Abstract
Objectives
To investigate liver function after pregnancy in women with chronic hepatitis B virus (HBV) and factors related to postpartum abnormalities.
Methods
A total of 317 pregnant women were included in this study and 138 had an HBV DNA level. In this trial, the highest number and proportion of hepatitis B surface antigen-positive mothers with postpartum hepatic inflammation were at 1 month after delivery.
Results
Baseline liver function of postpartum women with hepatic inflammation was significantly higher than that in those before delivery. The rates of hepatitis B e-antigen (HBeAg)-positive status, baseline HBV DNA levels, gestational diabetes mellitus, and antiviral therapy during pregnancy were significantly higher in the hepatic inflammation group than in the control group. Among the 138 women who received antiviral therapy, 83 withdrew from antiviral therapy immediately after delivery and 55 continued antiviral therapy for at least 1 month after delivery. Multivariate logistic regression analysis showed that HBeAg-positivity and gestational diabetes mellitus were associated with hepatic inflammation after delivery. Postpartum hepatic inflammation occurred mostly at 1 month after delivery in pregnant women with HBV infection.
Conclusions
Close monitoring of women with HBV during pregnancy is required, especially for those who are HBeAg-positive and have gestational diabetes mellitus.
Keywords
Introduction
Chronic hepatitis B virus (HBV) infection is still an important global health issue. Approximately 15% to 25% of patients who are infected by HBV may eventually develop liver cirrhosis, liver failure, or hepatocellular carcinoma. 1
In areas with a high prevalence of HBV, mother-to-child transmission is the main approach of transmission. 2 Postpartum vaccination of HBV and immunoglobulin can decrease the risk of perinatal transmission, but approximately 5% to 10% children still have HBV infection whose mothers have a high viral load.3–5 The use of nucleotide analogues in hepatitis B surface antigen (HBsAg)/hepatitis B e-antigen (HBeAg)-positive mothers who have a high viral load at the third stage combined with immunoprophylaxis of newborns at birth could significantly decrease the chance of HBV infection.6–9 This has been adopted by several chronic HBV management guidelines and is applied in clinical practice.10–12 Although attention has been paid to reducing HBV in the mother-to-child, little is known about the infection process of chronic HBV in pregnant women.
Pregnancy and puerperium have a close relationship with unique changes in the immune system, which may affect the natural history of autoimmune disease and immune-mediated infections. 13 These immunological changes might result in liver dysfunction, liver decompensation, and even maternal death. 14 Observational research has described that up to 50% of pregnant women who are infected with chronic HBV may experience alanine aminotransferase (ALT) flares (i.e., increased serum ALT levels that exceed the normal [ULN] within the first 6 months after delivery).15–17 Some studies have reported that the frequency and severity of flares are highly variable.18,19 However, some of these studies were limited by small sample sizes, which limited examining the potential risk factors for hepatitis flares. At present, the risk factors of postpartum hepatitis activity in pregnant women who are infected with HBV are still unclear. These data are important to ensure clinic correlation of recurrence of hepatitis in chronically infected pregnant women, and they should be informed of the need to monitor liver-related laboratory tests during and after pregnancy. Therefore, this study aimed to investigate the functional changes of the liver after delivery in pregnant women who are infected with chronic HBV, as well as the risk factors of hepatic flare.
Materials and methods
Patients
HBsAg-positive mothers who were admitted to the Department of Obstetrics and Gynecology of Beijing YouAn Hospital, Capital Medical University from January 2017 to December 2017 were included in this retrospective study.
The following inclusion criteria were used for the study: 1) women were aged 20 to 40 years; 2) women were infected with chronic HBV after diagnosis; and 3) a history of antiviral treatment was not recorded. HBsAg seropositive status and/or HBV DNA positivity at ≥6 months were considered chronic HBV infection. HBV-positive diagnostic criteria of patients were obtained in accordance with the Guideline of Prevention and Treatment for Chronic Hepatitis B (2015 Update). 12 The following exclusion criteria were used for the study: 1) women received antiviral therapy before pregnancy or accepted immune-modifying treatment during pregnancy; 2) there was other viral infection; 3) patients with hepatocellular carcinoma or liver decompensation; and 4) women had any immunologically compromised condition.
