Abstract
Objective
To evaluate the application of treatment planning system (TPS)-assisted large-aperture computed tomography (CT) simulator to percutaneous biopsy.
Methods
This retrospective study enrolled patients that underwent TPS-assisted large-aperture CT simulator-guided percutaneous biopsy from November 2018 to December 2019. Retrospective analyses of puncture accuracy were compared using paired
Results
A total of 38 patients were included in this study. There were no significant differences between the planned and actual puncture depth and angle. Pulmonary puncture was significantly associated with the accuracy of the puncture angle. The diagnostic rate of malignancy was 76% (29 of 38), of which 20 of 25 patients were in the group initially diagnosed with unconfirmed lesions and nine of 13 patients were in the group of treated patients that needed additional pathological analyses. For patients that underwent a pulmonary biopsy, 12 had minor pneumothorax and three suffered needle track bleeding. No other complications were observed. Regression analyses indicated a significant correlation between puncture angle and the incidence of pneumothorax.
Conclusion
TPS-assisted large-aperture CT simulator may improve the percutaneous biopsy procedure by combining the advantages of radiotherapy specialties with computer targeting.
Keywords
Introduction
Pathological results, as the standard for tumour diagnosis, provide vital information for qualitative diagnosis, treatment guidance and prognostic evaluation. Computed tomography (CT)-guided percutaneous biopsy, which has been extensively used in the clinic for pulmonary and abdominal lesions, plays an important role in obtaining tissue samples and making precise pathological diagnoses. Recently, a considerable body of literature has focused on improving the accuracy and efficiency of this puncture process, through the application of the puncture site-down positioning technique, automatic path proposal computation or large-core needles.1–3 Nevertheless, several major issues, such as inaccurate positioning, repeated adjustment of the puncture route and the risk of serious postoperative complications, remain unresolved.4–7
Considering that CT-guided percutaneous biopsy is generally performed by interventional radiologists or a thoracic surgeon, the present research aimed to exploit the advantages of a radiotherapy simulator and a treatment planning system (TPS) in a department of radiation oncology to compensate for technical defects. Since 2018, the clinical application of a TPS-assisted large-aperture CT simulator to percutaneous biopsy has been investigated and the initial experience is summarized in this current study, with the aim of offering new insights into the improvement of puncture operation and biopsy accuracy.
Patients and methods
Patient population
This retrospective study enrolled consecutive patients that underwent TPS-assisted large-aperture CT simulator-guided percutaneous biopsy at the Department of Radiation Oncology, Xiamen Humanity Hospital Fujian Medical University, Xiamen, Fujian Province, China between November 2018 and December 2019. The clinical data were retrospectively analysed. Inclusion criteria were as follows: (i) initially diagnosed with unconfirmed lesions (i.e. lesions were initially detected by imaging but qualitative diagnosis was unclear) or treated patients that needed additional pathology information for treatment guidance; (ii) available puncture path information; (iii) generally good health, Karnofsky Performance Score (KPS) ≥70, tolerable to puncture procedure. Exclusion criteria were as follows: (i) poor general condition or cachexia, KPS < 70, or intolerable to puncture procedure; (ii) coagulation dysfunction with haemorrhagic tendencies; (iii) severe cardiocerebrovascular disease; (iv) severe infection; (v) superficial lesions could be used to obtain tissue specimens without imaging or B ultrasound-guided puncture; (iv) compulsive position due to spinal disease or other reasons.
This study was approved by the Ethics Committee of Xiamen Humanity Hospital Fujian Medical University (no. XMHA2020004) in March 2020 and undertaken in accordance with the Declaration of Helsinki. All patients provided written informed consent before puncture. Verbal consent for publication was obtained from patients whose images are contained in the manuscript. In order to protect their identities, the details of all patients were de-identified.
