Abstract
Patients who commit suicide often deliberately hide their medical history. Given that taking pesticides is one of the most common methods of suicide, other forms of poisoning may be neglected in clinical practice. We report here a case of mixed-pesticide poisoning. The patient was poisoned by oral administration of a coumarin rodenticide in combination with an intramuscular injection of organophosphorus (OP) pesticide. The patient was treated with vitamin K1, cholinesterase reactivators, atropine, ventilator-assisted ventilation, and bedside debridement. Her condition gradually stabilized and she eventually recovered and was discharged. Assessment of the causes of delayed diagnosis and treatment suggests that we need to improve early detection and treatment of acute poisoning. It is especially important to ask about the patient’s medical history, conduct a careful physical examination, and track the clinical symptoms and differential diagnosis of common poisoning. In addition to the three common routes of poisoning—oral, inhalation, and cutaneous mucosal contact—intramuscular injection of OP can also lead to severe poisoning, which manifests as respiratory failure.
Introduction
Chlorpyrifos is an organophosphorus (OP) pesticide that is a neutral, organic, non-volatile poison derived from thiophosphoric acid; its chemical name is O,O-diethyl O-3,5,6-trichloro-2-pyridyl phosphorothioate. It is available commercially under various tradenames, including Dursban, Lorsban, and Empire 20. Acute chlorpyrifos poisoning is usually related to suicide. 1 Chlorpyrifos has an obvious inhibitory effect on cholinesterase, and cholinesterase activity recovers slowly, leading to a series of clinical symptoms. Clinical symptoms of chlorpyrifos poisoning are relatively mild, but respiratory failure, altered state of consciousness, and even death may occur. 2 OP pesticides can be absorbed not only through the digestive tract but also through the respiratory tract, skin mucosa, and even through intramuscular injection, which is more subtle and difficult to detect. 3 We report here a case of oral bromadiolone (rodenticide) poisoning combined with intramuscular chlorpyrifos poisoning. Because the patient did not provide a full medical history, diagnosis and treatment were delayed. The patient developed redness and swelling of the skin at the injection site, coagulation abnormality, and obvious symptoms of OP poisoning. These types of poisoning cases are rare.
Case report
A 59-year-old woman in good health reported that she had taken orally an unknown dose of the rodenticide bromadiolone 2 days previously; she did not undergo gastric lavage or induced vomiting. She reported nausea and vomiting of stomach contents 1 day earlier, and she had a dull pain in her upper abdomen. She felt fatigue and discomfort all over her body when she presented to our hospital for treatment. A physical examination after admission revealed that the patient was conscious and answering questions. Her pupil diameter was 1.5 mm bilaterally, she was sensitive to direct and indirect light reflection, and her heart rate was 99 beats/minute with a regular rhythm. She had mild tenderness in the upper abdomen but no rebound pain. A circular red swelling area about 2 cm in diameter was seen on her right medial malleolus, and slight elevation was noticed on the skin surface. She had a local increase in skin temperature with bilateral Babinski sign.
