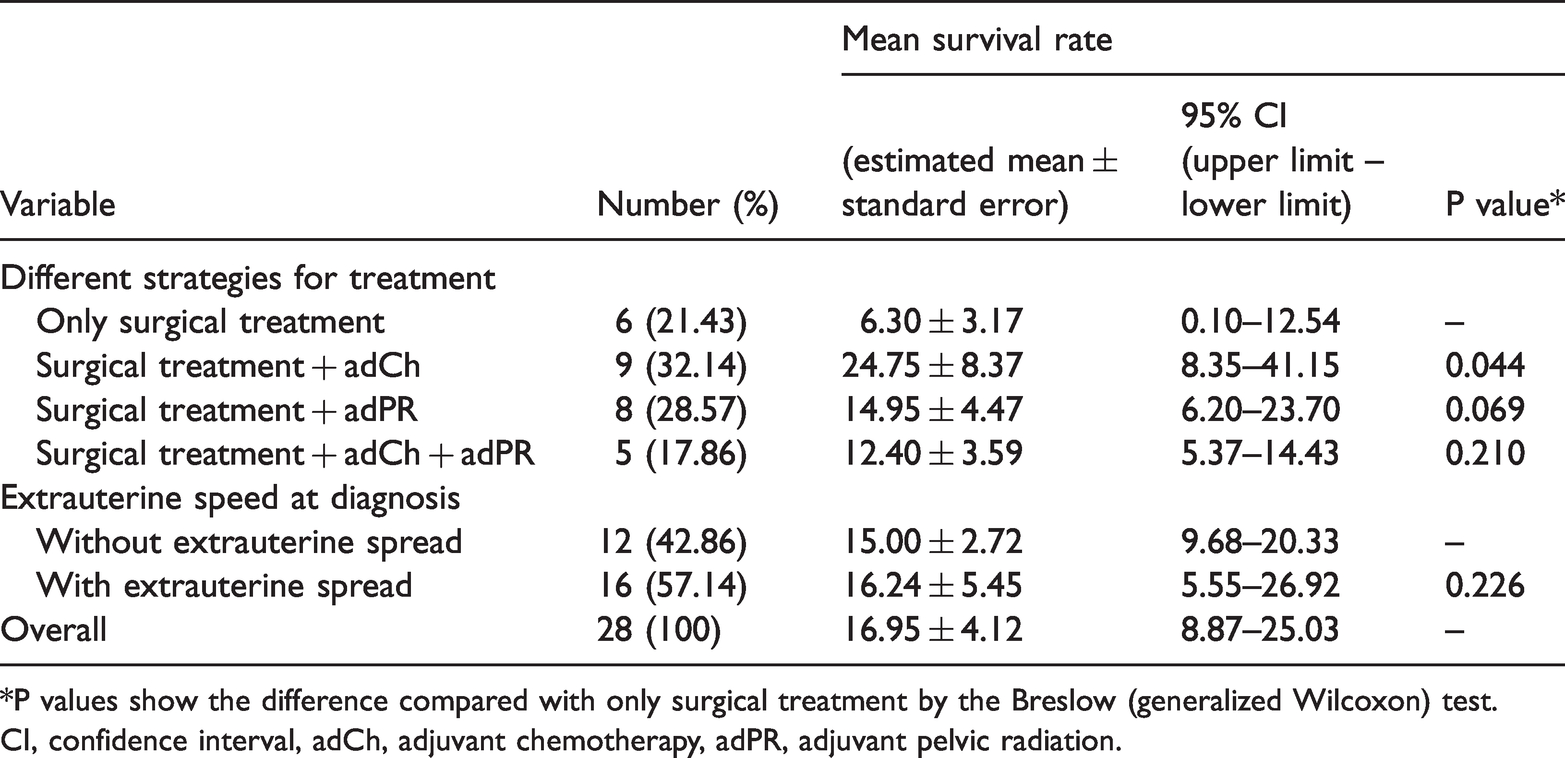
*P values show the difference compared with only surgical treatment by the Breslow (generalized Wilcoxon) test.
CI, confidence interval, adCh, adjuvant chemotherapy, adPR, adjuvant pelvic radiation.
Discussion
Conventionally, rhabdomyosarcoma originating from the uterus was classified as pleomorphic, embryonal, or alveolar sarcoma. 19 PRMSu is rare and occurs almost exclusively in adults with no cases reported in children. 3 We present the case of an 81-year-old woman with PRMSu due to a rare large tumor (5200 g, 26 × 18×12 cm3). The largest previously reported tumor in PRMSu was 18 cm in maximum diameter. 15 Secondary dyspnea of our patient was in complete remission following surgery, and the reason for this sign may have been caused by considerable uterine compression. The clinical symptoms of PRMSu, such as abdominal enlargement and abdominal pain, are non-specific. However, postmenopausal bleeding may be of clinical value for diagnosis of uterine malignancy. A review of the literature and our case showed that uterine bleeding (100%, 27/27) occurred in postmenopausal women.