Puncture procedures
The puncture workflow is shown in Figure 1. The preoperative preparation involved air sterilization of the CT simulator room, confirmation of the puncture supplies and rescue medicines, and measurement of the life indications of each patient. The following equipment was used: a Philips 16-multislice spiral large-aperture CT simulator (Philips Healthcare, Best, the Netherlands); a movable three-dimensional laser positioning system (Philips Healthcare); an immobilization device (vacuum fixation mat or prone board; Klarity, Guangzhou, China); an Eclipse 13.5 planning system (Varian Medical Systems, Palo Alto City, CA, USA); and a Bard 17G coaxial biopsy needle (Bard, Murray Hill, NJ, USA). For the immobilization and CT scan, the optimum position was selected for patients according to the location of each target lesion and a vacuum fixation mat or prone board was applied for fixing the patients in comfortable positions prior to biopsy. Then, location points were selected and spherical metal markers were pasted on the patients’ surface. Cross hatchings were marked following the laser lines and extension lines on the vacuum fixation mat or prone immobilization device were drawn to determine the relative position of the lesion. Location point-centred CT scan was then subsequently performed to a layer thickness of 3 mm and the images were transmitted to the radiotherapy server. For determination of the puncture path, CT images were imported into Varian TPS (Varian Medical Systems) and the origin was set on TPS. To design the optimum puncture path, the maximum cross-section level of the target lesion was taken when possible, avoiding bones and large blood vessels, and to ensure that normal tissue was pierced as infrequently as possible. The puncture target, known as the gross tumour volume (GTV), was delineated by a radiation therapist (X.K.) and a new plan was created by the radiotherapy physicist (Y.L.) according to the planned path. Subsequently, the source skin distance (SSD) was changed to 100 cm (SSD = 100 cm) and the relative coordinates (X, Y and Z values) of the location point and the puncture point were recorded. The puncture direction and depth were also recorded. With the assistance of the laser positioning system, the puncture point based on the relative coordinates was accurately located onto the patient’s body surface. The above operations were checked by two technicians before puncture. Verification of the puncture point involved the puncture points being marked and axial scans were performed to verify the location of each puncture point, avoiding set-up error. For the percutaneous biopsy, following routine sterilization and local anaesthesia administration, a coaxial biopsy needle was punctured into the proximal region of the lesion based on the planned direction and depth (calibrated by laser line and protractor). Additionally, the location of the biopsy needle was verified by CT scan (for adjustment if necessary). Two or three satisfactory specimens were collected in a specimen bottle with formalin and sent for pathological examination. The skin of the puncture region was then disinfected and bound up postoperatively. A CT scan of the puncture site and adjacent areas was performed to confirm whether there were signs of pneumothorax or bleeding. Close observation for 6 h was required for each patient enrolled in the study.

Flow diagram of treatment planning system (TPS)-assisted large-aperture computed tomography (CT) simulator guided percutaneous biopsy.
Outcome measures
The planned and actual puncture direction and depth were recorded for comparison separately. ΔH was defined as the difference between the planned and actual puncture depth; and Δα was defined as the difference between the planned and actual puncture angle. Other indices including puncture time, frequency of needle adjustment, pathological results and complications were recorded.
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp., Armonk, NY, USA). The parameters of puncture accuracy were analysed using a paired
Results
This retrospective analysis included 38 patients (25 males and 13 females) that underwent TPS-assisted large-aperture CT simulator-guided percutaneous biopsy. The median age of the study cohort was 62 years (range, 32–87 years). Percutaneous lung biopsy was performed in 34 patients, abdominal lesion biopsy was performed in three patients (including liver, pancreas and adrenal gland) and there was one patient that underwent a deep supraclavicular lymph node biopsy. Of the lesions, 36 of 38 (95%) were solid. The median maximum diameter of the lesions was 3.8 cm (range, 1.1–10.1 cm). The demographic and clinical characteristics of the patients are presented in Table 1.