After admission, the patient underwent routine blood tests, including a coagulation series, liver and kidney function, electrolyte series, cholinesterase, and myocardial injury markers. Results of blood biochemistry were as follows: glutamic oxaloacetic transaminase 56 IU/L (normal range 14–36 IU/L), cholinesterase <200 IU/L (4650–10,400), and K+ 2.6 mmol/L (3.5–5.5 mmol/L); other results were in the normal range. For the blood coagulation series, prothrombin time (PT) was 62.9 s (11–14.50 s), PT-international normalized ratio (PT-INR) was 7.06, and activated partial thromboplastin time was 46.10 s (28.00–45.00 s) (Table 1). Because of the significant decrease in cholinesterase activity, the patient was asked about her medical history. The patient and her family members denied any history of exposure to or poisoning with OP drugs and indicated that the cause of the skin lesion on her right ankle was unknown. The patient was given vitamin K1, proton pump inhibitors to protect the gastric mucosa, sodium creatine phosphate to support the cardiac muscle, and electrolyte supplements. She became unconscious 4 hours after admission. Her electrocardiogram showed SpO2 60%, a pulse of 88 beats/minute, blood pressure of 108/75 mmHg, and a Glasgow Coma Scale score of 8 points. The patient had bilateral pinpoint pupils, breathing sounds in both lungs were heavy, and scattered moist rales could be heard in the lower field of both lungs. Bilateral Babinski signs were absent. The patient’s partial pressure of oxygen was less than 60 mmHg, indicating respiratory failure. Tracheal intubation and assisted ventilation were initiated immediately. A computed tomography (CT) scan of the brain and chest showed pulmonary inflammation but no intracranial hemorrhage (Figure 1). At this time, family members found a “chlorpyrifos” insecticide bottle at the patient’s residence. Following a comprehensive review of the physical examination, blood test results, and medical history provided by the patient’s family, we considered that acute OP poisoning was likely. A blood test showed 206 ng/mL bromadiolone and 180 ng/mL chlorpyrifos. Therefore, 1 mg of penehyclidine hydrochloride was injected intramuscularly, 2.0 g of pralidoxime iodide was given intravenously, and 0.5 mg of atropine was administered by injection. Following the above treatments, the patient’s respiratory failure was gradually corrected, consciousness was restored on day 2 of admission, and the patient was successfully weaned and extubated 3 days after endotracheal intubation. However, on day 4, we noted that the redness and swelling of the right ankle had increased, with a clear boundary and fluctuance, increased local skin