Abstract
Heart surgery in patients from high-altitude areas is more challenging than usual. Few studies have been published on this issue, and none of them have discussed the effect of an altitude change (from high to low altitude) on a patient’s physiology or its effects on a patient’s perioperative management. Here, we present the case of a 46-year-old man who was a long-time resident of Tibetan area in Sichuan (altitude >3000 m) who underwent Stanford type A aortic dissection emergency surgery on the plain. Anesthetic management occurred through monitoring of the bispectral index (BIS) and transesophageal echocardiography (TEE), and we used a relatively loose fluid hydration strategy. The surgery was performed using cardiopulmonary bypass (CPB), deep hypothermia (DH), and selective antegrade cerebral perfusion. The most prominent anesthesia challenges for these patients are physiological changes due to habitation in an high-altitude environment (chronic hypoxemia), which can cause hyperhemoglobinemia, polycythemia, hypercoagulable blood, and even pulmonary hypertension, cor pulmonale, or congestive heart failure. Optimized perioperative management and close cooperation among the entire cardiac medical team were the key factors in the successful management of this rare case.
Keywords
Introduction
Human habitation in high-altitude environments may result in respiratory, cardiovascular, and hematological changes in relation to the oxygen uptake and transport. A sudden altitude change may lead to distribution of physiological function. A rapid or recent ascent into these environments may lead to high-altitude diseases such as high-altitude cerebral edema, pulmonary edema, and acute and chronic mountain sickness syndrome with headache, dizziness, fatigue, and insomnia. 1 However, long-term residency in high-altitude areas activates gene-coding programs for proteins that are involved in oxygen transport, blood vessel growth, and erythropoiesis, thus leading to unique physiological respiratory, circulatory, and hematological system phenotypes. 2 Previous studies have shown that the effects of anesthesia and surgery on the hemodynamics are distinctly different compared with those from individuals who live at low altitudes.3,4 There are increased perioperative risks and complications of a high-altitude-area inhabitant who undergoes surgery in a low-altitude area, and it is suggested to give these patients 3 to 5 days before surgery to de-acclimate.5,6
Stanford type A acute aortic dissection is a life-threatening emergency that requires swift open surgical repair on cardiopulmonary bypass (CPB). 7 Thus, cardiac emergency surgeries on patients from high-altitude areas are even more challenging. In this study, we report the case of a patient who resided in a high-altitude area who underwent Stanford type A aortic dissection emergency surgery in a low-altitude area. This case report will add to the limited body of knowledge on the anesthetic perioperative management strategy in these patients.
Case report
A healthy 46-year-old man was transferred to our center (Chengdu, China, 500 m above sea level) 52 hours after the onset of acute chest and upper abdominal pain. His medical history was normal. Noteworthy was the fact that he had lived in the Tibetan area of Sichuan (at an altitude >3000 m) since childhood and was transferred to a low-altitude area for only 3 hours, without a chance to acclimate to the altitude change. Upon the ambulance’s arrival and after the initial examination, the patient’s heart rate was 92 beats per minute (bpm), blood pressure was 165/95 mmHg (systolic/diastolic), respiratory rate was 22 breaths per minute, and the oxygen saturation was 92% (FiO2 = 21%). There was also no sign of jugular venous distension or swelling in his legs. The 12-lead electrocardiogram (ECG) indicated a sinus rhythm with a rate of 92 bpm, and there was no sign of ST elevation, pathologic Q-spikes, or dysrhythmia. The blood test yielded a red blood cell (RBC) count of 7.97 × 1012/L, hematocrit (Hct) level of 74.7%, and a hemoglobin concentration of 249 g/L. These values were all much higher than those of people who live in low-altitude areas. The white blood cell count and platelet results were unremarkable. The coagulation tests showed an increase in the D-dimer concentration of 6.5 mg/L and in the fibrin degradation product (FDP) concentration of 18.3 mg/L. The cardiac enzyme levels were not increased, high-sensitivity troponin T was 5.1 ng/L, creatine kinase-MB (CK-MB) was 1.6 ng/mL, and type B natriuretic peptide (BNP) was 95.8 pg/mL. In the emergency department, we performed a bedside transthoracic echocardiography (TTE), which unexpectedly revealed dilation of the aortic root with a sign of dissection. TTE demonstrated a mild degree of left ventricular (LV) hypertrophy, LV systolic function with an ejection fraction of 64%, and normal valve, atria, and right ventricular functions. ECG-gated computed tomography angiography (CTA) demonstrated that the dissection initially manifested from the aortic root, and it also showed the involvement of the aortic arch that extended in the inferior direction to the descending and abdominal aortae (Figure 1a and b). The maximum abdominal aorta diameter was 27 mm, and the celiac trunk, superior mesenteric artery, and the left renal artery arose from the true lumen, whereas the right renal and inferior mesenteric artery arose from the false lumen. There were no perfusion deficits in the kidneys.
