Abstract
Patients with essential thrombocythemia (ET) can experience hemorrhagic or ischemic vascular events. The prevention of these complications is challenging, and the overall risk of vascular events caused by ET is often overlooked. A 34-year-old man was admitted for a 10-day history of weakness and numbness in his right limbs. He had been diagnosed with ET in 2008 but had stopped receiving treatment half a year before admission. Physical examination showed a superficial sense of disturbance in the right limbs and decreased muscle strength in the right upper and lower limbs (4/5). His platelet count (459 × 109/L) was elevated. Magnetic resonance imaging showed acute watershed infarction, and he was treated successfully. However, he was readmitted for headache and left limb weakness 14 months later. A head computed tomography scan revealed spontaneous subdural hemorrhage. He underwent subdural hematoma removal and decompressive craniectomy. Surgery and pathological investigation revealed no venous sinus thrombosis or vascular malformation. His condition improved, and he exhibited a stable condition 1 year after discharge. Successive development of ischemic stroke and spontaneous subdural hemorrhage is rare in a patient with ET. This case suggests that ET is not only a risk factor for stroke but can also cause highly heterogeneous strokes.
Introduction
Patients with essential thrombocythemia (ET) can experience hemorrhagic or ischemic vascular events. Prevention of these complications, which is essential for a good prognosis, is a complex problem. The overall risk assessment of vascular events caused by ET is often overlooked. ET is characterized by megakaryocytic hyperplasia with subsequent thrombocytosis sustained in the peripheral blood, thereby increasing the risk of thrombo-hemorrhagic phenomena. Approximately 2% of patients with ET may develop arterial embolism. 1 The overall risks of bleeding and thrombosis are 0.33% and 6.6% per patient per year, respectively, in patients with ET. 2 It is rare to encounter a patient with ET experiencing both types of stroke sequentially. We present the case of a patient with ET who had an ischemic stroke and a hemorrhagic stroke in successive order. We also present a review of the literature. This report is aimed to increase awareness that patients with ET can be at risk for both stroke types.
Case report
In December 2017, a 34-year-old man was admitted to the hospital for weakness and numbness of the right limbs, which he had been experiencing for the past 10 days, the intensity of which had increased over the 3 days before admission. No obvious abnormality had been found on a digital subtraction angiogram 5 days before admission. In 2008, he was diagnosed with ET at the Hematological Specialist Hospital. At that time, the patient had undergone bone marrow biopsy, and his doctors had excluded chronic myeloproliferative disease and secondary thrombocytosis. He was treated with interferon-α, which he had discontinued for personal reasons 6 months before his recent admission. His medical history revealed multiple meningiomas, which were treated in 2015. He had no history of hypertension, infection, diabetes, or hereditary disease. He did not smoke or drink alcohol. A physical examination showed clear consciousness, vibration sense and touch sensory disturbance of the right limbs, and decreased muscle strength of the right upper and lower limbs (4/5). Laboratory analysis showed that his platelet count (459 × 109/L, reference range 100–300 × 109/L) and activated partial thromboplastin time (35.0 s) were elevated, as were his total bilirubin (25.7 µmol/L), uric acid (448 µmol/L), and triglyceride (4.05 µmol/L) levels. The red blood cell count, hemoglobin level, C-reactive protein level, and fasting blood glucose level were normal. A thromboelastogram, chest radiograph, 24-hour dynamic electrocardiogram, echocardiogram, and Doppler sonogram of the carotid artery showed no obvious abnormalities. Magnetic resonance (MR) imaging (MRI) indicated acute infarction (Figure 1a), enhanced MRI revealed multiple meningiomas (Figure 1b), and a brain MR angiogram indicated no obvious abnormalities (Figure 1c). The patient was administered intravenous Ginkgo biloba extract (35 mg/day), acetylsalicylic acid (100 mg/day), and atorvastatin calcium (20 mg/day), and his symptoms gradually improved. Six months after discharge, he had recovered completely and had stopped taking anti-platelet drugs and interferon-α. He was readmitted for headache and left limb weakness for 2 days in February 2019. He denied having a history of trauma. A physical examination showed muscle strength of 3/5 in the left upper and lower limbs and no other obvious positive neurological signs. An emergency head computed tomography (CT) scan revealed subdural hemorrhage (Figure 1d). Laboratory analysis showed that the patient’s platelet count (542 × 109/L) was elevated, as were the activated partial thromboplastin time (41.8 s), alanine aminotransferase level (107 U/L), C-reactive protein level (11 mg/L), and fibrinogen level (5.22 g/L). His plasma D-dimer level was in the normal range, and a chest CT scan showed splenomegaly. Enhanced MRI (Figure 1e) and MR susceptibility weighted imaging (Figure 1f) confirmed a subdural hematoma. An MR venogram (Figure 1g) and MR angiogram (Figure 1h) showed no obvious abnormalities. He underwent removal of the subdural hematoma and decompressive craniectomy because his condition worsened on day 3 post-hospitalization. No venous sinus thrombosis or vascular malformation was found during surgery (Figure 1i) or later during the pathological investigation of his lesions. His condition gradually improved after surgery. Consciousness and limb function recovered to a normal level 1 month after surgery. He continued receiving interferon-α treatment after discharge. At 1 year after discharge, the patient exhibited a stable condition. We have de-identified the details such that the identity of the patient may not be ascertained in any way, and the patient provided verbal consent for inclusion in this study. Our study complies with the CARE Checklist (2013).

