Abstract
Background
The causative virus of coronavirus disease 2019 (COVID-19) may cause severe fatal pneumonia. The clinical presentation includes asymptomatic infection, severe pneumonia, and acute respiratory failure. Data pertaining to acute renal injury due to COVID-19 in patients who have undergone renal transplantation are scarce. We herein report two cases of COVID-19 along with acute kidney injury following kidney transplantation.
Conclusion
Patients who develop COVID-19 after kidney transplantation are at risk of acute kidney injury, and their prednisone, immunosuppressant, and gamma globulin treatment must be adjusted according to their condition.
Keywords
Introduction
In December 2019, a series of cases of acute respiratory illness of unknown origin occurred in Wuhan, Hubei Province, China. 1 , 2 Because Wuhan is a city with one of the largest volumes of kidney transplantation in China, coronavirus disease 2019 (COVID-19) infection in renal transplant recipients deserves attention. 3 , 4
Factors that promote active invasive viral infection after organ transplantation include inflammation, graft rejection, tissue injury, and immune suppression. 5 , 6 In the current pandemic, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has been reported in kidney transplant patients from China and Spain. 3 , 4 , 7 Reduction of immunosuppressive agents along with low-dose methylprednisolone-based treatment regimens can improve COVID-19 pneumonia in renal transplant recipients with long-term immunosuppressive therapy. 3 Although little data are available on the incidence of COVID-19 in transplant recipients, preventive modalities and guidelines should be followed to prevent spread of the disease. 8 , 9 We herein report two cases of COVID-19 along with acute kidney injury (AKI) following kidney transplantation. Both patients were treated with immunosuppressant medication coupled with low-dose methylprednisolone-based therapy. Our intervention protocol thus serves as a template for the treatment of other such patients.
Case reports
Case 1
A 59-year-old man presented with an 8-day history of a fever and cough. The patient had a 20-year history of hypertension, for which he was undergoing treatment with oral valsartan and bisoprolol fumarate tablets. He also had a 6-year history of coronary heart disease, for which he was undergoing treatment with oral bisoprolol fumarate tablets and metoprolol. He had also been undergoing long-term maintenance immunosuppression with cyclosporine and mizoribine for 5 years after right kidney transplantation. These medications had been discontinued at the time of presentation. The patient’s wife had novel coronavirus pneumonia, and he had been in close contact with her. His peak body temperature was recorded at 38.2°C, and he exhibited dyspnea, headache, and myodynia; however, he had no palpitation, chest pain, chills, fatigue, or night sweats. He had a productive cough with white sputum. He had also experienced nausea without vomiting, abdominal pain, diarrhea, and exertional dyspnea without obvious aggravation, hemoptysis, or chest pain. The 2019-nCov nucleic acid test result was positive. The patient was hospitalized in an outside hospital 6 days prior to admission on 3 February 2020. A complete blood cell count was normal, and the total lymphocyte count was 1.28 × 109/L. Influenza A and B test results were negative. Computed tomography (CT) showed ground-glass opacities in the upper and lower regions of the left lung (Figure 1). The patient was administered arbidol hydrochloride tablets, cephalosporin, traditional Chinese medicine (Shuanghuanglian oral liquid and Lianhua Qingwen capsules), and antipyretic drugs from 4 to 8 February 2020.

Case 1: A 59-year-old man with fever, cough, and expectoration. Non-enhanced chest computed tomography showed multiple bilateral, small nodular ground-glass opacities and patchy ground-glass opacities with a subpleural distribution.
The patient’s vital signs on arrival were as follows: heart rate, 77 beats per minute; blood pressure, 136/76 mmHg; respiratory rate, 24 breaths per minute; peripheral capillary oxygen saturation, 82% (nasal cannula with 5 L/min oxygen flow); and body temperature, 36.5°C.