The trial was performed according to the ethical guidelines of the Helsinki Declaration of 1975. The study protocol was approved by the Institutional Review Board of Beijing YouAn Hospital, Capital Medical University (Approval No. 2019-3). The Institutional Review Board waived the need to sign informed consent because this research was a retrospective evaluation (Figure 1).
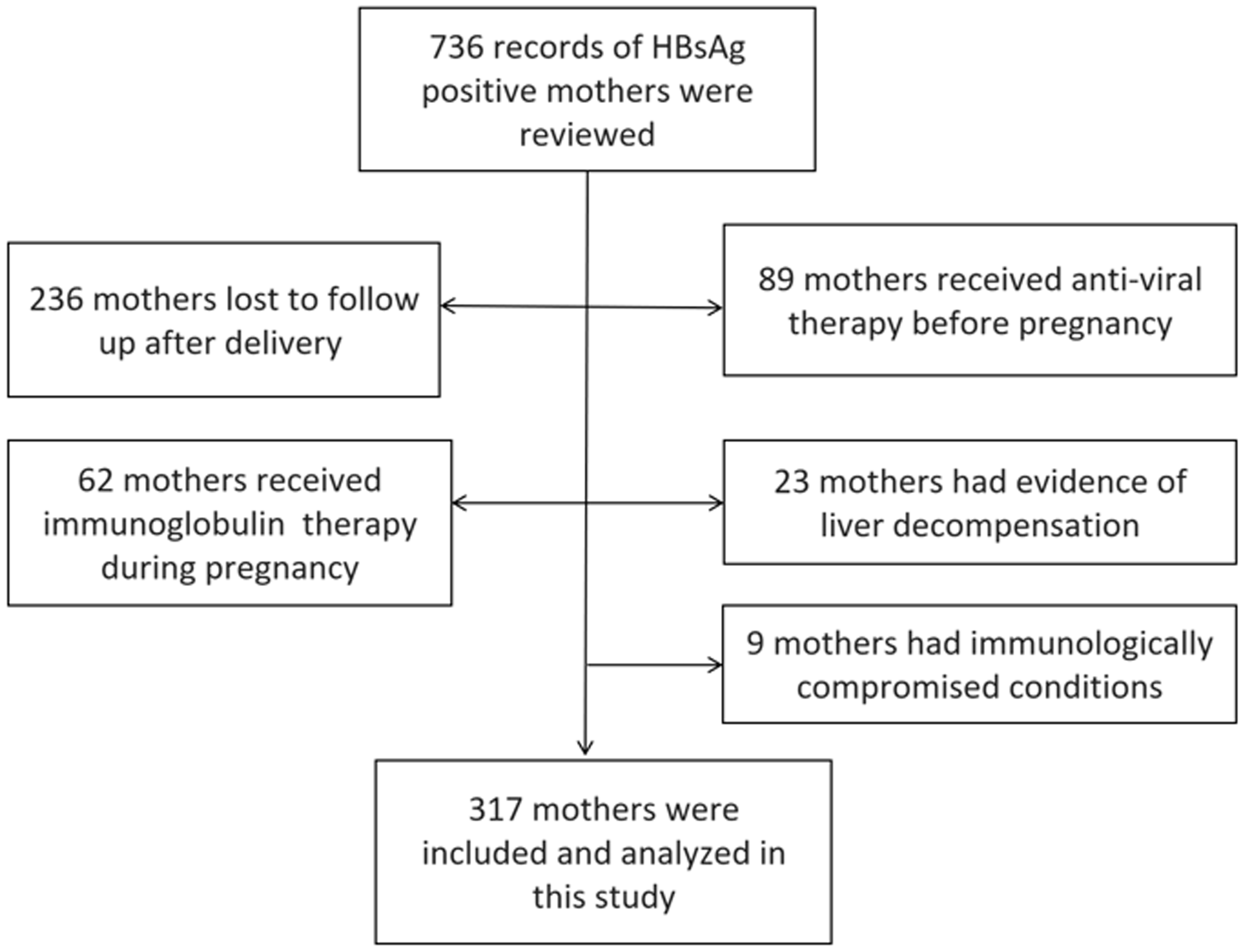
Enrolment of hepatitis B surface antigen (HBsAg)-positive mothers.