a. Diffusion-weighted magnetic resonance imaging (MRI) shows acute infarction at the junction (arrow) of the temporal and occipital lobes. b. Sagittal gadolinium-enhanced MRI shows multiple meningiomas (arrows). c. A brain magnetic resonance angiogram shows no obvious abnormality of the cerebral artery. d. A head computed tomography scan reveals a subdural arc-shaped lesion (arrows) with slightly higher density and right brain swelling, showing a midline shift and compression of the right lateral ventricle. e. Coronal gadolinium-enhanced MRI shows abnormal enhancement of the signal in the right subdural region (arrow) with an occupying effect, leading to a midline shift (arrows). f. Magnetic resonance (MR) susceptibility weighted imaging shows many signals indicating iron deposition on the right brain surface (arrows). g–h. An MR venogram (g) and an MR angiogram (h) show no obvious abnormality. i. Superficial filling of the cerebral veins (arrows) is obvious, and no vascular malformation was found after removing the bone flap and subdural hematoma during surgery.
Discussion
The occurrence of ischemic stroke and spontaneous cerebral hemorrhage successively in a young patient with ET has rarely been reported. This case suggests that ET is not only a risk factor for stroke but can also cause highly heterogeneous strokes. When caring for ischemic stroke, hemorrhagic strokes cannot be overlooked in the prevention of complications of ET. Watershed infarction is a common stroke subtype caused by ET. ET patients with a history of thrombosis are at high risk of stroke and may require more than two types of drugs for ET treatment.
Watershed infarction, as observed in this case, is one of the most common stroke subtypes caused by ET 3 , 4 along with lacunar infarction, 5 punctate subcortical infarction, 6 and giant infarction. 7 Cerebral ischemia is the most common ischemic manifestation in ET cases. 8 Platelets play an important role in the process of thrombus formation (Figure 2).