The patient’s body temperature and laboratory test results after admission are shown in Figures 2 and 3. His renal profile parameters were as follows: serum creatinine concentration, 166 µmol/L; blood urea nitrogen concentration, 9.2 mmol/L; and estimated glomerular filtration rate, 38.3 mL/min/1.73 m2. His angiotensin-converting enzyme (ACE) blood level was 28 U/L. The patient was negative for Mycoplasma pneumoniae, Chlamydia, respiratory syncytial virus, adenovirus, influenza A virus, influenza B virus, parainfluenza virus, and Legionella pneumophila immunoglobulin M antibodies. Laboratory testing also revealed the following: interleukin (IL)-1β, <5.0 pg/mL (reference range, <5.0 pg/mL); IL-2 receptor, 659 U/mL (reference range, 223–710 U/mL); IL-6, 57.47 pg/mL↑ (reference range, <7.0 pg/mL); IL-8, 16.4 pg/mL (reference range, <62 pg/mL); IL-10, 9.6 pg/mL↑ (reference range, <9.1 pg/mL); and tumor necrosis factor (TNF-α), 8.4 pg/mL↑ (reference range, <8.1 pg/mL). Factor G, galactomannan, and Candida mannan test results were negative. The patient was treated with supplemental oxygen using a nasal catheter at 5 L/minute.

Changes in critical laboratory test results during hospitalization (Case 1). There was a progressive increase in creatinine with a significant decrease in the estimated glomerular filtration rate after admission. With the progressive increase in leukocytes, the lymphocyte count dropped from 0.8 to 0.4 × 109/L and stabilized at this level. At the same time, the C-reactive protein level increased significantly to 100 mg/L on the fourth day of hospitalization, with platelets showing a downward trend. The dotted line in the figure refers to the reference range for these laboratory tests. SCr, serum creatinine; eGFR, estimated glomerular filtration rate; hs-CRP, high-sensitivity C-reactive protein; WBC, white blood cells; PLT, platelets.

Changes in body temperature (Cases 1 and 2). Case 1: The body temperature fluctuated. It was normal at the time of admission, increased to a fever in the daytime, and returned to normal the next day. On the fifth day of admission, the body temperature rose to 38.8°C and then returned to normal. Case 2: The body temperature was normal at the time of admission, and a remittent fever appeared the next day. From the seventh day of hospitalization, the body temperature dropped to normal and remained at normal level.
The day after admission, the patient’s blood oxygen level could not be maintained; therefore, he was treated with noninvasive ventilation (bilevel positive airway pressure) with the following parameters: spontaneous/timed mode; inspiratory positive airway pressure, 14 cmH2O; expiratory positive airway pressure, 8 cmH2O; fraction of inspired oxygen, 90%; and f = 12 times/minute. A 1440-mL injection of fat emulsion amino acids (17) and glucose (11%) was administered for intravenous nutritional support. The patient’s blood pressure was controlled by pump injection of urapidil hydrochloride (injection of 20 mL of urapidil via infusion pump followed by dilution to 50 mL), and the urine volume was controlled by furosemide (40-mg intravenous infusion). Three days after admission, the patient developed acid reflux, heartburn, chest distress and suffocation, progressive dyspnea, and progressive deterioration of renal function while undergoing the treatment. His renal function was reassessed (Figure 2), revealing a serum creatinine concentration of 467 µmol/L, blood urea nitrogen concentration of 25.8 mmol/L, estimated glomerular filtration rate of 11 mmol/L, bicarbonate concentration of 12.7 mmol/L, potassium concentration of 5.41 mmol/L, alanine aminotransferase concentration of 61 IU/L, aspartate aminotransferase concentration of 87 IU/L, albumin concentration of 38.4 g/L, and high-sensitivity C-reactive protein concentration of 96.7 mg/L.
The patient appeared to be in a state of delirium at 5 days after admission. His oxygen saturation dramatically dropped to 10%, and he developed cardiac arrest without restoration of spontaneous circulation. Because his family did not want tracheal intubation to cause pain to the patient, invasive rescue was not performed.
