Abstract
Objective
Preterm infants receive long-term parenteral nutrition (PN) for gastrointestinal immaturity. This study aimed to determine if mixed lipid emulsions containing fish oil decrease the incidence of PN-associated cholestasis by reducing oxidative stress and providing an anti-inflammatory effect.
Methods
This retrospective cohort study enrolled 399 very low birth weight premature infants (gestational age ≤32 weeks) between January 2009 and November 2017 at a single neonatal intensive care unit. Preterm infants received total PN with either mixed lipid emulsion including fish oil (SMOFlipid®, n = 195) or soybean oil-based lipid emulsion (Lipovenoes®, n = 204) for at least 7 days. We compared the outcomes of PN-associated cholestasis, comorbidities, and mortality between the groups.
Results
The incidence of PN-associated cholestasis was significantly lower in the SMOFlipid group than in the Lipovenoes group. The duration to full feeding days was significantly shorter in the SMOFlipid group compared with the Lipovenoes group. Relevant complications, such as severe retinopathy of prematurity and bronchopulmonary dysplasia, were also significantly reduced in the SMOFlipid group compared with the Lipovenoes group.
Conclusion
In premature infants, PN with fish oil-based lipid emulsions is associated with a lower incidence of PN-associated cholestasis compared with soybean oil-based lipid emulsions.
Keywords
Introduction
Preterm neonates rely on parenteral nutrition (PN) for several weeks after birth. Early administration of milk feeding is insufficient to support growth owing to gastrointestinal immaturity. However, the risk of infection, hypertriglyceridemia, hyperbilirubinemia, and PN-associated liver disease is increased in infants receiving long-term PN.1,2 There is a growing amount of evidence that soybean oil emulsions containing a high proportion of ω-6 long-chain polyunsaturated fatty acids (LC-PUFAs) and phytosterols induce inflammation and promote hepatic damage.1–5 The latest generation of mixed lipid emulsions are composed of soybean, medium-chain triglycerides (MCTs), olive oil, and fish oil (SMOFlipid®). Soybean oil provides essential fatty acids, such as linoleic acid and α-linolenic acid.2,3 MCTs are more beneficial for faster metabolic clearance than long-chain triglycerides.6–8 Olive oil is rich in monounsaturated fatty acids, which protect against lipid peroxidation.4,9,10 Fish oil provides ω-3 LC-PUFAs, eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA), which reduce oxidative stress, suppress inflammation, and prevent PN-associated cholestasis.2–5,11 A balanced fatty acid supply affects outcomes, such as growth, visual development, and cognitive development.12–14
Recent studies have investigated the effect of fish oil-containing lipid emulsion on markers of inflammation and immune responses.11,15 Commonly reported inflammatory markers are the proinflammatory cytokines interleukin 1 (IL-1), IL-6, and IL-8, the anti-inflammatory cytokine IL-10, the cell-signaling cytokine tumor necrosis factor alpha, and the ratio of CD4 and CD8 T-cell lymphocytes. A higher CD4/CD8 ratio indicates a satisfactory immune status, whereas a lower CD4/CD8 ratio is associated with immunological dysfunction. 15
Neonatal intermittent hypoxia, excessive and high levels of oxygen, oxidative stress, and inflammation may contribute to retinopathy of prematurity (ROP), which represents abnormal growth of retinal blood vessels in preterm infants.16,17 Aggressive ventilation with high positive pressure and excess volume could lead to alveolar damage and severe local inflammation in the lungs, resulting in bronchopulmonary dysplasia (BPD). 18 Necrotizing enterocolitis (NEC) is an emergent and critical disease characterized by inflammation and necrosis in the gastrointestinal tract. NEC is due to immaturity of multiple intestinal functions, altered anti-inflammatory control, abnormal gut bacterial colonization, inappropriate enteral feeding, and impaired host defense. 19 SMOFlipid emulsions containing a higher proportion of ω-3 to ω-6 LC-PUFAs has many immunomodulatory and anti-inflammatory properties, which may have a beneficial effect on these morbidities. Abdelkareem et al. 20 also reported that SMOF lipid emulsions may result in higher levels of soluble adhesion molecules, which could potentially affect the outcome of sepsis. Zhu et al. 21 showed that the protective effect of fish oil on rats with NEC may lead to alleviation of intestinal mucosal inflammation and injury, which are associated with endoplasmic reticulum stress. Therefore, this study aimed to determine if SMOFlipid is associated with a decrease in the incidence of PN-associated cholestasis and other morbidities compared with soybean oil-based lipid emulsion in preterm infants.