Laboratory methods
ALT, aspartate transaminase (AST), total bilirubin (TBIL), and direct bilirubin (DBIL) levels in mothers who were HBsAg-positive were detected by the Olympus Automatic Biochemical Analyzer AU5400 (Olympus, Tokyo, Japan). HBV seromarkers were measured by an electrical chemiluminescence immunoassay kit (Roche Laboratories, Penzberg, Germany). The level of HBV DNA was detected and measured by Beijing YouAn Hospital using the Cobas HBV Amplicor Monitor assay (Roche Diagnostics, Pleasanton, CA, USA). The test range for HBV DNA was 100 to 108 IU/mL.
Definition of hepatic dysfunction
Postpartum hepatic inflammation was defined as elevation of ALT levels to more than 1.5 times the ULN within 12 months after delivery (ULN: 40 U/L). A postpartum hepatitis flare was considered as elevated ALT levels to more than two times the ULN within 12 months post-delivery. A severe postpartum hepatitis flare was defined as an increase in ALT levels to more than five times the ULN within 12 months post-delivery.
Statistical analysis
EpiData 3.02 (EpiData Association, Odense, Denmark) was used to create a database. All data were input independently by at least two researchers, and another investigator checked the quality of data. The Wilcoxon signed rank test was used to compare continuous variables among groups, and the chi-square test or Fisher’s exact test was used to compare categorical variables. All data were analysed using the software IBM SPSS Statistics for Windows, version 24.0 (IBM Corp., Armonk, NY, USA). One-way analysis of variance was used to analyse the data. Data are expressed as the mean ± standard deviation. Statistical significance was defined as P<0.05.
Results
Characteristics of the patients
We included 317 mothers (Figure 1) and the mean age was 29.3±4.2 years. All of the mothers (317/317) were Han nationality. Among them, 46.4% (147/317) were HBeAg-positive. A total of 43.5% (138/317) of HBeAg-positive pregnant women who were infected with chronic HBV had an HBV DNA level >5 log10 IU/mL received antiviral therapy (Telbivudine, LDT; Novartis, Beijing, China) during pregnancy. All of them began to take antiviral therapy at 28±4 weeks of gestation.
Changes in maternal serum ALT levels at 12 months after delivery
Changes in maternal serum ALT levels from 1 to 12 months of delivery are shown in Table 1. The proportion of mothers with hepatic inflammation was significantly increased at 1 month after delivery compared with before delivery (5.7%, 18/317, P<0.001). A total of 16.7% (53/317) of mothers had hepatic inflammation (ALT levels ≥1.5×ULN). Postpartum hepatitis flare (ALT levels ≥2.0×ULN and <5.0×ULN) occurred in 9.8% (31/317) of women and 1.9% (6/317) had severe hepatitis flare (ALT levels ≥5.0×ULN) at 1 month after delivery. At 3 months after delivery, most mothers had normal serum ALT levels. After hepatoprotective treatment and antiviral therapy, hepatic function of these mothers gradually recovered. Twelve months after delivery, 98.1% (311/317) of mothers had normal serum ALT levels and the other six mothers continued to receive hepatoprotective treatment and antiviral therapy (Table 1).
Proportions and numbers of mothers with different serum ALT levels up to 12 months after delivery.

ALT, alanine aminotransferase; ULN, upper limit of normal.