Case 2
A 37-year-old woman presented with an 8-day history of a fever and cough. The patient also had a 1-year history of hypertension, for which she was undergoing treatment with 47.5-mg metoprolol tartrate tablets once daily and 30-mg nifedipine controlled-release tablets once daily. In 2014, she was diagnosed with immunoglobulin A nephropathy via renal biopsy and underwent hemodialysis for 3 months in 2015. She was selected as a candidate for kidney transplantation and maintained her creatinine concentration at 130 to 140 µmol/L postoperatively. She was treated with prednisone (20 mg once daily), tacrolimus (2 mg/morning, 1.5 mg/night), and mycophenolate mofetil.
The patient’s peak body temperature reached 40°C before hospitalization, and this fever was accompanied by chills, dry cough without phlegm, mild dyspnea, muscle pain, dizziness, headache, chest pain, chest distress, and pharyngeal pain. The patient visited Wuhan Union Hospital, where a chest CT scan revealed novel coronavirus infection in the lower region of her left lung on the day of presentation (Figure 4), and her 2019-nCov nucleic acid test result was positive. The patient was administered arbidol hydrochloride tablets from 5 to 13 February, moxifloxacin from 5 to 13 February, ceftriaxone from 5 to 12 February, and propylene spheres from 8 to 12 February. Her symptoms were thereafter alleviated, and her body temperature remained nearly stable for 3 days; however, her chest CT scan indicated deterioration on 12 February 2020.

Case 2: A 37-year-old woman with fever and cough. Non-enhanced chest computed tomography at onset of symptoms showed multiple bilateral, peripheral, patchy ground-glass opacities with a subpleural distribution and slight thickening of the adjacent pleura and interlobular septa.
The relevant changes in the patient’s laboratory examination results and body temperature are shown in Figures 3 and 5. Routine urinalysis showed 2+ red blood cells and 2+ protein. Biochemical tests showed a serum creatinine concentration of 167 µmol/L, blood urea nitrogen concentration of 7.7 mmol/L, estimated glomerular filtration rate of 33.4 mL/min/1.73 m2, and bicarbonate concentration of 19.5 mmol/L. The mycophenolate mofetil was stopped after admission. Tacrolimus was discontinued 10 days later, although methylprednisolone at 40 mg once daily and moxifloxacin were continued. Furosemide (40 mg) was administered according to the patient’s daily urine volume. The serum creatinine concentration exhibited a progressive upward trend. The patient rested well; received adequate nutrition, oxygen inhalation, and gamma globulin infusion; avoided nephrotoxic drugs; and received appropriate diuretic treatment according to the urine volume. The methylprednisolone therapy was changed from 40 mg once daily to oral prednisone 25 mg twice daily, and this was gradually reduced to 5 mg once daily. Eleven days after admission, tacrolimus (1.5 mg in the morning, 1 mg in the evening) was added to the treatment regimen. The patient had a satisfactory clinical recovery with respect to body temperature, absorption of lung lesions (Figure 6), and renal function (Figures 3 and 5).

Changes in critical laboratory test results during hospitalization (Case 2). The patient in Case 2 had stage 3 chronic kidney disease at the time of admission, and on the ninth day of hospitalization, she showed a progressive decrease in the serum creatinine concentration with the estimated glomerular filtration rate returning to 50 mL/min/1.73 m2. After admission, the leukocytes fluctuated from 1.0 to 8.0 × 109/L, and the platelets increased from 120 to 220 × 109/L. The lymphocytes decreased from 0.4 to 0.2 × 109/L and continued to rise to 1.5 × 109/L on the seventh day. In addition, the C-reactive protein level decreased to normal after peaking at 35 mg/L on the fifth day. The dotted line in the figure refers to the reference range for these laboratory tests. SCr, serum creatinine; eGFR, estimated glomerular filtration rate; hs-CRP, high-sensitivity C-reactive protein; WBC, white blood cells; LYM, lymphocytes; PLT, platelets.