Patients and methods
This retrospective cohort study was performed at the Department of Neonatology, Changhua Christian Children Hospital, Taiwan, between January 2009 and November 2017. A total of 399 premature neonates who were eligible, with a gestational age ≤32 weeks and birth weight ≤1250 g, and who received parenteral nutrition for at least 7 days, were enrolled. The study was approved by the Institutional Review Board at Changhua Christian Hospital, Taiwan, from 3 August 2017 (CCH IRB No.: 080707). We had de-identified all patient details in our databank to protect the patient’s privacy. Signed consent to publish was not required because this was a retrospective cohort study.
Infants with conditions associated with cholestasis independent of PN (i.e., infection with cytomegalovirus, HIV, and hepatitis B or C), hemolytic diseases, primary liver diseases, inborn errors of metabolism, multiorgan failure, or hypoxic–ischemic encephalopathy were excluded. The first cohort of preterm infants received soybean oil-based lipid emulsion (Lipovenoes® 20%, control group) from January 2009 to April 2013. The second cohort of preterm infants received SMOFlipid 20% (study group) composed of 30% soybean oil, 30% MCTs, 25% olive oil, and 15% fish oil as PN lipid emulsion from May 2013 to November 2017. Both cohorts were cared for by the same multidisciplinary team, including medical, pharmacy, dietetic, nursing, and nutritional care.
Regardless of whether PN contained SMOFlipid or Lipovenoes, the dosage of both groups started at 1 g/kg/day on day 1, and this was increased daily by increments of 1 g/kg/day up to a maximum of 3 g/kg/day. The lipid emulsion was administered as part of a PN regimen along with other nutrients (glucose, amino acids, vitamins, trace elements, and electrolytes) in two separate syringes. The dosages of other nutrients were standardized in accordance with current recommendations. 1 The preterm infants received nasogastric tube feeding supplement with formula or breast milk as early as possible. All preterm infants in both groups received probiotics, including Lactobacillus acidophilus and Bifidobacterium bifidum after they started feeding. PN was withdrawn when infants received enteral energy >80 kcal/kg/day.
Demographic data, medical history, concomitant diseases, clinical assessments, and medications of the preterm infants were recorded daily beginning on day 0. Blood samples for assessment of parameters for safety and efficacy evaluation were obtained weekly. Adverse events and their possible relation to study treatment were documented. The primary outcome, which was PN-associated cholestasis, was defined as serum direct bilirubin levels > 25.7 mmol/L, and >20% of serum total bilirubin levels.22,23 Peak levels of liver enzymes (aspartate aminotransferase, alanine aminotransferase, γ-glutamyl transferase, alkaline phosphatase), C-reactive protein, albumin, cholesterol, and triglycerides were also analyzed during hospitalization. Secondary clinical and developmental outcomes included severe ROP, intraventricular hemorrhage (IVH), NEC, BPD, sepsis, and mortality. ROP was screened by an ophthalmologist beginning at 4 weeks of age. Treatment with intravitreal bevacizumab was performed at ROP ≥ stage 3. IVH was diagnosed by cerebral ultrasound on days 0, 3, 7, and 21 and monthly. Severe IVH was identified as stage ≥3. NEC was diagnosed clinically (Bell’s stage ≥ IIa) or after surgical exploration. BPD was defined as requiring an oxygen supplement after 36 weeks’ postmenstrual age. 18 Sepsis was proven by blood culture.
All statistical analyses were performed using IBM SPSS software for Windows, version 22.0 (IBM Corp., Armonk, NY, USA). Data from all of the assessments were tabulated using descriptive statistics. Standard summary statistics and 95% confidence intervals (CIs) were calculated appropriately. To compare the mean values from parametric variables, data were analyzed using the Student’s t test. Secondary outcomes were compared between the groups with the χ2 or Fisher’s exact tests when appropriate for categorical data. A P value of <0.05 was considered statistically significant. Statistical evaluation of results was carried out for the entire patient population and according to stratification.