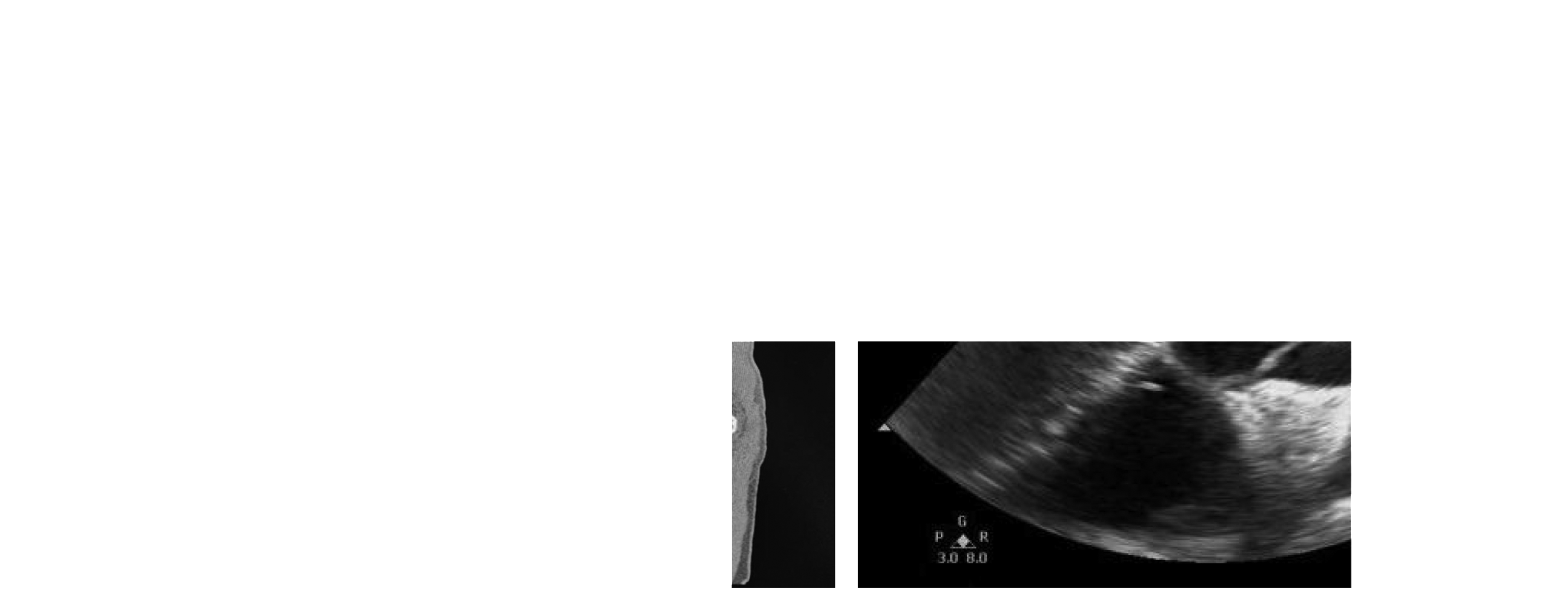
CTA showing the dissection from the aortic root (a) that extended in the inferior direction to the descending and abdominal aorta (b); and intraoperative TEE (c) image showing aortic root dilation.