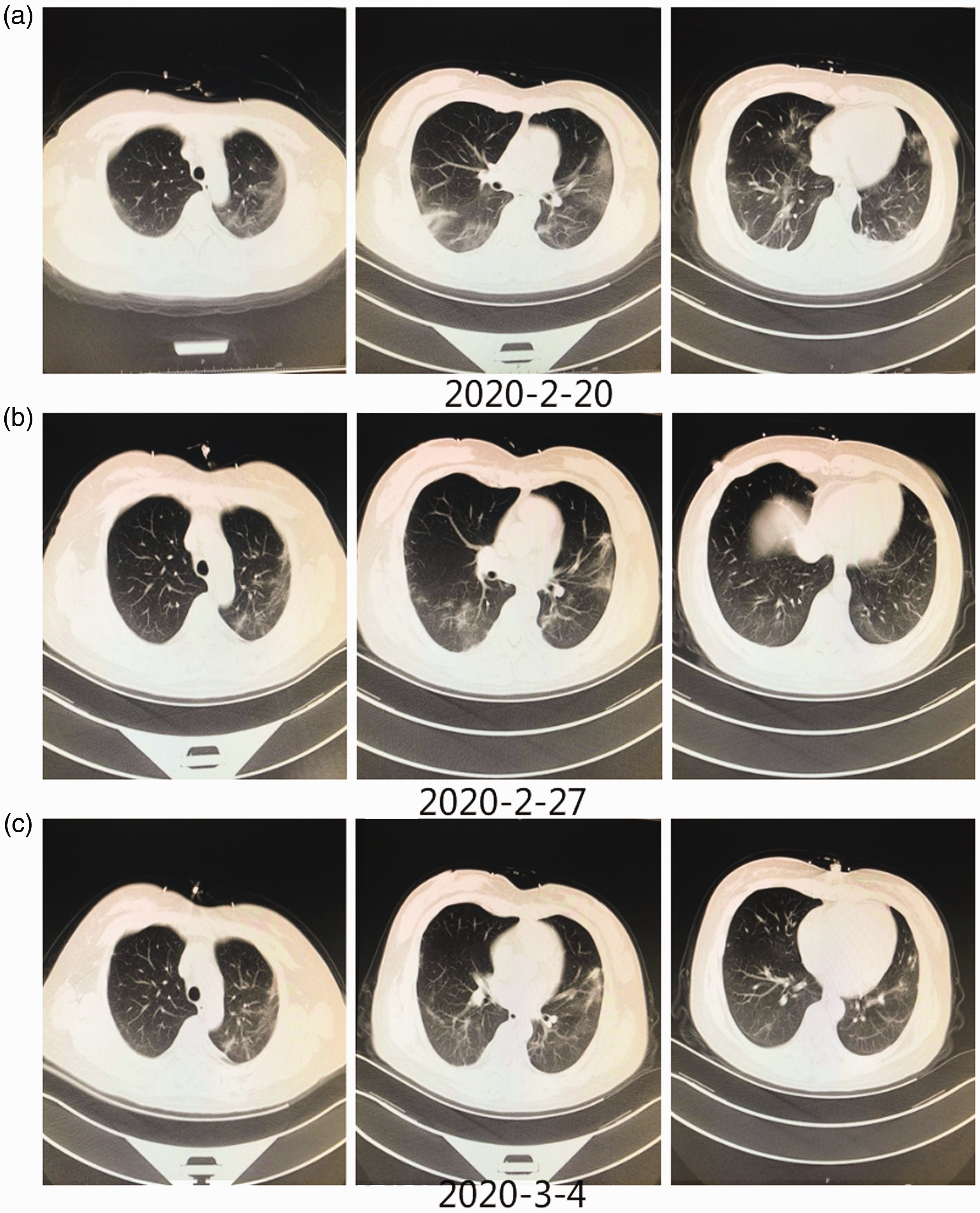
Chest computed tomography (CT) images (Case 2). (a) Chest CT in the axial plane shows multiple mixed ground-glass opacities and linear opacities in the bilateral lung lobes. (b) Chest CT in the axial plane shows improvement of the pneumonia with decreased ground-glass opacities and linear opacities in the subpleural area. (c) Chest CT in the axial plane shows minimal absorption of both the ground-glass opacities and linear opacities in the bilateral lung lobes.