Risk factors related to postpartum hepatic inflammation
To investigate the risk factors related to hepatic inflammation in pregnant women who were infected with chronic HBV after delivery, women in this trial were separated into the two following groups: the hepatic inflammation group (ALT levels ≥1.5×ULN) and the control group of hepatic function (ALT levels <1.5×ULN) based on hepatic function 1 month after delivery. Univariate analysis showed that baseline ALT and AST levels were significantly higher, and TBIL and DBIL levels were significantly lower in the hepatic inflammation group than those in the control group (all P<0.05). Additionally, the rates of HBeAg-positive status, baseline HBV DNA levels, gestational diabetes mellitus, and antiviral therapy during pregnancy were significantly higher in the hepatic inflammation group than in the control group (all P<0.05). However, the rates of hypertension syndrome, intrahepatic cholestasis of pregnancy, premature delivery, and caesarean section were not significantly different between the two groups (Table 2).
Univariate analysis of factors related to hepatic dysfunction at 1 month after delivery.
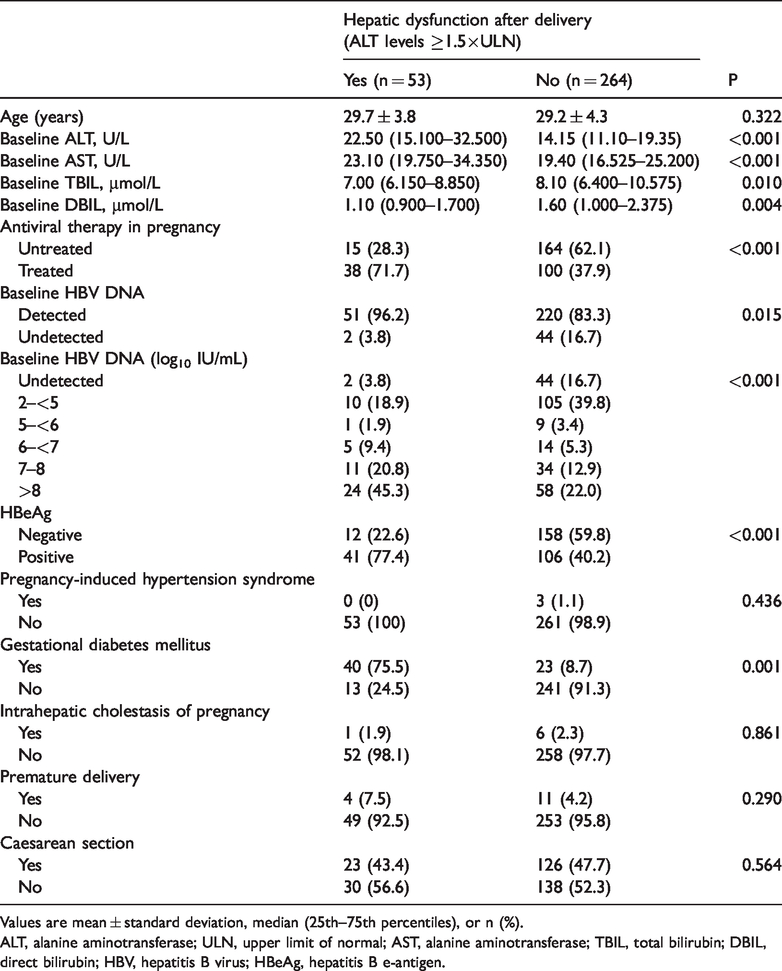
Values are mean ± standard deviation, median (25th–75th percentiles), or n (%).
ALT, alanine aminotransferase; ULN, upper limit of normal; AST, alanine aminotransferase; TBIL, total bilirubin; DBIL, direct bilirubin; HBV, hepatitis B virus; HBeAg, hepatitis B e-antigen.
According to univariate analysis results, antiviral therapy during pregnancy was a risk factor associated with hepatic inflammation after delivery. In this study, 138 HBeAg-positive pregnant women who were infected with chronic HBV received antiviral therapy during gestation. Among them, 83 stopped antiviral therapy immediately after delivery and 55 continued antiviral therapy for at least 1 month after delivery. We divided these women into two groups: the withdrawal group and the non-withdrawal group. The proportion of hepatic inflammation (ALT levels ≥1.5×ULN) at 1 month after delivery was 21.7% (18/83) in the withdrawal group and 36.4% (20/55) in the non-withdrawal group, with no significant difference between the groups (Figure 2).
