Abstract
The various presentations of osseous Langerhans cell histiocytosis (LCH) make it difficult to distinguish from other bone diseases. In addition, there is no universally accepted protocol for managing osseous LCH for single non-central nervous system-risk lesions. Here, the rare cases of two paediatric patients, aged 1 and 2 years, who presented with a solitary tibial lesion at time of LCH diagnosis, are reported. One patient progressed to multiple lesions after curettage of the original lesion. Subsequently, both patients received preventive chemotherapy using the Taiwan Paediatric Oncology Group (TPOG) revised protocol for treating low risk patients with LCH, namely, TPOG LCH2002-LR. After receiving this treatment, which included a schedule of prednisolone and vincristine for 6 weeks, followed by prednisolone, vincristine and 6-mercaptopurine for a further 48 weeks, both patients are free from recurrence or progression.
Introduction
Langerhans cell histiocytosis (LCH) is a sporadic disease that predominantly affects the paediatric population. It is characterized by idiopathic clonal proliferation of Langerhans cells accumulating in various organs, and approximately 80% of patients with LCH have bone involvement. 1 Osseous LCH is known as ‘the great imitator’ as it resembles many bone diseases, and is commonly misdiagnosed. Clinically, patients with LCH of the bone manifest localized bone pain with tender soft tissue swelling, and in children, LCH of the bone mostly affects the skull. Other relatively common sites include the proximal femur, ribs, pelvis, and vertebrae. 1 A solitary bone lesion of the tibia is relatively rare. Solitary osseous lesions are commonly treated with observation or curettage alone, however, there is still no consensus on the adequacy of treatment. Here, two paediatric cases of a single tibial lesion as the initial presentation, one of which progressed to multiple lesions after curettage of the original lesion, are reported.
Case reports
All patient data were deidentified, and verbal informed consent to publish the cases was obtained from the patients and their legal proxies
Case 1
A 1-year-old male presented at the Orthopaedic Outpatient Clinic of National Taiwan University Hospital, in June 2009, with a 2-week history of limp, fever and difficulty in moving the left leg. At the emergency room, a physical examination showed no gross deformity, swelling, skin lesion, pitting oedema or local tenderness at the left leg. There were no recent body weight loss, jaundice, polyuria or palpable lymph nodes noted. Except for leucocytosis, all laboratory values were within reference range (white blood cell (WBC) count, 14.67 × 103/µl; red blood cell (RBC) count, 4.17 × 106/µl; haemoglobin (Hb), 11.5 g/dl; haematocrit (HCT), 34.2%; mean corpuscular volume (MCV), 82.0 fl; mean corpuscular haemoglobin (MCH), 27.6 pg; MCH concentration (MCHC), 33.6 g/dl; platelets, 363 × 103/µl; aspartate aminotransferase, 28 U/l; alkaline phosphatase, 121 U/l; lactate dehydrogenase (LDH), 220 U/l; and creatinine (CRE), 0.4 mg/dl). Plain radiography revealed a radiolucent lesion at the left proximal tibial meta-diaphysis with periosteal reaction (Figure 1A and 1B). Osteomyelitis was suspected and the patient underwent curettage of the lesion and grafting with tobramycin-impregnated OSTEOSET® T Bone Graft Substitute (Wright Medical Technology Inc., Arlington, TN, USA; Figure 1C and 1D). The pathology report revealed diffuse mononuclear cell infiltration with positive immunostaining for T-cell surface glycoprotein CD1a (CD1a), protein S-100 (S-100), and C-type lectin domain family 4 member K (CD207) and features of histiocytic origin (Figure 1E, 1F and 1G). Immunohistochemical analysis showed that there was no serine/threonine-protein kinase B-raf (BRAF)-V600E mutation (Figure 1H). Except for the left tibia, the skeletal survey and bone scan did not show additional skeletal lesions. Brain magnetic resonance imaging (MRI) was also arranged to assess for possible multisystem involvement, and this revealed negative findings. The patient was diagnosed with unifocal osseous LCH.