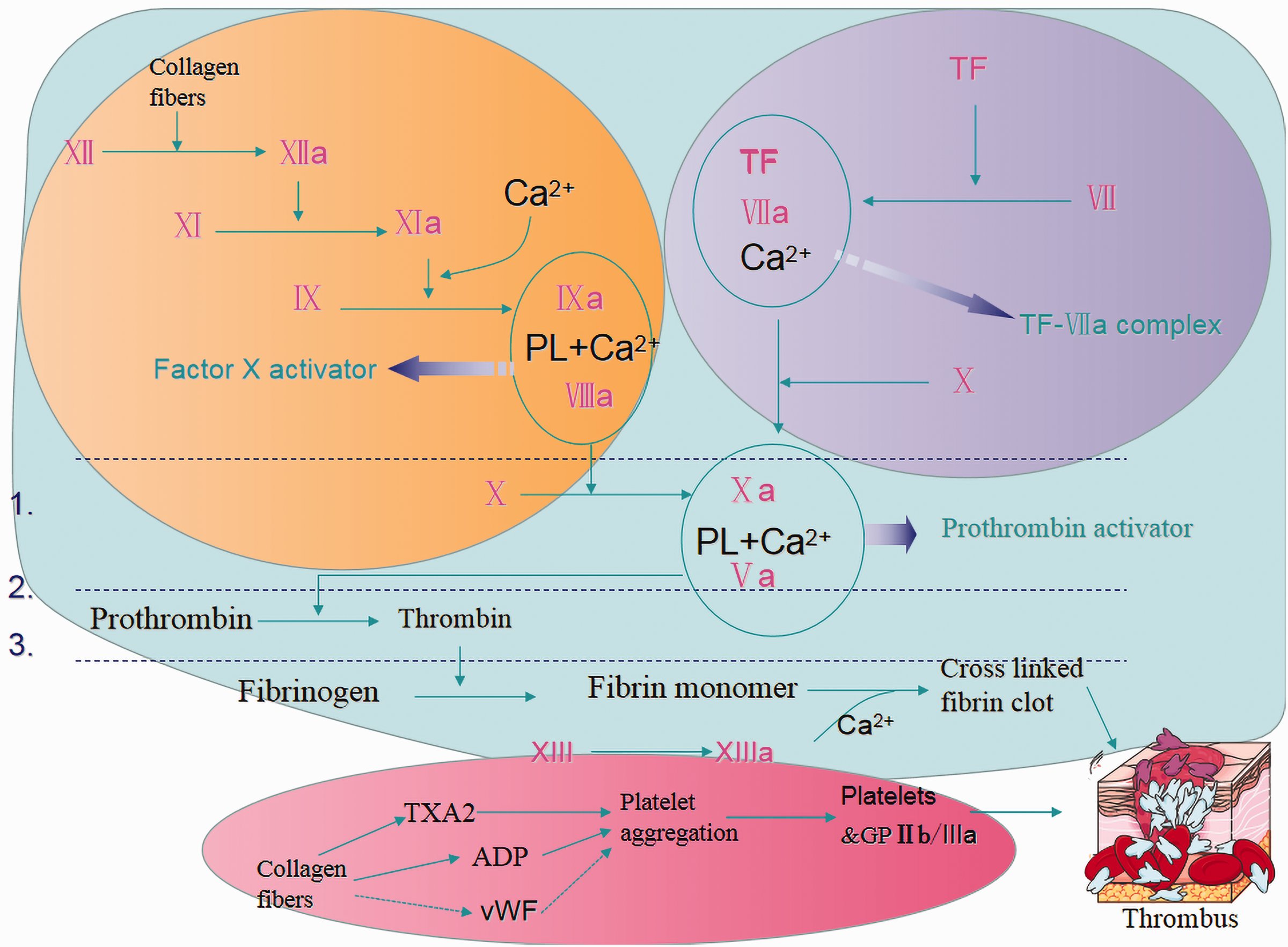
(Graphical Abstract) The role and function of platelets in the process of thrombus formation. The figure shows platelet activation (red circle) and the extrinsic (purple) and intrinsic (yellow) coagulation pathways. The three parts work together to eventually lead to thrombus formation.
ET-related bleeding is often mucocutaneous, and the most severe bleeding event is intracerebral bleeding, 9 including subdural hematoma, 10 as noted in this case. Spontaneous subdural hematomas are uncommon in patients with ET. A search of PubMed revealed only one similar case report. 10 The main cause of death in patients with ET is hemorrhage or thrombosis of vital organs.
A normal platelet count is associated with a reduction in the occurrence of vascular events. However, stroke risk may not necessarily be proportional to the degree of increase in the platelet count, as observed in this case. ET patients with a history of thrombosis are at high risk of stroke, and a history of thrombosis was shown to be independently predictive of future thrombotic events. 11 Cytoreductive therapy, including interferon-α and hydroxyurea, is recommended in high-risk patients. High-risk patients with ET should be treated with more than two types of drugs (dual anti-platelet therapy and a combination of anti-platelet drugs and anticoagulation/chemotherapeutic drugs; Table 1). Moreover, dual anti-platelet therapy is superior to individual anti-platelet therapy in preventing cardiovascular events. 12 The presence of immature platelets serves as a marker of a poor response to anti-platelet treatment. 12 Patients with ET often have splenomegaly, as was observed in this case; however, a splenectomy tends to aggravate the condition.
Clinical characteristics of cerebral vascular events related to thrombocythemia.