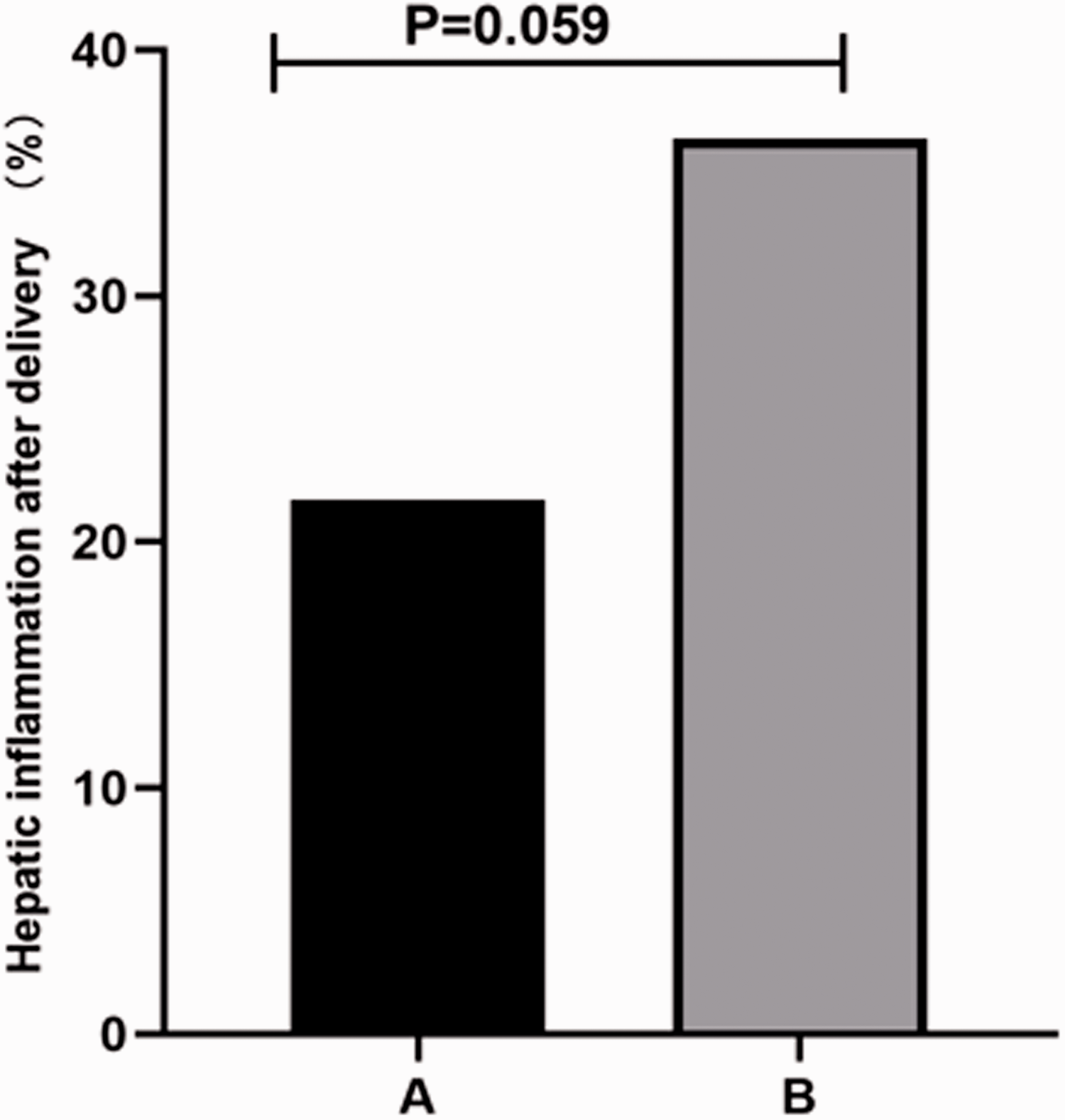
Proportion of hepatic inflammation at 1 month after delivery with or without withdrawal of antiviral therapy. A, withdrawal group; B, non-withdrawal group.