temperature, and obvious pain (Figure 2). We again questioned the patient about her medical history and the cause of redness and swelling of the right ankle. The patient reported that she had used a syringe to inject about 5 mL of the chlorpyrifos insecticide into her right ankle 2 days before admission. After the patient was treated with vitamin K1, her coagulation function returned to normal and we decided on surgical debridement of the ankle. A 4-cm incision was made in the area of redness and swelling, and a lot of bloody necrotic material was seen. A sample of the secretion was sent for bacterial culture and drug sensitivity analysis. After cleaning the incision, a cavity with a diameter of about 3 cm was observed. After thorough disinfection of the cavity, a vacuum sealing drainage system was installed to allow continuous bedside drainage (Figure 3). Changes in the amount of drainage fluid and its properties were monitored continuously and the dressing was changed regularly. Bacteria were not cultured in the secretion sample, so it was considered aseptic inflammation. The patient’s condition improved, and she was discharged 15 days after admission. The area of redness and swelling of the right ankle decreased, no abnormal secretions were found, and the surface of the wound was clean and dry.
Changes in coagulation series and cholinesterase results during treatment.

PT, prothrombin time (reference value: 11.00–14.50 s); PT-INR, PT-international normalized ratio (reference value: 0.8–1.2); CHE, cholinesterase (reference value: 4300–11,500 U/L).
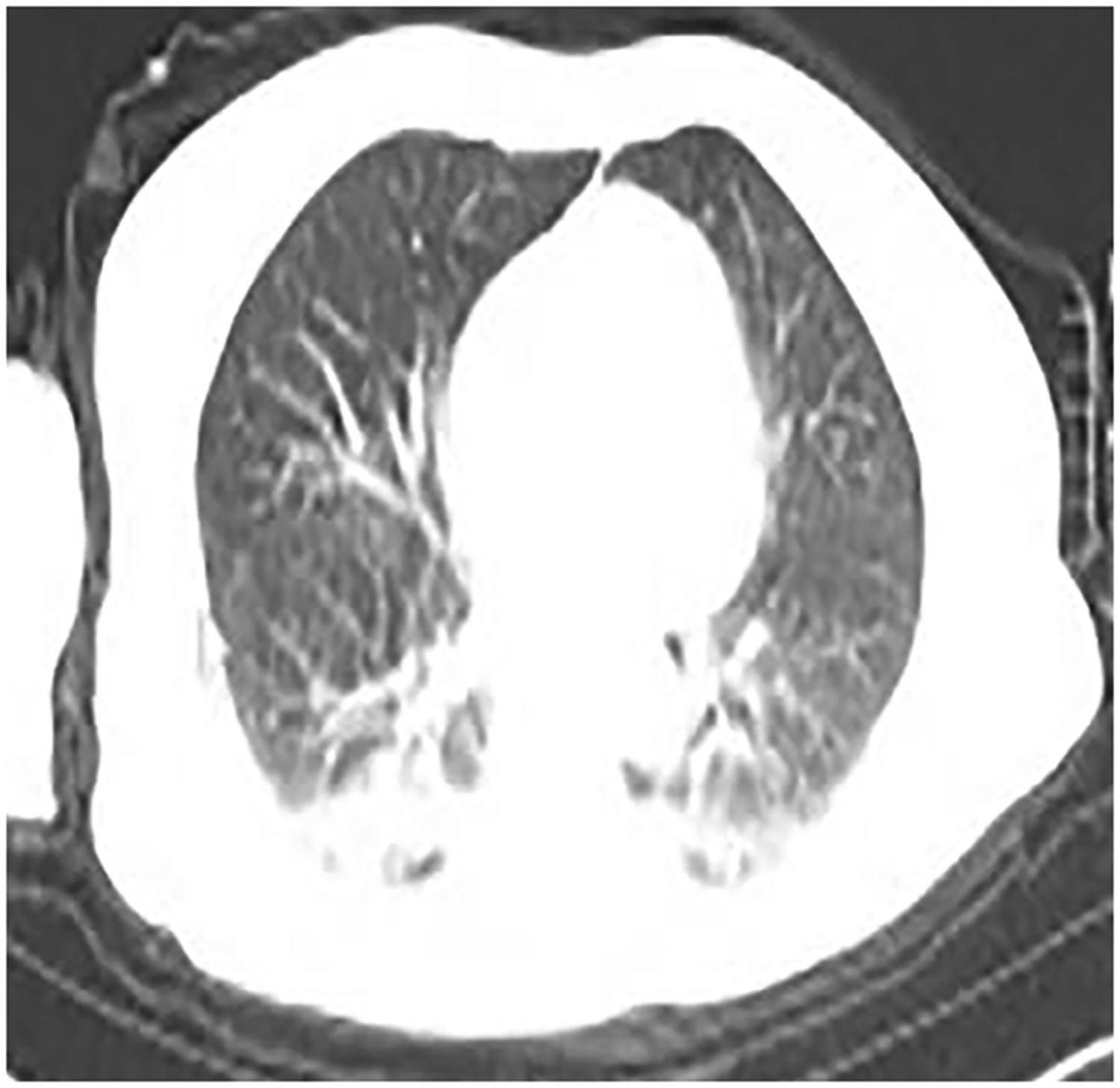
Computed tomography (CT) scan of the chest showing pulmonary inflammation.
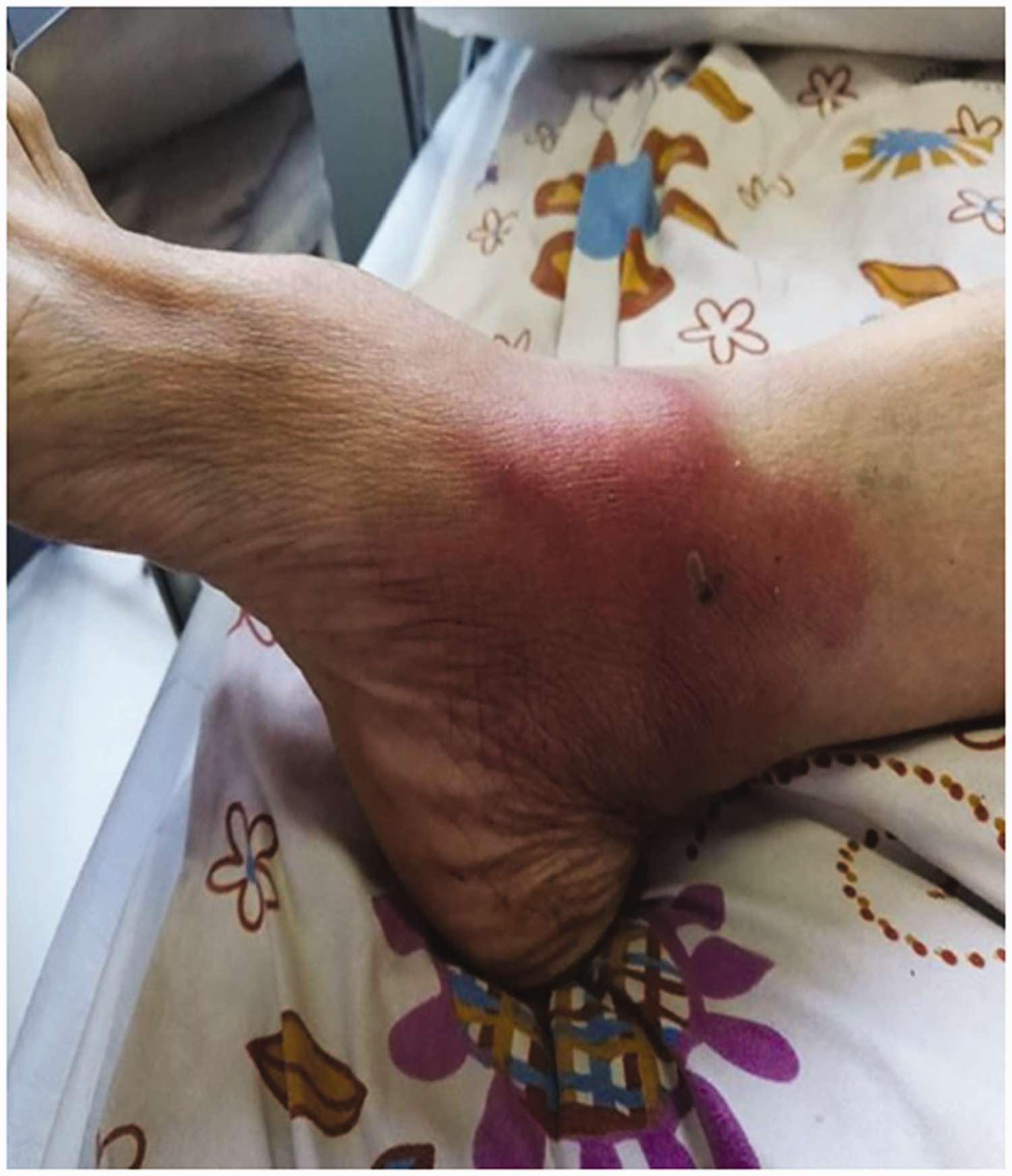
Redness and swelling at the injection site on the ankle.

Surgical drainage was performed at the site of injection of organophosphorus in the patient’s right ankle.
Discussion
Poisoning with OP pesticides is common. OP poisoning inhibits cholinesterase activity so that it can no longer decompose acetylcholine (ACh). When acetylcholinesterase is inhibited at the neuromuscular junction, it can only be replenished by new enzyme. However, recovery of acetylcholinesterase in the neuromuscular region is slow; it takes approximately 4 weeks for patients with mild to serious OP poisoning to recover completely. 4 When ACh accumulates in significant amounts, it induces a series of symptoms involving the muscarinic, nicotinic, and central nervous systems, including hyperhidrosis, salivation, tears, runny nose, lung rales, tremors, myasthenia, and even death. 