Clinical and demographic characteristics of the patients (

Data presented as median (range) or
The puncture time for each patient was within 20 min. The entire cohort achieved a one-time successful biopsy. Thirty-four patients had CT scans four times, while the remaining four patients underwent one additional CT scan due to slight deviation adjustments. The actual puncture depths were close to the planned puncture depths and there was no significant difference between the mean ± SD planned and actual puncture depths (5.34 ± 2.01 cm versus 5.28 ± 1.95 cm, respectively) (Table 2). The mean ± SD deviation of the puncture depth ΔH was 0.37 ± 0.32 cm, which indicated the precision of the puncture depth. With regard to the puncture angle, there was no significant difference in the median planned and actual angles, which were 90° and 88°, respectively, demonstrating the high reproducibility of this technique.
Puncture accuracy in patients (

Data presented as mean ± SD or median.
No significant between group differences (
Data presented in Table 3 show that the deviations in the puncture depth and angle were well controlled regardless of the size and location of the lesions. Although no significant differences were detected in the puncture accuracy between the different lesion size subgroups, ΔH decreased with increasing lesion size. In addition, Δα in the superior lobe of the lung was significantly smaller than that in the middle or lower lobes (χ2 = −2.129,
Puncture accuracy in patients (

Data presented as mean ± SD.
aWilcoxon rank sum test; NS, no significant between group differences (

Representative computed tomography scans of an example of the biopsy planning. (a) A plan was created using the treatment planning system to design an optimum puncture path. The relative coordinates (X, Y and Z values) were generated automatically when the puncture point was determined. (b) The position of the biopsy needle was verified. GTV, gross tumour volume (i.e. puncture target).
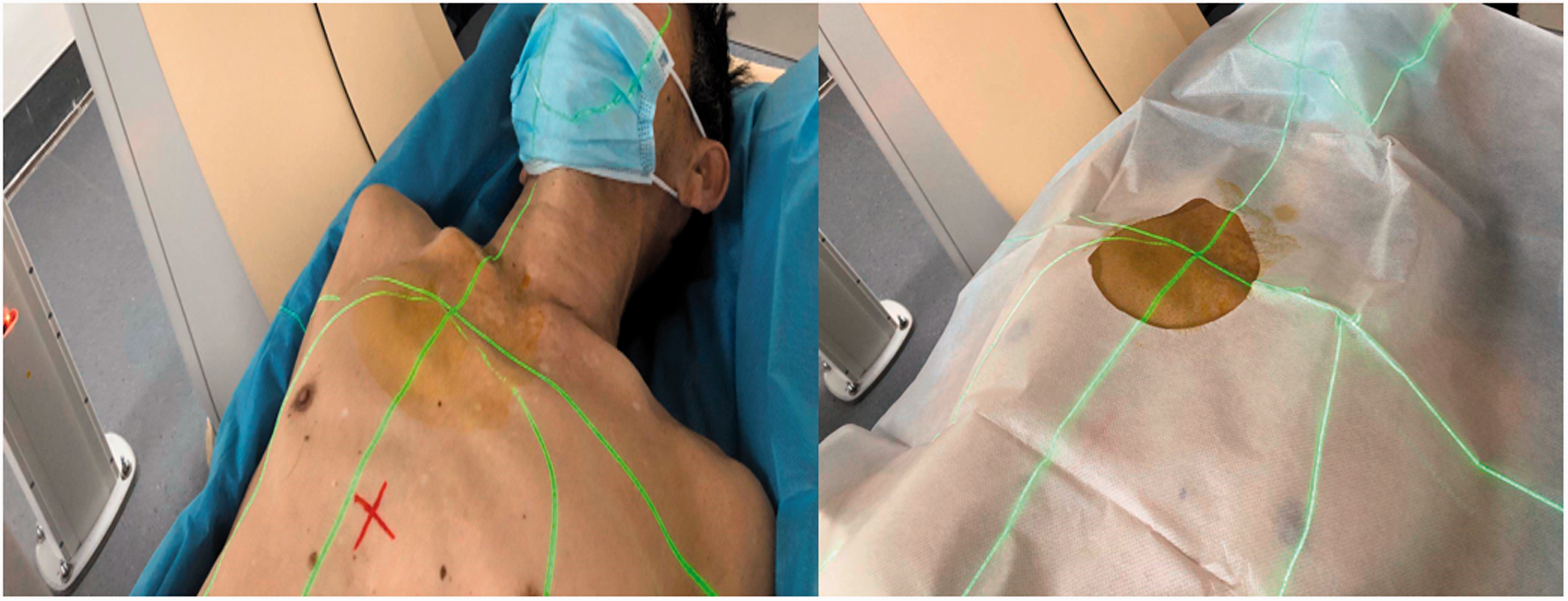
Representative photographs showing the patient positioning. With the help of the laser positioning system, a puncture point based on the relative coordinates was accurately located onto the body surface of the patient.