Baseline ALT, AST, TBIL, DBIL, and HBV DNA levels, HBeAg status , antiviral therapy during pregnancy, and gestational diabetes mellitus were further analysed using multivariate logistic regression. We found that only being HBeA positivity and gestational diabetes mellitus were significantly associated with hepatic dysfunction after delivery (both P<0.05, Table 3).
Multivariate analysis of hepatic dysfunction after delivery.

OR, odds ratio; CI: confidence interval; ALT, alanine aminotransferase; AST, alanine aminotransferase; TBIL, total bilirubin; DBIL, direct bilirubin; HBeAg, hepatitis B e-antigen; HBV, hepatitis B virus.
Discussion
We retrospectively investigated 317 pregnant women who were infected with chronic HBV. Changes in liver function after delivery in these women were observed. We found that the highest incidence of hepatic inflammation occurred at 1 month after delivery. At 3 months postpartum, liver function gradually returned to normal. Among patients who developed hepatic inflammation, the incidence of hepatitis flare occurred in 9.8% (31/317) of women and 1.9% (6/317) had ALT levels ≥5.0×ULN at 1 month after delivery. No women developed hepatic decompensation.
At present, there are few reports on follow-up of pregnant women who were infected with chronic HBV after delivery in China and overseas. In China, Wang et al. found that the incidence of elevated ALT activity postpartum was 62.2% in 114 pregnant women with chronic HBV infection 1 month after delivery. 20 At present, the cause of abnormal postpartum liver function is believed to be related to a change in immunological function in postpartum women. Maternal autoimmune function recovery and immune clearance after delivery can lead to an increase in hepatic enzyme activity. With the passage of time, immune tolerance is gradually improved, and the function of hepatocytes is restored. This immune clearance usually occurs 1 to 3 months after delivery.21,22 This timing is consistent with the results of our study, suggesting that the mechanism of postpartum liver dysfunction is related to immune clearance of HBV. The morbidity rate of hepatitis flare in our study is much lower than that in other studies of pregnant women who were infected with HBV. 15 , 16 , 20 This difference between studies might be due to differences in the research subjects of whom 43.5% (138/317) of patients in our study accepted antiviral treatment in pregnant women who were infected with chronic HBV. Additionally, the postpartum withdrawal time was different between studies, which might reflect different disease activities in the condition of pregnancy. Therefore, for pregnant women who are infected with chronic HBV, hepatic inflammation is prone to occurring within 1 to 3 months after delivery. Consequently, detecting indicators during this period is important, as well as timely intervention and treatment to prevent the occurrence of severe hepatitis.
In our study, we also analysed the factors that affect postpartum liver dysfunction. In univariate analysis, baseline ALT, AST, TBIL, and DBIL levels, HBeAg-positive status, baseline HBV DNA levels, gestational diabetes mellitus, and the antiviral treatment period affected postpartum hepatic inflammation. However, pregnancy-induced hypertension syndrome, intrahepatic cholestasis of pregnancy, premature delivery, and caesarean section were not related to postpartum liver dysfunction. In multivariate analysis, only HBeAg-positive status and gestational diabetes mellitus were associated with postpartum hepatitis flare. Some studies have shown that hepatitis flares or exacerbations are found in mothers with high ALT levels at delivery. 18 ,23,24 The possibility of postpartum liver dysfunction increases with an increase in ALT levels during pregnancy. Additionally, liver function during pregnancy may have clinical significance in assessing the risk of postpartum hepatitis flare. A previous study reported that a cut-off HBV DNA level of 5 log10 IU/mL at delivery predicted ALT levels. 20 However, in our study, although univariate analysis showed that postpartum liver dysfunction was associated with liver function during pregnancy and baseline HBV DNA levels, multivariate analysis did not show these associations. This finding may have been related to the method of grouping and the number of patients enrolled.