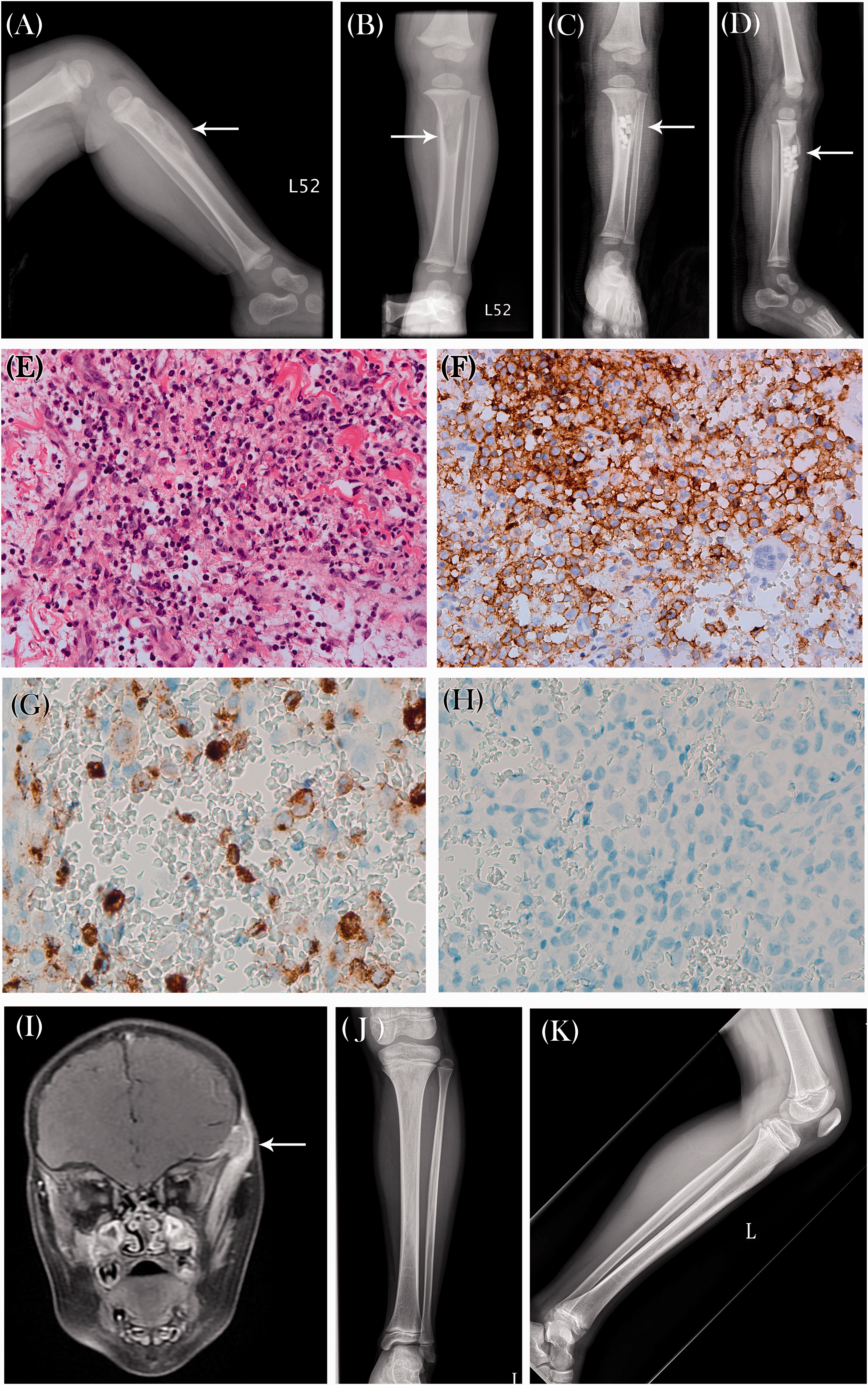
Radiographs showing: (A and B) an osteolytic lesion locating eccentrically at the anterior proximal third of the left tibia with laminated periosteal reaction and adjacent mild soft tissue swelling; and (C and D) OSTEOSET® T Bone Graft Substitute pellets at the anterior aspect of the left proximal tibia; photomicrographs showing: (E) haematoxylin and eosin-stained histiocytoid cells with indented or grooved nuclei, and a few eosinophils in the background; immunohistochemical staining of tumour cells with (F) positive signal for T-cell surface glycoprotein CD1a; (G) positive signal for C-type lectin domain family 4 member K (CD207), supporting the diagnosis of Langerhans cell histiocytosis; and (H) negative signal for serine/threonine-protein kinase B-raf (BRAF)-V600E mutation (all original magnification × 400); (I) magnetic resonance image showing a mass lesion near the left temporal facial region, with left temporalis involved. The lesions were moderately enhanced by contrast medium, indicating possible tumour involvement; and (J and K) follow-up radiographs at 9 years after surgery showing no new bone lesion.
