Abstract
Objective
To assess the safety and efficacy of prostatic arterial embolization (PAE) for elderly patients with lower urinary tract symptoms secondary to large benign prostatic hyperplasia.
Methods
Twenty-eight patients (>80 years of age) with prostate volume >80 mL were enrolled from October 2016 to October 2019. PAE was performed using microspheres and functional results were evaluated at 1, 3, 6, and 12 months postoperatively. The following data were recorded: International Prostate Symptom Score (IPSS), quality of life (QoL), maximum urine flow rate (Qmax), post-void residual urine volume, prostate volume and total prostate-specific antigen level.
Results
Selective prostatic arterial catheterization and embolization were achieved in 27 of 28 patients. Follow-up data were available for those 27 patients until 12 months postoperatively. Significant improvements were found at all postoperative time points in terms of the mean IPSS, mean QoL score, mean Qmax, mean post-void residual urine volume, mean total prostate-specific antigen level, and mean prostate volume. The overall complication rate was 46.4%.
Conclusions
PAE is an efficacious and safe treatment for elderly patients with large prostate volume; it may offer an effective approach for patients who are not candidates for open or endoscopic surgical procedures because of comorbidities.
Keywords
Introduction
Benign prostatic hyperplasia (BPH) is a common public health concern that affects older men. 1 Although it usually is not life-threatening, BPH has been associated with reduced quality of life and constitutes a substantial burden to the healthcare system. Furthermore, global life expectancy has increased in recent decades, such that a growing number of elderly men require better treatment for BPH. 1 The European Association of Urology recommends transurethral resection of the prostate and laser techniques for the surgical treatment of BPH, although these procedures carry some risks, especially in patients older than 80 years of age with large prostate volume (>80 mL). 2 Moreover, some elderly patients are not suitable for open or endoscopic surgical procedures because of comorbidities. Therefore, various minimally invasive techniques have been proposed to reduce patient morbidity.3–5
Prostatic arterial embolization (PAE) is an interventional radiological technique that involves the injection of small particles directly into bilateral prostatic arteries, which causes devascularization of hypervascular nodules. 6 It has been identified as an effective and safe procedure for the treatment of BPH following its initial clinical implementation in 2010. 7 However, to the best of our knowledge, treatment of large prostate volume (>80 mL) in elderly patients (≥80 years of age) using PAE has not been adequately investigated. In this study, we aimed to assess the safety and efficacy of PAE as a primary treatment for elderly patients (≥80 years of age) with large prostate volume (>80 mL) after failed drug treatment. Importantly, our patients were not suitable candidates for open or endoscopic surgical procedures because of comorbidities.
Materials and methods
Study population and patient evaluation
The study protocol was approved by the local ethics committee of the Ninth People’s Hospital of Suzhou City (approval number: 17-01). Elderly male patients (≥80 years of age) were enrolled in this study from October 2016 to October 2019. The inclusion criteria included severe lower urinary tract symptoms (LUTS; International Prostate Symptom Score [IPSS] ≥25, quality of life score [QoL] ≥4, and maximum urine flow rate [Qmax] ≤8 mL/s) because of BPH after failure of at least 3 months of drug therapy, as well as a prostate volume >80 mL on trans-rectal B-ultrasound or magnetic resonance imaging (MRI). The exclusion criteria included urethral stricture, bladder neck contracture, bladder stones, active urinary tract infection, neurogenic bladder, detrusor failure, and prostate cancer. Patients underwent ultrasound-guided prostate biopsy prior to study inclusion if they had both a total prostate-specific antigen (PSA) level >4 ng/mL and free PSA/total PSA ratio <0.16, a total PSA level >10 ng/mL (regardless of free PSA/total PSA ratio), or abnormal findings during digital rectal examination or iconography. Patients were included in the study if their pathology results were negative. Patient selection was performed in a multidisciplinary manner by a single urologist (MJ) and a single interventional radiologist (CZ). Upon assessment by the same urologist (MJ) and an anesthesiologist, all patients in this study were determined not to be suitable candidates for open or endoscopic surgical procedures because of cardiopulmonary insufficiency. All included patients provided written informed consent for surgery and inclusion in the study, prior to the PAE operation.
Routine preoperative diagnostic procedures (detailed medical history, physical examination, blood tests, urine tests, and sterile urine culture) were performed for all patients. Computed tomography angiography of the hypogastrium and MRI were used for all enrolled patients to assess the statuses of prostatic arteries. Prostate-related characteristics were evaluated using MRI and B-ultrasound. Data analysis included demographic characteristics, prostate-related characteristics, surgical details, and perioperative outcomes.