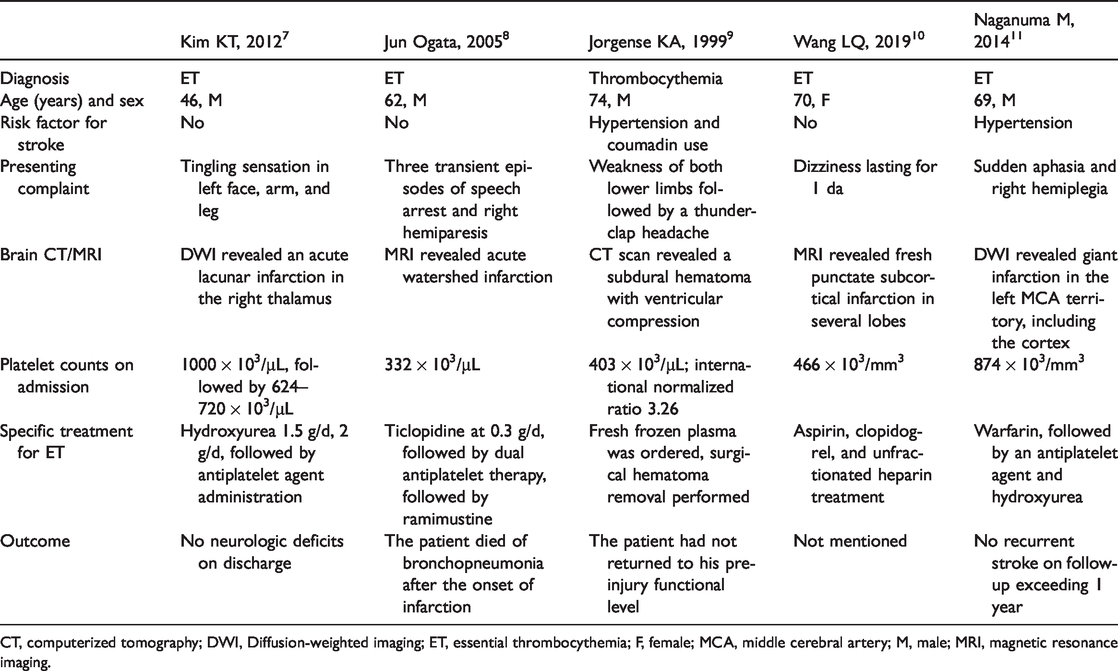
CT, computerized tomography; DWI, Diffusion-weighted imaging; ET, essential thrombocythemia; F, female; MCA, middle cerebral artery; M, male; MRI, magnetic resonance imaging.
Aspirin and interferon-α were applied for our patient based on the following guidelines: interferon-α is preferred in young individuals, 13 and interferons are included as options for symptomatic patients. 14 The platelet count needs to be reduced by platelet-lowering treatment in patients who have a history of major thrombosis or major bleeding. 15 For patients younger than 40 years of age who are candidates for platelet-lowering therapy and are either male or female with no childbearing potential, the first-line therapy should be interferon (grade D). 15 Aspirin is recommended for the primary treatment of acute ischemic stroke according to stroke guidelines. 16 Anti-platelet therapy is also recommended for ET patients with a recent ischemic stroke, and the appropriate anti-platelet therapy is aspirin. 15 The traditional therapy for ET is the prevention of thrombotic events with antiproliferative agents in association with aspirin, and pegylated interferon is a candidate drug for slowing the evolution to myelofibrosis or leukemia. 17
This case is significant because a patient with ET developed ischemic stroke and hemorrhagic stroke successively. We searched PubMed for the medical subject heading terms “Brain Infarction/Hematoma, Subdural” and “Thrombocythemia/Thrombocythemia, Essential” in different combinations but found no similar cases. Unfortunately, because the patient had been diagnosed with ET in a local hospital a long distance away 12 years prior, we were unable to obtain a bone marrow biopsy specimen or the JAK2V617F test results. Although we were unable to find any cause of stroke other than ET in the patient, we cannot rule out unknown causes of stroke (e.g., patent foramen ovale).
Conclusion
A patient with ET developed successive ischemic and hemorrhagic strokes. This rare case indicates that ET is not only a risk factor of stroke but can also cause highly heterogeneous strokes. Watershed infarction is a common stroke subtype caused by ET. ET patients with a history of thrombosis are at high risk of stroke and may require more than two types of drugs for ET treatment.
Footnotes
Ethics approval
The study design was approved by the ethics review board of the Third Affiliated Hospital of Shenzhen University (No: 2020SZLH-LW-013).
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