Results
A total of 399 preterm infants were enrolled, including 195 in the SMOFlipid group and 204 in the Lipovenoes group. There were no significant differences in baseline characteristics regarding gestational age, birth weight, height, head circumference, and the Apgar score at 1 and 5 minutes between the SMOFlipid and Lipovenoes groups (Table 1). The proportion of breast milk feeding was similar in the SMOFlipid and Lipovenoes groups (P = 0.083). However, the mean duration of parenteral nutrition and duration to full enteral feeding were significantly shorter in the SMOFlipid group compared with the Lipovenoes group (both P < 0.001).
Demographic and clinical characteristics.
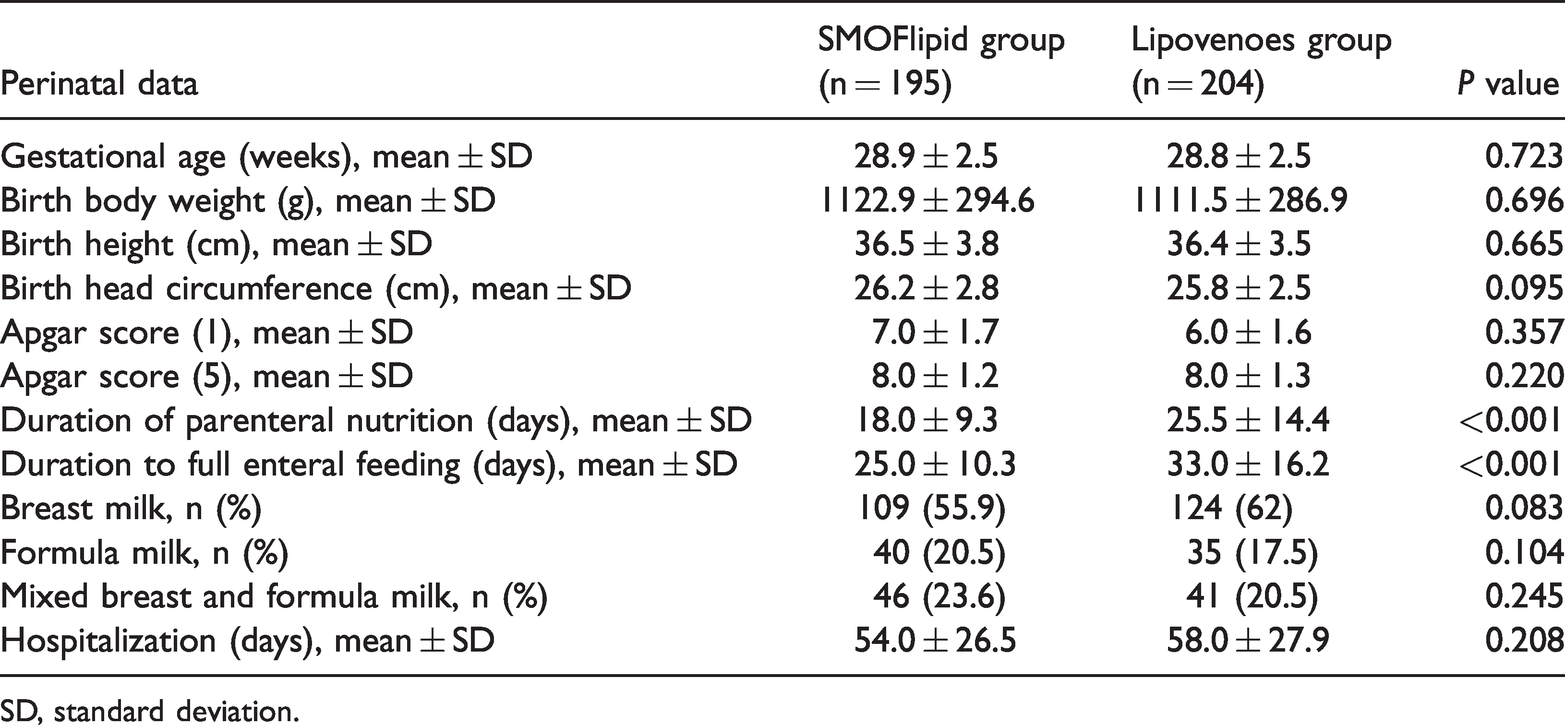
SD, standard deviation.