PAE technique
The PAE procedure was performed under local anesthesia by a single senior interventional radiologist (CZ). Patients underwent angiography and PAE in a specialized angiography unit equipped with a digital flat-panel detector system (Artis Zee; Siemens Healthcare, Erlangen, Germany) with non-ionic contrast medium (iohexol 300 mg I/mL; GE Healthcare, Chicago, IL, USA). Embolization was usually performed by a right femoral approach; the femoral artery was cannulated with a 5-Fr vascular sheath (Radifocus; Terumo, Tokyo, Japan) using Seldinger’s technique. Selective digital subtraction angiography was performed with a 5-Fr cobra catheter (Terumo) to evaluate the iliac artery by using the ipsilateral anterior oblique projection. The prostatic artery was identified by digital subtraction angiography; super-selective catheterization was then carried out with a coaxial 2.7-Fr microcatheter (Progreat 2.7; Terumo). Following confirmation of the embolization position, slow flow injection of microspheres (100–300 µm; Merit Medical, South Jordan, UT, USA) was performed with a complete occlusion endpoint (Figure 1a, b). The PAE procedure was generally performed bilaterally, but was performed unilaterally in the event of failed catheterization of one side. A urinary catheter was routinely inserted during the PAE procedure and was removed at 2 weeks postoperatively. All patients were hospitalized for 1 to 3 days for observation and appropriate hydration was administered for 2 days postoperatively. Antibiotics were administered to prevent infection in all patients.

(a) Angiography before/after super-selective catheterization of the right prostatic artery; (b) angiography before/after super-selective catheterization of the left prostatic artery.
Outcome measures
Safety was evaluated in terms of perioperative data (e.g., operative time, fluoroscopy time, changes in hemoglobin level within 24 hours postoperatively, hospitalization days, postoperative duration, and overall cost) and complications. The complications were graded using the Clavien–Dindo classification system. 8
Functional results (e.g., prostate volume, total PSA level, IPSS, QoL score, Qmax, and post-void residual urine volume [PVR]) were used to assess PAE efficacy. Follow-up of functional results including patient-reported complications was performed at 1, 3, 6, and 12 months after the PAE procedure. The prostate volume was measured by MRI and images were independently assessed by a single senior radiologist (JW), while PVR was measured by B-ultrasound by a single senior radiologist (GZ); both radiologists were unaware of the outcomes of PAE during imaging assessments. The prostate volume was determined using the ellipsoid formula: length × width × height × 0.523.
Technical success was defined as selective prostatic arterial catheterization and embolization on at least one side of the pelvis (i.e., unilateral or bilateral PAE). 9 Clinical failure after the PAE procedure was recorded when at least one of the following criteria was met: IPSS ≥18, QoL score ≥4, Qmax increase ≤2.5 mL/s, and Qmax ≤7 mL/s. 10
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics, version 20.0 (IBM Corp., Armonk, NY, USA). Quantitative data were expressed as mean ± standard deviation (range), whereas qualitative variables were expressed as numbers and percentages. Statistical analysis was carried out using the chi-squared test for categorical variables and Student’s t-test for continuous variables. A P-value <0.05 was considered statistically significant.
Results
Twenty-eight patients with severe LUTS were enrolled in this study. Technical PAE success was achieved in 27 patients (96.4%). PAE failure occurred in one patient (3.6%) because of tortuosity and atherosclerotic changes of in the bilateral iliac arteries. Bilateral PAE was performed in 25 patients (92.6%), while the remaining two patients underwent unilateral PAE because of severe atherosclerotic stenosis of a unilateral prostatic artery. Patients were followed for at least 12 months. Only one patient (the patient with PAE failure) was lost to follow-up at 3 months; the remaining 27 patients completed all postoperative follow-up visits. The baseline characteristics of the patients who underwent PAE are shown in Table 1.
Baseline characteristics of patients who underwent prostatic arterial embolization.

Data are shown as mean ± standard deviation or n (%).
The perioperative outcomes are shown in Table 2. Notably, the mean hemoglobin level was significantly reduced after the PAE procedure (P < 0.05), but it remained acceptable (i.e., no patients required blood transfusion or experienced any discomfort). Table 3 shows the intraoperative and postoperative complications of patients who underwent PAE. Of the 28 patients, 12 had minor complications (42.9%) and one had a major adverse event (technical failure) (3.6%). Minor complications included post-embolization syndrome, hematuria, and urinary tract infection; all were cured within 1 week of specific treatment. Acute urinary retention after the PAE procedure was successfully resolved by temporary bladder catheter placement for 7 days.