Two months following curettage, a painless mass was observed at the left fronto-temporal area. Skull radiographs and MRI images revealed an osteolytic bone lesion at the left frontal region of the skull, and a mass near the left temporal facial region with temporalis involvement (Figure 1I). The patient was diagnosed with multifocal osseous LCH, and started chemotherapy with vincristine and prednisolone following the Taiwan Paediatric Oncology Group (TPOG) protocol for treating LCH in low-risk (LR) patients, namely, TPOG-LCH2002-LR (Table 1). The lesions subsided. A follow-up assessment at 9 years showed no recurrence or new lesion (Figure 1J and 1K).
Taiwan Paediatric Oncology Group (TPOG) chemotherapy protocol for treating Langerhans cell histiocytosis (LCH) in low risk (LR) patients, TPOG LCH2002-LR.

aEvaluated at weeks 4–6: if no evidence of active disease or improved, start continuation therapy; if evidence of disease progression, shift to initial therapy protocol for high-risk patients.
Case 2
A 2-year-old female was brought to the emergency room of National Taiwan University Hospital, in October 2017, with a history right lower leg painful contusion after a fall 1 week previously. There was a palpable mass without cutaneous lesion at the right middle calf. No other mass like lesion, pitting oedema, recent body weight loss, cough, chest tightness or polyuria was noted. Standard laboratory test results were within normal limits (WBC, 11.08 × 103/µl; RBC, 5.14 × 106/µl; Hb, 13 g/dl; HCT, 38.6%; MCV, 75.1 fl; MCH, 25.3 pg; MCHC, 33.7 g/dl; platelets, 315 × 103/µl; C-reactive protein, 0.34 mg/dl; LDH, 241 U/l; and CRE, 0.3 mg/dl). Radiographs revealed an osteolytic lesion at the tibial diaphysis, with erosion of the medical cortex and laminated periosteal reaction (Figure 2A). The MRI showed uneven cortical thickening with amorphous bone marrow hyperaemia and oedema in the right anterior proximal tibial shaft (Figure 2B).

(A) Plain radiograph showing osteolytic lesion with laminated periosteal reaction and ill-defined margin at the right tibial mid shaft; (B) magnetic resonance image showing uneven cortical thickening with amorphous bone marrow hyperaemia and oedema in the anterior portion of the right mid tibial shaft; photomicrographs showing: (C) haematoxylin and eosin-stained sheets of histiocytoid cells with indented or grooved nuclei and occasional multinucleation, with a few eosinophils; immunohistochemical staining of tumour cells with (D and E) positive signal for T-cell surface glycoprotein CD1a and C-type lectin domain family 4 member K (CD207), supporting the diagnosis of Langerhans cell histiocytosis; and (F) negative signal for serine/threonine-protein kinase B-raf (BRAF)-V600E mutation (all original magnification × 400).
Osteomyelitis was suspected, and the patient underwent right tibial shaft debridement. The pathology report revealed scattered dendritic Langerhans cells (Figure 2C) with positive immunohistochemistry signal for CD1a (Figure 2D) and CD207 (Figure 2E), but negative result for the BRAF-V600E mutation (Figure 2F), indicative of Langerhans cell histiocytosis. The follow up bone survey showed no other definite bone lesion. In light of the clinical course in case 1, the patient received prophylactic chemotherapy following the TPOG-LCH2002-LR protocol (Table 1). At the one-year follow-up assessment, the patient showed no sign of recurrence.