The rate of the primary outcome of PN-associated cholestasis was significantly lower in the SMOFlipid group compared with the Lipovenoes group (P = 0.006) (Table 2). With regard to laboratory results, γ-glutamyl transferase levels (P = 0.019), triglyceride levels (P < 0.001), and cholesterol levels (P = 0.023) were significantly lower in the SMOFlipid group than in the Lipovenoes group (Table 2). Hemoglobin and hematocrit levels were significantly higher in the SMOFlipid group than in the Lipovenoes group (both P < 0.05). C-reactive protein levels, the white blood-cell count, the platelet count, and the rate of sepsis were not significantly different between the SMOFlipid and Lipovenoes groups.
Primary outcome and laboratory data.
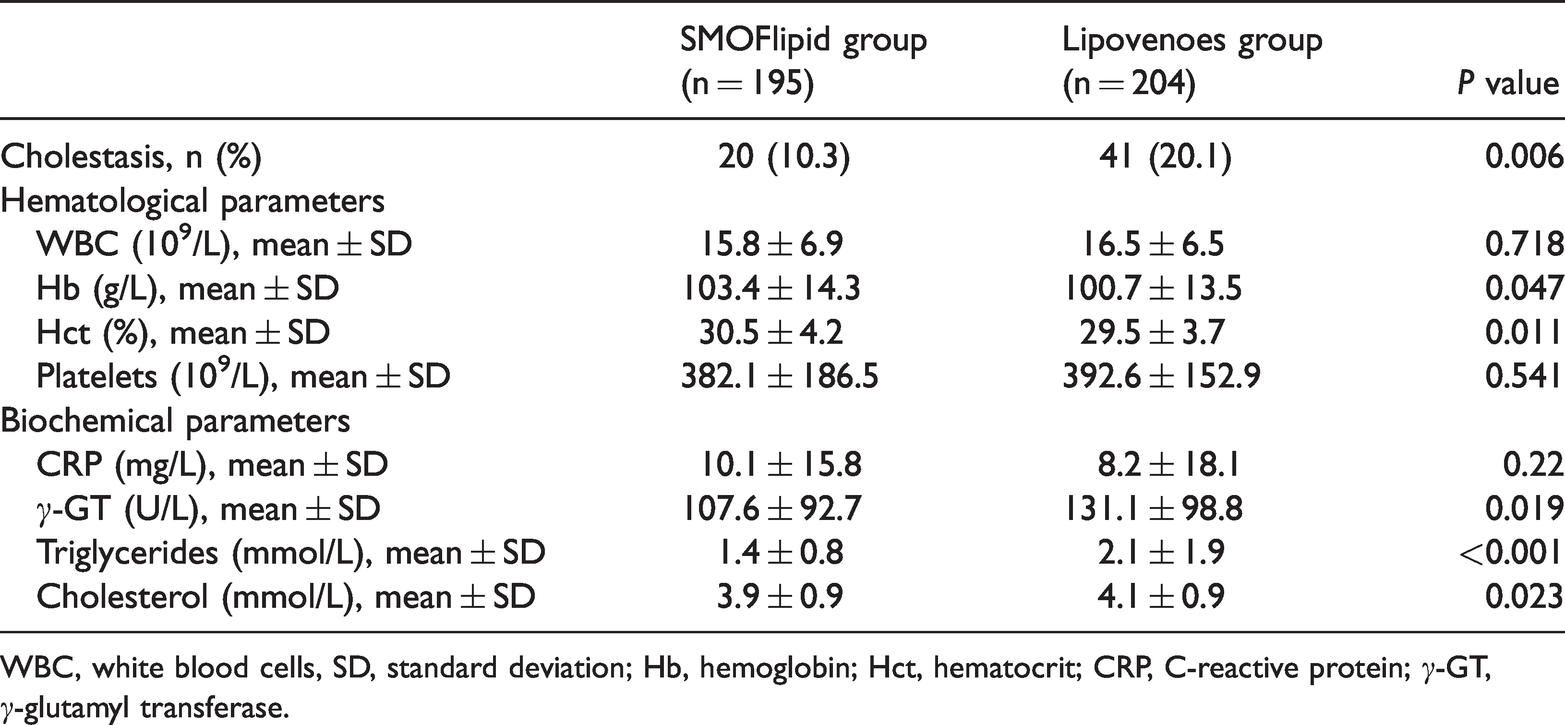
WBC, white blood cells, SD, standard deviation; Hb, hemoglobin; Hct, hematocrit; CRP, C-reactive protein; γ-GT, γ-glutamyl transferase.