Perioperative outcomes of patients who underwent prostatic arterial embolization.

Data are shown as mean ± standard deviation or n (%).
Intraoperative and postoperative complications of patients who underwent prostatic arterial embolization.
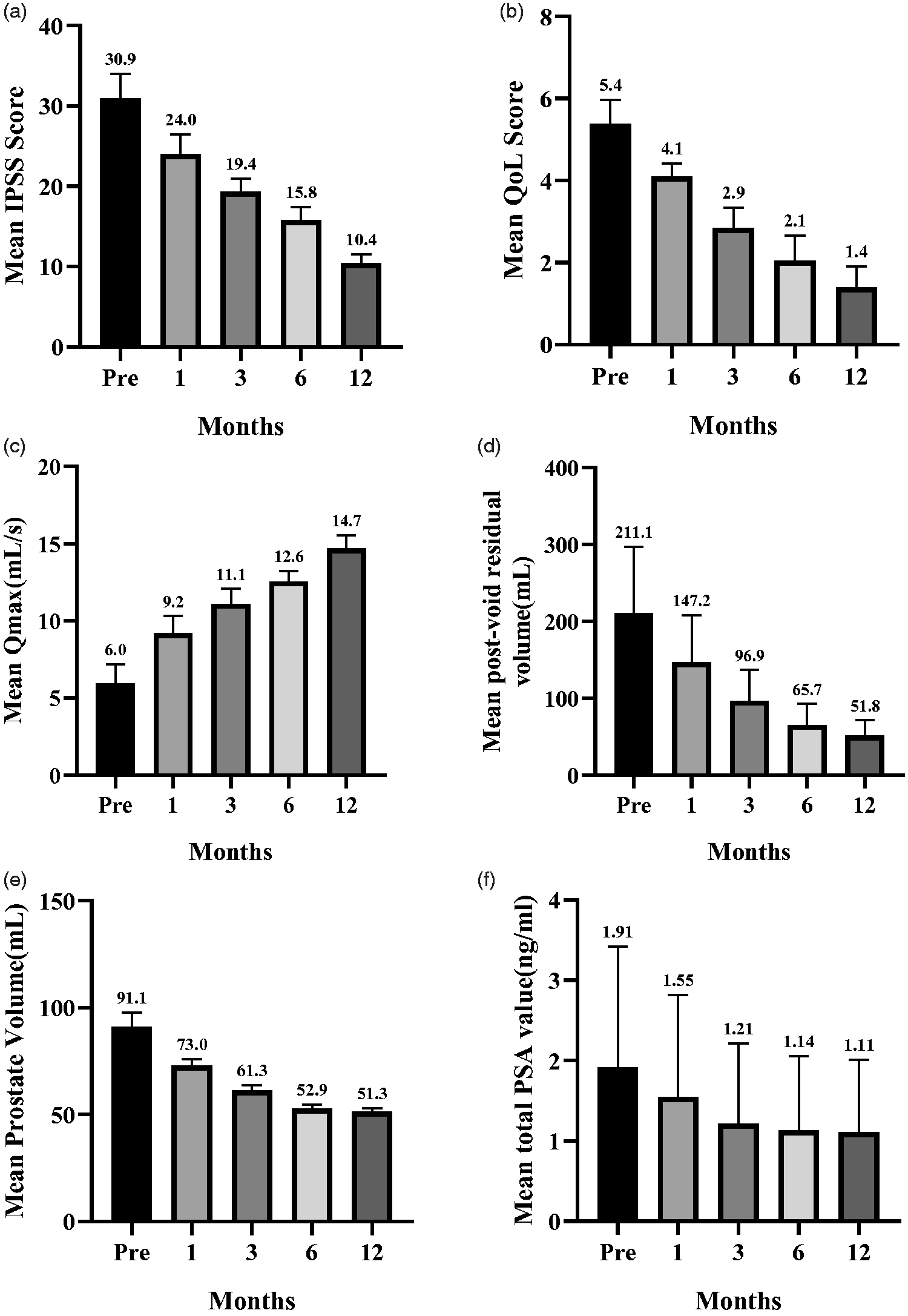
In patients with technical PAE success, the mean IPSS, QoL score, Qmax, and PVR were significantly improved at 12 months postoperatively (Figure 2a–d). The mean IPSS decreased from 30.9 ± 3.2 points to 10.4 ± 1.1 points (P < 0.05), mean QoL score decreased from 5.4 ± 0.6 points to 1.4 ± 0.5 points (P < 0.05), mean Qmax increased from 6.0 ± 1.3 to 14.7 ± 0.9 mL/s (P < 0.05), and mean PVR decreased from 211.1 ± 88.2 mL to 51.8 ± 19.9 mL (P < 0.05). Furthermore, the mean prostatic volume significantly decreased from 90.1 ± 6.7 mL to 51.3 ± 1.9 mL (mean reduction of 43.7%, P < 0.05, Figure 3a, b) and the mean total PSA level also significantly decreased from 1.9 ± 1.5 points to 1.1 ± 0.9 points (P < 0.05) (Figure 2e, f).