Many studies have suggested that PRMSu is a highly malignant sarcoma, and it is composed of multiform, sometimes fusiform, frequently large multinuclear neoplastic cells with an acidophilic cytoplasm.20,21 Additionally, neoplastic cells are chaotically distributed in connective tissue stroma and transverse striation is almost never observed. The above-mentioned histopathological changes were mostly present in the current case. Immunohistochemical analysis is an essential tool for verifying histological types of rhabdomyosarcomas. Desmin expression is notable in PRMSu, 20 Additionally, MyoD1 and myogenin are skeletal muscle-specific markers found in PRMSu, especially MyoD1, 21 and overexpression of desmin and low expression of Ki-67 (only 20%) in our case may have been related to a high differentiation of PRMSu.22,23 Thus, the accuracy of diagnosing PRMSu entirely depends on histopathology and immunohistochemistry.
With regard to our systematic review of the literature, patients with adjuvant chemotherapy showed the best outcome, while patients with only surgical treatment showed the shortest survival after diagnosis. The mean survival rate of patients without extrauterine spread was similar to that in patients with extrauterine spread. These findings are different to those found in clinical practice of rhabdomyosarcomas. 24 The reason for this discrepancy may be that the number of reported cases in our retrospective analysis was too small (n=28, Table 1), resulting in error and deviation of statistical results.
With regard to surgical treatment in all cases (28/28, 100%), total hysterectomy and bilateral salpingo-oophorectomy were performed in 20 (71.43%) patients. This approach may be the basic surgical strategy for treatment of PRMSu, especially for patients without extrauterine metastasis. Whether the omentum and lymph nodes should be removed remains unclear, especially for patients without extrauterine metastasis. However, diagnostic lymph node resection is not necessary unless there is clinical or imaging evidence that lymph nodes are involved. 25
In children, multimodal treatment protocols for rhabdomyosarcoma are well defined and lead to an improved survival rate, especially in those with non-metastatic disease.25,26 In adults, however, the same protocols show much less response.1,27 Because of the rareness of this disease in adults, use of a variety of chemotherapeutic agents in studies involving adult patients have not led to a standardized chemotherapy regime.27–29 Surgery (hysterectomy + bilateral salpingo-oophorectomy, 75%) combined with chemotherapy based on actinomycin and doxorubicine 21 may be the most effective treatment at present. Radiotherapy does not appear to increase the mean survival rate of PRMSu, especially for patients without extrauterine metastasis, as supported by Zeisler et al.’s study. 30
Currently, there is no definite follow-up plan for PRMSu, while follow-up measures of uterine leiomyosarcoma are mostly accepted in the literature. However, life-long follow-up for PRMSu is necessary. Elevated CA125 levels can be used to evaluate whether a uterine malignant tumor has extrauterine metastasis after the operation. This especially applies in advanced metastatic stages of a uterine sarcoma.9,11 In the present case, the patient presented with a mildly elevated CA125 level of 55 U/mL before surgery. Similar to this case, Holcomb et al. 9 reported a case of a patient with PRMSu with an elevated CA125 level, but elevated CA125 levels do not directly reflect the tumor burden. This elevation in CA125 levels may be related to extrauterine metastasis or recurrence of PRMSu.
Conclusions
PRMSu is a rare malignant tumor, which shows a common clinical sign of postmenopausal bleeding and non-specific clinical symptoms, tumor markers, and imaging findings. The accuracy of diagnosing PRMSu is completely based on histopathological analysis, and it often displays multiform and large multinuclear neoplastic cells with an acidophilic cytoplasm. Additionally, overexpression of desmin and positive expression of MyoD1 and myogenin are helpful in differential diagnosis in an immunohistochemical examination. Although there is no standardized approach in the treatment of this rare disease, the treatment strategy of a surgical operation combined with adjuvant chemotherapy may show a good prognosis. Moreover, the basic surgical procedure is total hysterectomy and bilateral salpingo-oophorectomy, which may be especially suitable for patients without extrauterine metastasis.
Ethics statement
Approval for a study protocol was not required because this was a case report. The authors have obtained the patient’s written informed consent for print and electronic publication of this case report.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
Initial drafting of the manuscript and subsequent revisions: Zi-Jun Li; surgical and medical practices: Zi-Jun Li and Xiao-Ying Fu; statistical analysis: Zi-Jun Li and Cheng-Lie Li; literature search and clinical data collected: Wei Wang and Ya-Qin Zhen. All authors have read and approved the final manuscript.