The patient spent 6 hours in the emergency center where he underwent a preoperative examination and basic treatment, he was transferred to the operating room on blood pressure-lowering treatment (nicardipine), and his blood pressure, ECG, and oxygen saturation (SPO2) were monitored. Subsequently, his left radial and left dorsal arteries were catheterized following the administration of a local anesthesia to monitor arterial pressure invasively. The arterial pressure before induction was 95/57 mmHg with a heart rate of 90 bpm. Anesthetic induction was performed by intravenous injection of midazolam (0.1–0.2 mg/kg), etomidate (0.3 mg/kg), sufentanil (1–2 µg/kg), and cisatracurium (0.15 mg/kg), in accordance with the guidance for the bispectral index (BIS). An endotracheal tube (7.5#) was placed, and anesthesia was maintained with sevoflurane (1.5%–2.5%), sufentanil (1–2 µg/kg/hour), and dexmedetomidine (0.4 µg/kg/hour), together with intermittent intravenous of cisatracurium. Mechanical ventilation was performed with a respiration ratio from 1:1.5 to 1:2.0, the respiration rate in the range of 10 to 12 breaths per minute, and end-tidal carbon dioxide pressure (PetCO2) maintained in the range of 35 to 40 mmHg (1 mmHg = 0.133 kPa). After anesthetic induction, a triple-lumen central venous catheter was placed in the vena cava through the right internal jugular vein. Nasopharyngeal and rectal temperatures were monitored. Intraoperative transesophageal echocardiography (TEE) revealed aortic root dilation with a sign of dissection (Figure 1c).
Compound sodium chloride solution, hydroxyethyl starch 130/0.4, and blood products were used for intraoperative fluid infusion. Stanford type A aortic dissection surgery (ascending aortic replacement + aortic arch replacement + descending aortic stent implantation) was performed with CPB, deep hypothermia, and selective antegrade cerebral perfusion. The vasoactive drugs, norepinephrine, epinephrine, and nitroglycerin were used to maintain stable hemodynamics. CPB lasted for 215 minutes. The intubated patient was transferred to the intensive care unit (ICU) after surgery. The endotracheal tube was removed after the patient awoke, spontaneous breathing improved, and arterial blood gas showed satisfactory oxygenation and hemodynamic stability. The postoperative course in the ICU was stable, and there were no complications. Epinephrine and norepinephrine infusions were discontinued within 24 hours post-ICU admission. The TTE conducted on postoperative day (POD) 3 did not identify any obvious abnormalities in the artificial blood vessels. The patient was discharged from the hospital on POD 7, and he lived in a low-altitude area for 2 weeks before he returned to his permanent residence in the local Tibetan area.
Discussion
This is a rare case of a patient who had been a long-time inhabitant in a high-altitude area who underwent emergency surgery for Stanford type A aortic dissection. Long-term residency in high-altitude areas leads to unique physiological respiratory, circulatory, and hematological system phenotypes. Sudden altitude changes may lead to distribution of physiological functions. There is no published literature that discusses the altitude change (from high to low altitude) and its effect on the patient’s physiology as well as its effects on the patient’s perioperative management. In the present study, we summarized the physiological changes that occur to acclimate a person to a hypoxemic environment, reviewed the cases that reported surgery and complications in high-altitude inhabitants, and suggested perioperative management for these patients.