Comparisons of the following parameters from the preoperative assessment (0 months) through 1, 3, 6, and 12 months postoperatively: (a) mean International Prostate Symptom Score (IPSS); (b) mean quality of life (QoL) score; (c) mean maximum urine flow rate (Qmax); (d) mean post-void residual urine volume; (e) mean prostate volume; (f) and mean total PSA level.
Changes in axial contrast-enhanced T1-weighted magnetic resonance images from a 93-year-old man with lower urinary tract symptoms due to benign prostatic hyperplasia who underwent bilateral prostatic arterial embolization (preoperatively [panel a] and 1 month postoperatively [panel b]). Significant infarction areas were evident on both sides of the prostate (straight arrows) at 1 month postoperatively, with a prostate volume reduction of 19.8%.
Discussion
The treatment of BPH has changed markedly in recent decades. Medical therapy is recommended for patients with mild BPH symptoms (IPSS ≤7) and for patients with moderate or severe symptoms (IPSS >8) who do not exhibit complications. However, surgical treatment for BPH is recommended after failure of medical therapy, especially for patients with moderate or severe LUTS and substantial PVR with gross hematuria or recurrent urinary tract infection. 11 These patients typically undergo endoscopic enucleation or open surgery, which is determined in accordance with their prostate volume. For patients with prostate volume >80 mL, open prostatectomy or endoscopic enucleation of the prostate (e.g., holmium laser or bipolar enucleation of the prostate) is recommended as the primary surgical option. 2 However, these surgeries are associated with substantial morbidity and require lumbar anesthesia or general anesthesia; therefore, many patients ≥80 years of age are not suitable candidates because they have multiple comorbidities. Novel treatment approaches are necessary for such patients. PAE was proposed relatively recently and has been the focus of increasing interest in both radiological and urological communities since its first successful use for BPH with acute urinary retention in 2010. 7
Consistent with the findings of previous investigations,6,12–14 our study showed that PAE is an efficacious treatment for LUTS secondary to large prostate volume (>80 mL) in elderly patients (≥80 years of age) who were not suitable candidates for open or endoscopic surgical procedures; notably, we found that this surgical approach had a favorable safety profile. All six of the monitored functional results were significantly improved at 12 months postoperatively in the present study, similar to the results reported by Gao et al. 9 Furthermore, Lebdai et al. 15 reported a mean reduction in IPSS of 11.9 points at 1 month postoperatively, which persisted at 6 months postoperatively. Pisco et al. 10 described an IPSS improvement of 6.5 points and a Qmax improvement of 3.85 mL/s at 6 months postoperatively. In 2020, Pisco et al. 16 also reported that the improvements in IPSS and QoL score were superior after the PAE procedure, compared with the effects of a sham procedure for BPH. In the present study, only prostate volume, total PSA level, and PVR remained stable at 6 months postoperatively; IPSS, QoL score, and Qmax continued to increase at 6 months postoperatively. We presume that these results may have been related to the slow and complicated histopathologic changes that occur in the prostate after PAE. Specific follow-up intervals are needed and 6 months postoperatively may not be sufficient to determine the real-world effectiveness of the procedure. 17 Thus, PAE is not regarded as an immediate ablative technique. Furthermore, one-quarter of patients in a previous study had no significant improvements of IPSS or Qmax, although technical PAE success had been achieved in some of those patients. 18 We hypothesize that the baseline characteristics of those patients may have been better and some degree of unilateral PAE may have provided minimal symptomatic improvement, compared with bilateral PAE.