Discussion
Langerhans cell histiocytosis occurs throughout the neonatal period to adulthood, with the highest prevalence observed in children aged 1–4 years. 2 , 3 LCH is classified as a single system disease (SS-LCH) or multisystem disease (MS-LCH), based on the extent of system involvement. Totadri et al. 4 reported that patients with SS-LCH have favourable prognoses, while patients with MS-LCH have lower rates of either five-year event-free or overall survival, particularly those with risk-organ involvement. MS-LCH involving risk organs, such as the central nervous system (CNS), haematopoietic system or liver, may lead to a rapidly fatal outcome. Both patients in the present report were children under three-years-of-age, and lesions were confined to the skeleton, therefore, they were diagnosed with SS-LCH. Skeletal presentation of LCH with CNS-risk bone involvement, such as orbital, temporal, sphenoid, or ethmoid bones, may indicate that the patient is in the higher risk group. A comprehensive survey and evaluations, such as a bone survey, are essential.
The initial presentations of the current cases were isolated osteolytic tibial lesions with symptoms that mimicked osteomyelitis. Factors for differential diagnosis include infection, and benign or malignant tumours, and the present diagnosis of LCH was solely based on histologic confirmation. This osseous lesion, also called eosinophilic granuloma, can manifest in any location depending on the age of patients. In a review of 686 cases of eosinophilic granuloma, Slater et al., 5 stated that there is a predilection for the axial skeleton, with calvarial lesions accounting for around 42% of eosinophilic granuloma of bone. In the long bones, the proximal femur is the most commonly affected. Tibial involvement was reported as one of the multifocal lesions. The only report of solitary tibial LCH was a case with a lesion at a growing epiphysis, 6 which differs from the present cases of metaphysis and diaphysis involvement.
Treatment of eosinophilic granuloma depends not only on disease extent, but also risk stratification, which is based on whether or not ‘CNS-risk’ bones are involved. Diverse therapeutic options consist of conservative treatment (observation, immobilization), local treatment (local excision, curettage, low-dose irradiation) or systemic therapy (chemotherapy, corticosteroid or indomethacin injections), and combined therapy. 1 , 7 , 8 There is no universally accepted protocol for the management of osseous eosinophilic granuloma for single non-CNS-risk lesion. In general, a solitary bone lesion tends to spontaneously regress with time, particularly in the paediatric population. 7 Hence, conservative therapy or curettage are often recommended. Nevertheless, once the disease progresses to the multifocal type, or MS-LCH, treatment should become more aggressive. In the current report, the patient in case 1 first presented with an isolated lesion. However, the disease progressed to multifocal osseous LCH with CNS-risk bone involvement, within two months following initial treatment. Therefore, with the progressive course of case 1 in mind, the patient in case 2 was treated with prophylactic chemotherapy using the TPOG-LCH2002-LR schedule (Table 1).
The Taiwan Paediatric Oncology Group revised the schedule for treating LCH in 2002, and stratified the treatments according to organ involvement. The high-risk group comprised patients with haematopoietic, liver, spleen or lung involvement. Treatment for the low risk group, TPOG-LCH2002-LR, consists of high dose prednisolone and low dose vincristine, while therapy for the high risk group includes the addition of high dose methotrexate. 9 A retrospective study compiled data from 132 patients, and found that unifocal osseous LCH had a reactivation rate of about 7.6% after local treatment. 10 The present authors believe that preventive chemotherapy with TPOG-LCH2002-LR may reduce the risk of disease reactivation, however, further studies are needed to verify the efficacy of this management strategy.
The present case report supports the importance of careful evaluation of possible systemic involvement and diligent follow-up in unifocal bony LCH. Patients with a solitary lesion as first presentation have the possibility of developing a second lesion within months. The study also raises a question whether preventive chemotherapy, particularly the TPOG-LCH2002-LR protocol for high-risk patients, is applicable, since adverse reactions associated with TPOG-LCH2002-LR for low-risk patients were minimal. Methotrexate, particularly at a high dose, may be associated with multiple adverse effects, such as hepatotoxicity, renal toxicity, encephalopathy, skin photoreactivation and photoenhancement, thus the TPOG-LCH2002 protocol for high-risk patients may result in more adverse reactions.
The term ‘maximal extent of disease’, defined by Bernstrand et al., 11 suggests that reactivation of LCH may extend to previously uninvolved organs or systems. To date, there are no available published reports to determine the correlation between primary lesion site and incidence of disease extent. Physicians should be aware of the possibility of progression from unifocal LCH to multifocal, or even MS-LCH. Also, due to the variability of clinical symptoms and location of lesions, eosinophilic granuloma should always be included in the differential diagnosis of solidary bony tibial lesions.