Ethics statements
Informed consent was obtained from the patients for the collection of their medical history and blood samples in compliance with the Declaration of Helsinki. This report was approved by the local ethics committee (Peking University Third Hospital Medical Science Research Ethical Committee Approval No. IRB00006761-M2020060). The patients also provided informed consent for publication of their cases.
The clinical and research activities being reported are consistent with the Principles of the Declaration of Istanbul as outlined in the Declaration of Istanbul on Organ Trafficking and Transplant Tourism. The study procedures were approved by the institutional review board (IRB) at Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology (IRB approval number: TJ-C20200120).
Discussion
We have herein described two patients with allograft kidneys who developed COVID-19, one of whom died of the disease and the other of whom cleared the infection. The disease course, symptoms, radiologic findings, and key laboratory values are described for each patient. The number of patients in this study is too small to draw meaningful conclusions; however, because such cases are rare, it is important to describe them in the literature so that a meta-analysis can eventually be performed.
Pulmonary infection is one of several major infections that may occur after renal transplantation and is characterized by its occult onset, rapid progression, and serious complications. 10 , 11
The causative virus of COVID-19 binds to human ACE2 to enter the host cells. One of the activities of ACE2 is hydrolyzing the active bradykinin metabolite [des-Arg973] BK (DABK). Decreased activity or reduced expression of ACE2 by the virus impairs the inactivation of DABK. This enhances signaling through the bradykinin B1 receptor and can lead to fluid extravasation and leukocyte recruitment to the lung. Targeting the bradykinin system by either blocking bradykinin production or blocking bradykinin receptors may open a new therapeutic window for the treatment of COVID-19–induced acute respiratory distress syndrome, particularly before patients enter the irreversible stages. 12
Coronavirus-induced AKI is common and mainly manifests as renal tubular injury. In their recent report of 1099 patients with COVID-19, Guan et al. 13 indicated that the incidence of AKI was 0.5% and that 5 of 173 severely affected patients showed AKI (2.9%). The most recent investigation by Cheng et al. 14 showed that the incidence of AKI was 3.2% among 710 consecutive patients diagnosed with COVID-19. Nevertheless, the mechanism of how coronavirus leads to AKI remains unclear. As indicated by the above-mentioned recent study, 14 SARS-CoV-2 particles are large and must actively crawl through all three layers of the glomerular filtration barrier to reach Bowman’s space. 15 SARS-CoV-2 particles can also be identified in the cytoplasm of podocytes by electron microscopy, causing collapsing focal segmental glomerulopathy. 16 From Bowman’s space, SARS-CoV-2 particles easily flow into the proximal tubules, where they enter the ACE-2–rich tubular epithelium. Several studies have shown clusters of SARS-CoV-2 particles individually or wrapped by cell membranes in the cytoplasm of cells in the proximal tubules as evidence of direct viral infection.17,18 With respect to immune activation, earlier studies have shown that both severe acute respiratory syndrome-related coronavirus and Middle East respiratory syndrome-related coronavirus infections can remarkably raise the levels of inflammatory factors and chemokines. 19 The major secretions include chemokines, proinflammatory cytokines, and inducible nitric oxide synthase through the M1 subtype of macrophages to form cytotoxic peroxynitrite and mediate renal injury. Moreover, dendritic cells release TNF-α for additional promotion of the inflammatory response. 20 A recent report indicated that cytokine storm syndrome plays a crucial role in various types of infection-mediated multiple organ failure. 21 It can promote the activation of a cytokine cascade reaction and release large numbers of cytokines to cause systemic inflammation. In the present cases, the TNF-α, IL-1, and IL-6 levels in both patients increased remarkably, indicating that the patients may have had cytokine storm syndrome. Chen et al. 22 reported that in a group of 29 patients with COVID-19, IL-6 and granulocyte-macrophage colony-stimulating factor caused further release of inflammatory factors by activating monocytes, thus amplifying the immune effects and eventually causing AKI.