Physiological changes in response to high altitude
High-altitude areas have a low-ambient barometric pressure, which leads to a decrease in the partial pressure of oxygen, and this results in hypobaric hypoxemia. Key physiological changes in high-altitude residents are shown in Figure 2. Hyperhemoglobinemia and polycythemia develop in response to facilitated loading of O2 in the lung. Platelets are also activated and lead to increased blood coagulability. Hypoxic pulmonary vasoconstriction is a vasomotor response to alveolar hypoxia; it increases pulmonary vascular resistance and contributes to significant pulmonary hypertension. Studies have shown that high-altitude-area residents have a larger lung volume with increased vital and total lung capacities.8,9 For the cardiovascular system, systemic blood pressure and LV systolic function increase in response to hypoxia during early adaptation, and mild to moderate hypertension and LV myocardial hypertrophy may occur with chronic habitation. Cardiac and cerebral blood flow is selectively increased. Previous studies showed that Tibetan highlanders achieved normal O2 delivery because of a ten-fold increase in the plasma concentration of circulating nitric oxide (NO), which leads to vasodilation and increased tissue blood flow. 10 Some serious cases may lead to cyanosis, cor pulmonale, and congestive heart failure. 11 In the present case, the patient did not have any severe complications such as pulmonary hypertension and heart failure described above. The laboratory tests demonstrated that hyperhemoglobinemia and polycythemia (RBC was 7.97 × 1012/L and the hemoglobin concentration was 249 g/L) had developed, and the blood was in a hypercoagulable state (D-dimer of 6.5 mg/L and FDP 18.3 mg/L). In the acute phase of aortic dissection, the non-endothelial false lumen surface thrombosis activated coagulation and caused an increase in D-dimer. D-dimer (>0.5 mg/L) is recommended as a diagnostic biomarker of aortic dissection. 12 However, a previous study also showed higher D-dimer levels in high-altitude-area inhabitants. 13 These multiple factors might cause the increased coagulation markers that were observed in our patient.

Key physiological changes in high-altitude residents.
Surgery in high-altitude inhabitants
Table 1 shows a summary of the cases that reported surgery and complications in high-altitude inhabitants. Pan et al. 5 and Li et al. 6 showed the increased perioperative risks and complications for high-altitude-area inhabitants who underwent surgery in a low-altitude area. Zhang et al. 14 reported that living in a moderate high-altitude area (∼1800 m) may mimic the hypoxic preconditioning and decrease the incidence of ICU admission and postoperative serious adverse events in older patients.
Summary of surgery or complications in high-altitude inhabitants.

HA, hip arthroplasty; CH, chronic hypoxemia; pSAEs, postoperative serious adverse events; LG, laparoscopic gynecological; TIVA, totally intravenous anesthesia; CIVIA, combined intravenous–inhalation anesthesia; PONV, postoperative nausea/vomiting; ICA, intraoperative cardiac arrest; -, no data.
Perioperative management for high-altitude inhabitants
In the preoperative period, early diagnosis of aortic dissection is important. 15 In the present case, the patients’ hemodynamic parameters were stable. Because of his poor medical condition in the high-altitude area, he was transferred to our cardiac center and was managed by experienced teams. As discussed above, hyperhemoglobinemia, polycythemia, hypercoagulable blood, pulmonary hypertension, cyanosis, cor pulmonale, and congestive heart failure need to be evaluated when patients are from high-altitude areas.
For the Stanford type A aortic dissection, emergency surgical treatment with CPB is an important rescue strategy to save the lives of patients with increased cerebral injury and other life-threatening risks. In the present case, the induction process was stable, and it was conducted in accordance with BIS guidelines. Previous studies reported that high-altitude dwellers require significantly larger amounts of intravenous anesthetic propofol (rather than sevoflurane) to achieve a similar target BIS during both the induction and maintenance phases of anesthesia. This phenomenon may be a result of the higher cardiac output in the high-altitude area. In the present case, we did not find any difference in propofol use compared with the normal population.