5 Therefore, early diagnosis of OP poisoning, timely use of atropine and longtonine against cholinergic crisis, treatment with cholinesterase reactivators, and correction of respiratory failure are especially important. Chlorpyrifos is a highly effective, low toxicity OP insecticide and acaricide, and it is mainly used to treat grains, including rice, sorghum, wheat, and legumes, before storage or sowing to prevent worm infestation. 6 OP insecticides have a strong inhibitory effect on cholinesterase and cholinesterase activity. OP poisoning occurs mainly through the gastrointestinal tract, respiratory tract, and skin, and exposure by other means may cause different symptoms. The type and severity of symptoms depend on the dosage of OP and route of exposure. In this case, the signs of systemic toxicity caused by the intramuscular injection of OP did not appear immediately, which may have been due to the low toxic dose and small degree of involvement of the circulatory system, 7 which slowed the onset of symptoms. However, a significant decrease in cholinesterase was detected in our patient, and muscarinic symptoms such as miosis, nausea and vomiting, and abdominal pain were observed upon admission. Serious symptoms affecting the nervous system and respiratory failure occurred 4 hours after admission, which reflected the serious harm and potentially life-threatening effects of an intramuscular injection of OP. Organophosphorus metabolizes slowly, is absorbed continuously by the body (the log Kow value of chlorpyrifos is 5.11), and can cause death in severe cases of poisoning. Therefore, it is necessary to administer anticholinergic drugs (such as atropine and penehyclidine hydrochloride) and cholinesterase reactivators (such as pralidoxime chloride and pralidoxime iodide).8,9 Symptoms such as swelling, necrosis, and bloody exudation may occur at the injection site. In severe cases, compartment syndrome and chronic cholinergic crisis may occur and amputation may be necessary.7,10,11 Before surgical debridement, the serum cholinesterase level of this patient was consistently less than 200 IU/L. However, after surgery, serum cholinesterase level increased rapidly. Therefore, we recommend immediate surgical debridement to completely remove pesticide residues from soft tissue to prevent subsequent local and systemic symptoms.
The active ingredient of bromadiolone is warfarin, a derivative of dicoumarin, and its chemical structure is 3-(α-phenylacetone)-4-hydroxy coumarin. Bromadiolone inhibits the synthesis of vitamin K-dependent coagulation factors II, VII, IX, and X in liver microparticles, leading to coagulation disorder. Its action is slow and there are no obvious symptoms in the early stages. Patients often do not show clinical symptoms immediately after anticoagulant rodenticide poisoning, but a comprehensive analysis of poisoning history and coagulation test results can often ensure a clinical diagnosis and timely treatment. 