The operative and fluoroscopy times in our study were similar to those in several published papers.9,13,19 Although PAE has been presumed to carry some risks of radiation exposure, no radiation exposure-related adverse events were found in the published studies or in our investigation. However, in a patient with a complicated clinical presentation, excessive fluoroscopy time has been reported; notably, that patient developed radio-dermatitis and subsequent skin atrophy at the procedure site. 20 Therefore, additional long-term data might be needed to assess both the short- and long-term effects of radiation exposure from PAE. The length of stay in our study was longer than in several previous studies, both in terms of overall stay and postoperative duration; the majority of patients in previous studies underwent PAE as outpatient or day case procedures. 6 However, the patients in our study usually were admitted a few days preoperatively to undergo routine examinations such as computed tomography or MRI angiography; they then remained hospitalized for 1 to 3 days during postoperative observation. Notably, patients in our study were elderly (age ≥80 years) and had multiple comorbidities. However, total preoperative examinations (e.g., computed tomography or MRI angiography) carry greater costs in the outpatient setting under the current health insurance system in rural China, whereas a portion of the costs may be reimbursed if they are incurred during hospitalization.
In contrast to most published reports, we found a significant difference between preoperative and postoperative hemoglobin levels.9,13 However, we considered this difference to be reasonable because as the mean hemoglobin level decreased from 123.3 ± 6.6 g/L to 118.6 ± 6.7 g/L, which did not constitute a severe health risk. We presumed that this change was related to the routine postoperative hydration, because no instances of severe hemorrhage occurred during the PAE procedure.
The overall cost of the PAE procedure in our study is likely to be acceptable for most patients in rural China. Many other minimally invasive procedures have been developed with advances in surgical technology. Rezūm water vapor therapy has been used in the treatment of BPH; it can be performed under sedation alone and as a day case procedure, which enables cost-effective treatment. However, this therapy is reportedly unsuitable for patients with urinary retention; 21 most patients in our study required the use of a catheter. Jones et al. compared the use of Urolife and PAE for patients with BPH; they recommended the Urolife as an attractive treatment choice for younger patients (i.e., those with suitable prostate anatomy), whereas PAE may be more effective for elderly patients with symptoms refractory to pharmacotherapy. 22
Several studies have reported complication rates of PAE ranging from 40.3% to 78.3%.6,13,23 In the present study, the overall complication rate was 46.4%. One patient exhibited technical failure because of intraoperative tortuosity and stenosis of the prostatic arteries. Some researchers have recommended preoperative computed tomography or MRI angiography assessments to evaluate the features of prostatic and iliac arteries, with the aim of improving the technical success rate.16,24,25 In our patient who exhibited technical failure, preoperative computed tomography angiography had shown narrow prostatic arteries, although the patient had insisted on PAE treatment. Nevertheless, we recommend routine computed tomography or MRI angiography before PAE for most patients. Minor complications of PAE in our study included post-embolization syndrome, hematuria, urinary tract infection, and acute urinary retention. All these minor complications generally disappeared within 1 week after definitive therapy. Fever was common in our patients, presumably because of necrosis within ischemic prostatic tissue. Only two patients exhibited acute urinary retention after PAE, which was relatively less common than in published studies.9,13,26 This might have been because we extended their catheterization to 2 weeks postoperatively, which helped to reduce ischemic edema in the peri-urethral prostatic tissue after PAE. For patients with acute urinary retention after PAE, a temporary bladder catheter was routinely inserted in combination with antibiotic treatment (determined on the basis of urine culture results); decongestant medications were also administered. In our study, the catheter was removed 7 days later and the patients’ symptoms were resolved. These results imply that PAE could serve as effective treatment for patients with BPH who are not suitable candidates for open or endoscopic surgical procedures.
There were several limitations in our study. First, the sample size was small. Second, the postoperative follow-up duration was limited to 12 months; additional follow-up is necessary to assess the safety and efficacy of the PAE procedure. Third, we did not examine radiation doses during PAE, although these data are necessary to fully determine the level of radiation exposure. Published studies have shown that similar durations of fluoroscopy time might provide different radiation doses.9,27 Finally, we did not perform a comparison between treatments for this population. Despite these limitations, we presume that our findings are meaningful and will aid clinicians in the selection of surgical treatment options for patients with BPH.
Conclusion
Our results indicate that PAE is an efficacious and safe treatment for elderly patients (≥80 years of age) with large prostate volume (>80 mL) following failed medical therapy, especially for those who are not suitable candidates for open or endoscopic surgical procedures because of comorbidities. The functional results of PAE were satisfactory in this study, with an acceptable complication rate.
Footnotes
Author contributions
CX: Project development, data collection, data analysis, manuscript writing/editing. GZ: Project development, data collection, manuscript editing. JW: Data collection, project development. CZ: Project development, manuscript editing. MJ: Project development, data collection and management, manuscript editing.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Project of Wujiang City (grant no. wwk201701) to MJ and CZ and the Livelihood Technology Project of Soochow (grant no. SYS2019012) to CX.