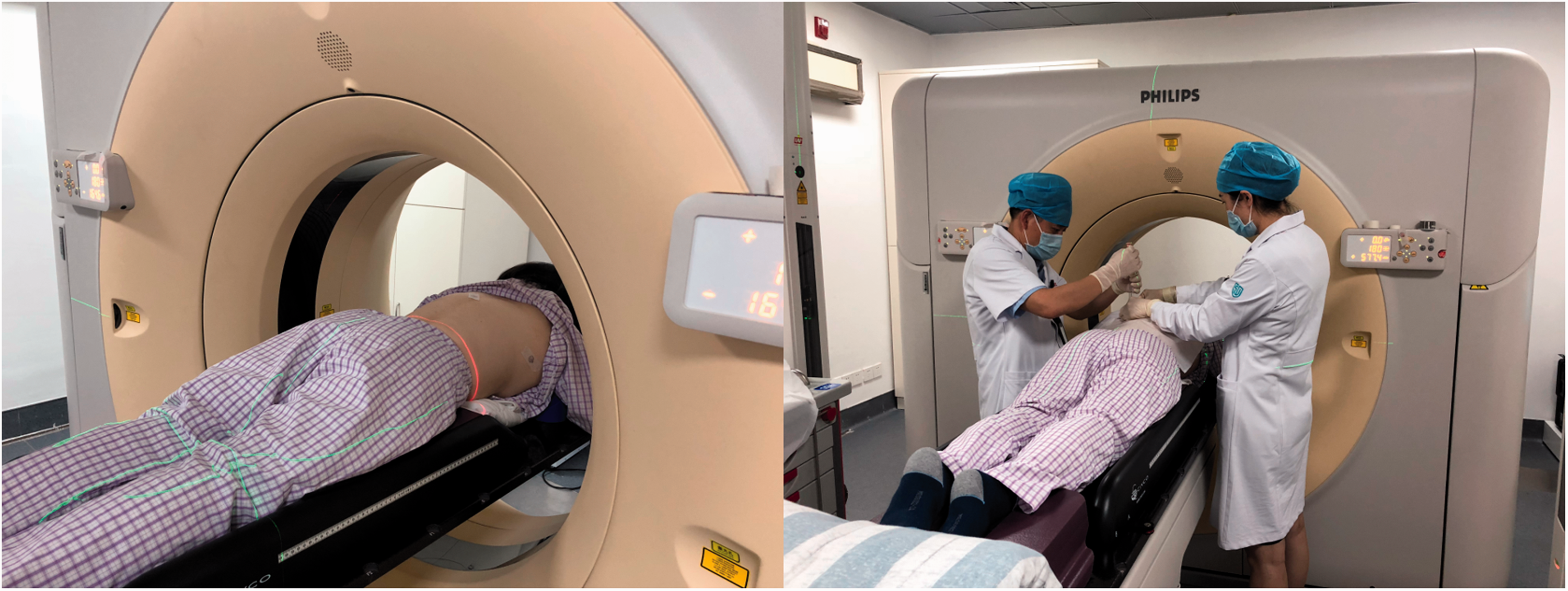
Representative photographs showing a patient in the prone position and the biopsy being undertaken.