12 Bromadiolone does not affect synthesized coagulation factors, and the half-life of the in vivo coagulation factors II, VII, IX, and X is 60 to 70, 6 to 8, 12 to 24, and 48 to 72 hours, respectively. Therefore, when the coagulation factors in the body become insufficient, bleeding will occur. Because the clinical half-life of bromadiolone is relatively long, patients must return to the hospital regularly to check PT and PT-INR indices in outpatient clinics even if no abnormalities are observed in the coagulation series. Patients should be advised to watch for gingival bleeding, skin ecchymosis, and other conditions so that vitamin K treatment is not discontinued prematurely, which could lead to a reversal of clinical improvement.
Our patient had a strong willingness to commit suicide and deliberately concealed her full medical history in the early stages of treatment, confessing only to oral consumption of bromadiolone, which resulted in the missed diagnosis of OP poisoning. During admission, the patient’s pupil diameter was smaller than normal, and she had decreased levels of cholinesterase. On the basis of these symptoms, we should be alert to encephalitis, cerebrovascular accident, or poisoning by opioids, carbamate insecticides, nereistoxin, toadstools, and other substances. The patient had been poisoned for 2 days when she was admitted to our hospital. Because the combination of carbamate pesticides and cholinesterase is reversible, their metabolism in the body is very fast. Generally, successful care allows patients to begin to recover in about 4 hours, returning to normal in about 24 hours. However, in this case, the patient suddenly suffered altered consciousness and acute respiratory failure 2 days after the poisoning, and craniocerebral CT excluded a diagnosis of acute stroke. At this time, acute OP poisoning should be strongly suspected. Thus, it is particularly important to carefully examine the patient’s medical history, check the physical findings in detail, and recognize the clinical manifestations and differential diagnosis of common poisonings. In addition, monitoring of toxic insecticides should be improved and insecticides used wisely. The use of pesticides should be planned well to avoid large stocks.
Footnotes
Ethical approval
Ethics approval was obtained from the Qilu Hospital Ethics Committee for Human Research (approval no. KYLL-2019-296). Written informed consent was obtained from the patient for publication of the case details and accompanying images.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