Hyperhemoglobinemia and polycythemia are most common among high-altitude residents. Thus, blood protection and volume management strategies for these patients need to be discussed. The changes in RBC, Hct, and hemoglobin are listed in Table 2. Hyperhemoglobinemia can cause cerebral infarction, and there is a positive linear correlation between hemoglobin levels and risk factors for ischemic stroke. 16 In the present case, the patient’s blood was in a hypercoagulable state. Thus, we adopted a relatively loose fluid therapy strategy, and crystal or colloidal fluids were infused at a rate of 10 mL/kg/hour. CPB perfusion also required a hydration strategy, and hemoglobin was maintained at levels that were decreased by 30% compared with basal levels. Heparin was routinely used in cardiac surgery, and it played an important role in regulating blood viscosity. Blood protection and volume management also depended on the heart and respiratory function, peripheral vascular resistance, and laboratory test results. TEE or TTE are helpful for evaluating volume states and heart function. We will apply these techniques in the future.
Red blood cells, hematocrit, and hemoglobin at different time points.

ABG, arterial blood gas analysis; CPB, cardiopulmonary bypass; POD, postoperative day.
In the present case, general deep hypothermia (DH) (20–25°C) and arrested circulatory CPB were used to reduce the metabolic rate and oxygen consumption, and the DH period was approximately 35 minutes. Cerebral protection was achieved by the bilateral antegrade cerebral perfusion (approximately 25 minutes, cerebral blood flow around 5–10 mL/kg/minute) via the innominate artery and left common carotid artery. For patients with hypercoagulability, measures should be taken to control the coagulation state to avoid thrombosis, which may cause neurological complications such as cerebral infarction. Tranexamic acid was reported to increase the incidence of stroke, and it should be used with caution in this kind of patient. 17 DH that lasts more than 45 minutes could result in significantly increased brain injury. Furthermore, long-term CPB induced by DH can lead to serious systemic inflammatory response. Recently, moderate hypothermia (MH) (25–28°C) due to anesthesia has been encouraged by some scholars.18,19 Future studies should investigate if MH is better for the reduction of the risk of neurological disorders and other complications than DH in this type of population. No neurological complications occurred in this patient case after 6 months of follow-up. Studies suggest that monitoring cerebral saturations using regional cerebral oxygen saturation (rScO2) are more appropriate for assessing the brain oxygen supply and demand, and if the dissection impacts spinal function, somatosensory- and motor-evoked potential monitoring are needed. 20
In postoperative management, the focal aim of the ICU treatment was to achieve hemodynamic and organ functional stability. The catalytic activity and expression of certain cytochrome P450 isoenzymes at high altitudes are modified and lead to relative changes in drug metabolism and pharmacokinetics. 21 Thus, the patient’s recovery from anesthesia must be carefully managed. During follow-up, it is better for these patients to stay at low altitudes for a period of time to decrease the potential risk for physiological changes, which may harm the heart in its fragile postsurgical state.
In summary, for patients who have been long-time inhabitants of a high-altitude area and who undergo surgery in a low-altitude area, the most prominent anesthesia challenges are the physiological changes due to habitation in chronic hypoxemia. The detailed preoperative evaluation should consider hyperhemoglobinemia, polycythemia, hypercoagulable blood, pulmonary hypertension, cyanosis, cor pulmonale, and congestive heart failure. Optimized perioperative management and close cooperation among the entire cardiac medical team were the key factors the in successful management of this rare case.
Footnotes
Authors' contributions
H.Z. and Q.L. participated in the anesthesia and care of the patient, and they were responsible for obtaining the patient's informed consent, collecting the data from the patient, follow-up, and preparation of the manuscript. X.W. and T.Y collaborated on writing and review of the manuscript. All authors read and approved the final manuscript.
Consent
Written informed consent was obtained from the patient for publication of this case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Sichuan Science and Technology Program (2019YJ0577), the Science and Technology Project of the Health Planning Committee of Sichuan (20ZD011, 19PJ126), and the Science and Technology Project of Sichuan Overseas Students (2019-58).