Biopsy specimens were successfully obtained in all 38 patients. The success rate of puncture was 100% (38 of 38 patients). As summarized in Table 4, the diagnostic rate of malignancy was 76% (29 of 38 patients), of which 20 of 25 patients were in the group initially diagnosed with unconfirmed lesions and nine of 13 patients were in the group of treated patients that needed additional pathological analyses. Among the 29 patients with malignant tumours, adenocarcinoma was confirmed in 16 patients, squamous cell carcinoma in six patients, small cell lung carcinoma in two patients, metastatic disease in three patients and the remaining two patients had epithelial carcinoma. The other nine patients were diagnosed with benign diseases.
Pathological results of patients (

Minor needle track bleeding was observed in three patients. For the 33 patients that underwent a pulmonary biopsy, 12 had a minor pneumothorax (36%), of which two progressed to a massive pneumothorax at re-examination after 24–48 h (6%). Patients with a massive pneumothorax were treated successfully with closed thoracic drainage and the minor pneumothorax absorbed themselves without further intervention. No other complications, such as haemoptysis, haemothorax or subcutaneous haematoma, were found in the cohort. Moreover, no puncture-related complications, such as bleeding, infection or pancreatitis, were observed in the three patients that underwent an abdominal biopsy.
Based on the regression analyses (Table 5), a significant correlation was found between puncture angle and pneumothorax; the incidence rate of pneumothorax in punctures with an angle of 80–100° was significantly higher than that with other angles (11 of 23 patients [48%] versus one of 10 patients [10%], respectively;
Regression analysis of factors affecting the incidence of pneumothorax in patients (

Data presented as
NS, no significant association (
Discussion
In the era of individualized precision treatment, especially with the advancements in molecular targeted therapy and immunotherapy, pathology is still a prerequisite for further gene detection, exploration of drug resistance mechanisms and evaluation of patient conditions.8–10 CT-guided percutaneous biopsy, as an important solution for the acquisition of pathological information, is playing an increasingly critical role in clinical practice, 11 which requires improved operability and accuracy. Unfortunately, there still exists defects in this process in terms of inaccurate positioning, low one-time success rate, heterogeneity of operators’ experience and the poor compliance of some patients who are prone to serious complications.4–7 This is the first study to combine TPS and radiotherapy positioning systems with the puncture process and it has refined the success of this operation. The initial results from this current study suggest that this method has advantages in puncture accuracy and a higher success rate, with satisfactory safety. Thus, it has the potential to be an important contribution to the field of CT-guided percutaneous biopsy.
The advantages of combining TPS with CT-guided percutaneous biopsy are summarized as follows: (i) the pre-puncture area (GTV) can be freely delineated by operators on TPS based on their clinical experience and the imaging features of lesions; (ii) regions of tumour liquefaction or necrosis can be avoided; and (iii) the detection rate can be improved. Furthermore, with accurate coordinate information conversion between TPS and a CT simulator system, the optimal puncture point is projected onto the patient’s body surface based on relative coordinates, which are accurate to the millimetre. Therefore, the accuracy of positioning is greatly improved compared with the traditional method of using a standard lead point mesh and the restriction on the choice of puncture point is reduced to a certain extent.
Several attempts had been made to improve puncture accuracy,12,13 but these did not take into account the inevitable errors between actual operations and ideal pre-set parameters. This current study compared actual and planned puncture data and the results showed a good agreement between the two. There was no statistical difference between the actual and planned puncture depths (5.28 ± 1.95 cm versus 5.34 ± 2.01 cm, respectively) or angles (88° versus 90°, respectively). The reason that the actual puncture depth was less than the planned depth might be due to different levels of skin retraction during the puncture process. Ultimately, the deviations were controlled within a satisfactory range and the one-time success rate reached 100%, demonstrating that this technology was suitable for clinical application.
The success rate of CT-guided percutaneous biopsy reported varies between different techniques. For example, a previous study reported a puncture success rate of 98% with template-assisted CT-guided percutaneous biopsy, of which the one-time success rate was 50%. 12 Another study used an electromagnetic navigation system for CT-guided biopsy, reducing puncture time and radiation exposure. 14 The success rate was 84.4% and the one-time success was only 58.3%. 14 Similarly, large-core biopsy (10G needle) was successful in 88.6%, 3 which was lower than that of the present study. As for puncture time, 20 min would not appear to be an advantage compared with other methods (range 9–34 min),3,13,14 but it was acceptable.