Multivariable logistic regression showed that higher alanine aminotransferase levels (odds ratio [OR] 1.012; 95% CI: 1.001–1.022, P = 0.024), lower hematocrit levels (OR 0.964; 95% CI: 0.947–0.982, P < 0.001), hypoalbuminemia (OR 0.353; 95% CI: 0.163–0.767, P = 0.008), and hypoglycemia (OR 0.973; 95% CI: 0.954–0.992, P = 0.005) were significantly associated with cholestasis.
For secondary outcomes (Table 3), ROP was significantly lower in the SMOFlipid group than in the Lipovenoes group (P < 0.001). The incidence of BPD was slightly lower in the SMOFlipid group than in the Lipovenoes group (P = 0.046), but there was no difference in severe IVH, NEC, or mortality between the groups.
Secondary outcomes.

ROP, retinopathy of prematurity; IVH, intraventricular hemorrhage; NEC, necrotizing enterocolitis; BPD, bronchopulmonary dysplasia.
Discussion
PN-associated cholestasis is the most concerning morbidity in preterm infants with long-term use of PN. In this retrospective cohort study, a mixed lipid emulsion composed of soybean oil, MCTs, olive oil, and fish oil significantly reduced the incidence of PN-associated cholestasis compared with a soybean oil-based lipid emulsion. The large sample size analyzed in our study may have been sufficient to prove a clinically meaningful effect. The preterm infants in our study received intravenous lipid emulsion as early as < 24 hours after birth. In the present study, we recruited preterm infants in a single center, thereby reducing possible bias by using one multidisciplinary team and a consistent protocol. Furthermore, we found that SMOFlipid provided a beneficial effect by decreasing the duration to full enteral feeding days and preventing relevant complications, such as ROP and BPD.
The etiology of PN-associated cholestasis is multifactorial, with causes including prematurity, lack of enteral nutrition, sepsis, and factors relevant to the duration and composition of PN.24–27 Numerous adult studies have shown that fish oil containing lipid emulsion has a positive effect on liver enzyme tests and prevention of PN-associated liver disease in surgical or critically ill patients.28–30 Three studies showed a 15% to 25% faster plasma lipid clearance or 25% to 45% shorter half-life of experimental fish oil containing lipid emulsion, even with different fish oil content.31–33 Liang et al. 34 showed that fish oil-enriched PN produced a reduction in IL-6 levels, a high CD4+/CD8+ ratio, and higher CD3+ and CD4+ lymphocytes compared with soybean oil of PN in patients undergoing radical colorectal cancer resection. These findings assumed that supplementation of EPA and DHA by the parenteral route may modulate immunocompetent cells in seriously ill surgical patients. Socha et al. 35 reported that infants with biliary atresia after surgery and intrahepatic cholestasis receiving a long-chain PUFA-supplemented formula had higher levels of DHA and arachidonic acid (AA). Cholestasis status improved in these infants receiving long-chain PUFA supplementation. However, the number of cases in their study was small, and no relevant studies have supported these findings.
Retrospective animal research has suggested that lipid emulsions containing high concentrations of ω-6 LC-PUFAs and phytosterols, which produce proinflammatory prostaglandin E2 and thromboxane A2 mediated by cyclooxygenase pathways, may induce an immune response and promote hepatic injury. 36 In contrast, fish oil containing lipid emulsions that are rich in EPA and DHA might reduce an inflammatory reaction and improve liver function. Additionally, PN containing fish oil increases gene expression of canalicular bile acid transporters, resulting in greater secretion of hydrophobic bile acids and increased bile flow in neonatal piglets37,38 and rats. 39 These alterations may prevent cholestasis and explain the hepatoprotective effects of fish oil containing lipid emulsion. Pscheidl et al. 40 also reported that a parenteral diet supplemented with fish oil normalized nutritive blood flow to the gut, ameliorated bactericidal defense of the splanchnic region, and advanced killing of translocated bacteria in a low-dose endotoxin rat model. These findings are consistent our finding of a decreased number of full feeding enteral days in the SMOFlipid group.