Kidney involvement in patients with COVID-19 is not uncommon, but the results of clinical studies are conflicting, and very few biopsy-based studies are available. 14 , 23 Most studies have suggested that the tubulointerstitial compartment of the kidney is the predominant site of injury. More recently, however, case reports (mostly from the United States) have interestingly detected the development of collapsing glomerulopathy in the setting of COVID-19 infection.16,24,25 The first renal biopsy-based report on renal pathology in COVID-19 raised the possibility that APOL1 high-risk alleles may augment the risk of renal injury in patients with COVID-19. 26
Both patients described in this report had severe COVID-19 at the time of admission. The patient in Case 1 had heart failure, high age, and hypertension, which contributed to the development of chronic kidney disease. The patient in Case 2 had hypoxemia and hypertension at the time of admission. Moreover, both patients were using antibiotics and antiviral drugs. All of these unfavorable risk factors could lead to renal tubular injury, increasing the risk of AKI. Importantly, we should be careful to identify signs of allograft rejection in renal transplant patients. We speculated that allograft rejection might have occurred in Case 2, although a renal biopsy was not performed.
Given the extent of lymphocytes that are consumed by SARS-CoV-2, 27 mycophenolate mofetil treatment was withdrawn in the second patient. Moreover, the glucocorticoid and tacrolimus dosages were also adjusted. Whether SARS-CoV-2 can be inhibited by tacrolimus is unclear, although replication of SARS-CoV was diminished after tacrolimus treatment in one report. 28 Notably, cyclosporine, another calcineurin inhibitor, may be used as a host-directed therapy for COVID-19. 29 Interestingly, kidney impairment was detected in Patient 2, implying the possibility of acute rejection. After the creatinine concentration increased, the patient recovered with a small dose of methylprednisolone, resulting in a decrease in the creatinine concentration. Aggressive reduction or withdrawal of immunosuppressants should be considered with caution for recipients with severe pneumonia or acute respiratory distress syndrome. 28 More importantly, renal function should be monitored frequently because renal injury resulting from acute rejection could make treatment more difficult.
The general population is also susceptible to the novel coronavirus; it is not a conditional pathogen, which differs from the common infections that occur after kidney transplantation. Therefore, in contrast to opportunistic pathogens, immunosuppressants should be removed in a step-by-step instead of radical manner. Intravenous administration of small doses of methylprednisolone is recommended because such treatment can help to prevent rejection and reduce inflammation. Close observation of the urine volume and serum creatinine concentration is needed, and biopsy can be performed if necessary.
COVID-19 is caused by a coronavirus and has become a major public health hazard to human society. COVID-19 in combination with AKI is a clinical problem that not only increases the mortality rate but may also cause chronic kidney disease; therefore, it calls for immediate attention. Because such investigations are just beginning, active exploration and multidisciplinary cooperation are essential.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ contributions
Qiuyu L. wrote and revised the original draft. Qiuyu L., Ning S., and Qinggang G. contributed to the design of the work. Qiuyu L., Qin C., Zhiling Z., Chao L., Junhong W., Shu L., Ning S., and Qinggang G. performed the data acquisition and analysis. Qiuyu L., Qin C., Zhiling Z., Lan Z., Wei G., Chao L., Junhong W., Shu L., Ning S., and Qinggang G. contributed to data interpretation. Qiuyu L., Lan Z., and Wei G. were responsible for creation of the new software used in the study. Nini D. contributed to revision of the manuscript. Qinggang G. is the corresponding author of this article. All authors read and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81900641), the Medjaden Academy & Research Foundation for Young Scientists (Grant No. nCoV_MJA20200228), the Special Campaign to Fight Novel Coronary Pneumonia (COVID-19)(2020) of Subject Construction Project of Peking University Health Science Center (Grant No. BMU2020HKYZX011), the Fundamental Research Funds for the Central University: the Special Research Fund of PKU for Prevention and Control of COVID-19 (Grant No. BMU2020HKYZX011), and the National R&D Program of China (Grant Nos. 2020YFC0844500 and 2020YFC0844900).