Puncture accuracy is thought to be associated with detection rate and it is influenced by multiple variables. For example, a previous study reported on 123 patients with pulmonary nodules and found that diagnostic accuracy decreased in proportion to a decrease in lesion size.
15
Similarly, the diagnostic accuracy of CT-guided percutaneous lung biopsy in lung nodules ≤ 1.5 cm was lower than that in nodules > 1.5 cm.
16
In this current study, the detection rate was 100%. The diagnostic rate of malignancy was 76% (29 of 38), of which 20 of 25 patients were in the group initially diagnosed with unconfirmed lesions and nine of 13 patients were in the group of treated patients that needed additional pathological analyses, which was slightly higher than recent literature reports.
12
Although lesion size showed no effect on puncture accuracy, ΔH tended to decrease with increasing lesion size. Additionally, pulmonary puncture location was found to be a risk factor for puncture angle accuracy (
The most common complication of pulmonary puncture reported is pneumothorax, followed by haemorrhage, but there is no consensus on the incidence rates of these complications. For example, according to several studies that reported on large sample sizes in recent years,18–22 pneumothorax rates range from 3.1% to 49.3% and the incidence of haemorrhage was 6.9%–29.1% after CT-guided biopsy. Similarly, other studies have shown pneumothorax rates between 5.3% and 42%, of which the incidence of the requirement for closed thoracic drainage was between 2% and 21%.23–26 The incidence of pulmonary haemorrhage was 5%–30% and severe pulmonary haemorrhage occurred in roughly 3.1% of cases.23–26 In this current study, the incidence rate of minor pneumothorax was 36% and only 6% progressed to massive pneumothorax. Only three patients suffered needle track bleeding and no other rare complications were observed. The complication rates were consistent with previous studies.18–26 Pulmonary haemorrhage seemed to occur less often than the reported rates,23–26 possibly due to the improvement of positioning accuracy and patients’ comfort in this method. A high one-time success rate also helped to avoid lung tissue and vessel damage caused by repeated needle adjustment. Some researchers have reported that small lesions and repeated puncture had prognostic value for pneumothorax. 27 A previous study reported that pneumothorax was associated with lesion size and the depth of needle penetration. 28 This current investigation revealed that puncture angle was the main risk factor for pneumothorax, while sex, age, lesion location, lesion size and puncture depth had no effect. This might be related to an insufficient sample size of this study. For this current cohort, the occurrence of complications was less affected by clinically relevant factors, which reflected the safety of this technology to some extent. Yet, these results still need to be verified in a larger research cohort in the future.
This current study had several limitations. First, although the 38 eligible patients that were enrolled in the study were evaluated as thoroughly as possible, the sample size was small. A more comprehensive analysis was limited by this small sample size, particularly when identifying risk factors of puncture accuracy and complications. For small lesions with high puncture difficulty, it would be more convincing if a subgroup analysis was performed on a larger sample size to verify the technical superiority of this method relative to the current standard of care. Secondly, the number of patients that underwent abdominal biopsy was very limited, making it hard to explore more details in this subgroup. Further investigation in different tissues is required.
In conclusion, this current study summarized the initial experience and results of the clinical application of a TPS-assisted large-aperture CT simulator-guided percutaneous biopsy to improve the biopsy procedure by combining the advantages of radiotherapy specialties. This emerging technology may enhance the puncture accuracy and success rate under the premise of ensuring safety and efficiency, which is worth promoting in clinical applications.
Footnotes
Acknowledgements
We are grateful to our colleagues from the departments of oncology and surgical oncology that provided expertise that greatly assisted this research. We also express our immense appreciation to Professor Jiade Lu for sharing his knowledge and his comments on an earlier version of the manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