Repa et al. 41 recently found that SMOFlipid did not significantly reduce the incidence of PN-associated cholestasis in extremely low birth weight infants. In that study, the intervention started within the first 120 hours of life, which is different from our study’s intervention beginning within 24 hours of birth. However, those authors’ study was powered to detect a reduction of PN-associated cholestasis from 25% to 10%. They found that the incidence of PN-associated cholestasis was 16% in the soybean oil-based lipid emulsion group and 10% in the SMOFlipid group. The outcome of cholestasis was lower using SMOFlipid compared with soybean oil-based lipid emulsion, but the small population size and the low incidence of cholestasis limited the statistical significance of the difference in Repa et al.’s study. 41
We also found that the incidence of BPD and ROP was lower in the SMOFlipid group than in the Lipovenoes group. Recent evidence indicates the importance of inflammation in the pathogenesis of BPD and ROP.11,17 A higher ratio of ω-3 to ω-6 LC-PUFAs might improve the balance of pro- and anti-inflammatory mechanisms. In preterm infants, early supplementation of fish oil containing lipid emulsion significantly decreases IL-1β and IL-6 levels in serum and bronchoalveolar lavage fluid, and is associated with a shorter duration of ventilator support and less BPD.11,42 Additionally, several studies have shown that DHA and EPA, which is a metabolic precursor of DHA, may protect the retina against ischemia, hypoxia, and hyperoxia through anti-inflammatory action. 43 The metabolites of ω-3 LC-PUFAs decrease vascular endothelial growth factor levels, promote insulin-like growth factor-1 secretion, reduce neovascularization, and normalize retinal layers in rats.44,45 These findings suggest that an anti-inflammatory effect is associated with a lower incidence of BPD and ROP, as well as PN-associated cholestasis.
Nevertheless, SMOFlipid did not reduce the risks of other morbidities, such as sepsis, NEC, severe IVH, or mortality in our study. Although much is known about the mechanism of action of ω-3 LC-PUFAs and their effect on cytokines and inflammatory mediators from research with animal models, their exact clinical effects in preterm infants are still unclear. Preterm infants are a heterogeneous population and may present with a wide variety of severe medical illnesses. Several review studies have reported that there are insufficient high-quality data to demonstrate any differences in the rate of sepsis or infection after administration of fish oil-containing lipid emulsion.46,47 Najm et al. 48 noted that there is a large deficit of DHA and AA in preterm infants, and considered that studies on more prolonged or different ratios of ω-3 and ω-6 LC-PUFAs administration are warranted.
A limitation of our study is that it was a retrospective cohort study, not a randomized, controlled trial. Despite having the same multidisciplinary team and using a consistent protocol, there were some factors that could not be controlled. We also did not quantitate the relative effect of DHA and AA levels in red blood cells to prove a direct correlation, and did not measure DHA and AA levels in breast milk to assess the effect of dietary intake. A well-designed, randomized, controlled trial is required to evaluate the long-term effects of mixed lipid emulsion on growth, neurodevelopmental, and visual outcomes in very low birth weight infants.
Conclusions
Parenteral nutrition of SMOFlipid is safe and well tolerated. SMOFlipid shows a potential beneficial effect on cholestasis and anti-inflammation because of the relatively low ω-6: ω-3 ratio in premature infants requiring PN. This may be associated with a reduction in the incidence of BPD and ROP. Additionally, SMOFlipid is associated with a shorter duration of parenteral nutrition and time to full enteral feeding. However, SMOFlipid does not affect other morbidities and mortality. Randomized, controlled trials are required to prove the effect of different fish oil-based lipid emulsions on prevention of PN-associated cholestasis.
Footnotes
Acknowledgements
The authors thank the staff at the Epidemiology and Biostatistics Center, Department of Changhua Christian Hospital for statistical assistance. The authors are grateful to the infants and their parents, as well as to all of the staff in the neonatal intensive care unit of Changhua Christian Children’s Hospital for their assistance.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