In this study, we found that the HBeAg-positive status was associated with postpartum hepatic inflammation, which is consistent with Kushner et al.’s study. 19 HBeAg positivity is typically related to high HBV DNA levels, which in turn may increase the risk of HBV flare. However, in multivariate logistic regression in our study, HBV DNA levels were not associated with postpartum hepatitis flare. We consider that the main cause of postpartum liver dysfunction was related to a change in immune function and not HBV DNA levels. Previous studies have observed spontaneous immune clearance, disappearance of HBeAg and/or seroconversion in mothers infected with HBV postpartum, and a decrease in the HBV DNA load. 22 ,25,26 These studies also suggested that postpartum immune recovery in women who are infected with HBV may induce immune clearance of HBV by the immune system. Therefore, HBeAg should be detected during pregnancy to perform closer surveillance of HBV for these patients.
Prenatal antiviral therapy for women with high viral loads is being supported because of increasing awareness of perinatal HBV transmission due to failure of immunoprophylaxis. Although the advantages of this therapy for the infant are well recognized, the effect on maternal health remains relatively unknown. At present, there are differences between domestic and foreign guidelines for therapy of chronic HBV in recommending the timing of postpartum withdrawal of antiviral treatment during pregnancy to block mother-to-child transmission.10–12 In our study, 138 HBeAg-positive pregnant women with a high viral load received antiviral therapy during pregnancy. Eighty-three women stopped antiviral therapy immediately after delivery and 55 mothers continued antiviral therapy for at least 1 month after delivery. We found that the proportion of hepatic inflammation was not significantly different between them. Therefore, we speculate discontinuing antiviral treatment after delivery within a short period might be safe. Stopping medication immediately after delivery can reduce maternal and infant exposure to antiviral drugs, reduce economic burden, and facilitate breastfeeding. However, we are not sure whether this is also safe in the long term. In the future, a large cohort study is required to confirm whether discontinuing antiviral therapy in pregnant women with HBV after delivery is beneficial.
Interestingly, we found that gestational diabetes mellitus was an important risk factor for hepatitis flare. The liver is the main location for intake, storage, production, and metabolism of glucose, and it plays an important role in maintaining stability of blood sugar. Some researchers have found that chronic HBV infection is a risk factor for gestational diabetes mellitus. 27 , 28 Che et al. reported that most pregnant women who have chronic HBV have a high HBV load, serious liver damage, and poor ability of the liver to regulate blood sugar compared with pregnant women who have chronic HBV and normal pregnant women. 29 Therefore, the occurrence of gestational diabetes mellitus is relatively serious. Consequently, for pregnant women who are infected with chronic HBV, management of pregnancy and blood sugar monitoring should be strengthened. Additionally, close monitoring and early intervention should be carried out to achieve the ideal level of blood sugar and help to reduce adverse pregnancy outcomes.
There are some limitations in this study. First, the study design was retrospective, which may have produced bias for collecting all electronic files of patients or pregnant women who were infected with HBV. Second, there is no generally accepted definition for obviously increased liver disease activity. Therefore, we used an ALT level ≥1.5×ULN. Finally, the number of available samples during and after pregnancy was limited. Prospective, large-sample cohort studies are required to solve these issues in the future.
Conclusion
Our retrospective cohort study of pregnant women who were infected with HBV showed that hepatic inflammation developed in 16.7% (53/317) of women within 1 month after delivery, and most were subclinical in nature. Positive HBeAg status and gestational diabetes mellitus are risk factors for hepatitis flare. Pregnant women who are infected with HBV with a high risk of flare-up should be closely monitored during pregnancy to be aware of this possibility as early as possible.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Science and Technology Key Project on Major Infectious Diseases such as HIV/AIDS, Viral Hepatitis Prevention and Treatment (No. 2017ZX10201201-001-001 and No. 2017ZX10201201-002-002), Beijing Municipal Science and Technology Commission (No. Z151100003915096 and No. Z161100000516084) and “Beijing Municipal Administration of Hospitals” Ascent Plan (No. DFL20151601).
