Abstract
Objective
Human urinary kallidinogenase (HUK) is a glycoprotein extracted from human urine that is used to treat stroke by triggering positive regulation of the kallikrein–kinin system. Our aim was to evaluate the efficacy and safety of HUK treatment for acute ischemic stroke.
Methods
We searched the online databases PubMed, Embase, Cochrane Library, Google Scholar, and China National Knowledge Infrastructure (CNKI) for papers published between January 2015 and December 2019. The quality of each trial was assessed using the Cochrane Reviewers’ Handbook. Randomized controlled trials of HUK in patients with acute ischemic stroke were included.
Results
Sixteen trials with 1326 participants were included. The HUK injection groups had more neurological improvement than the control groups in National Institutes of Health Stroke Scale scores (mean difference, –1.65; 95% confidence interval [CI], –2.12 to –1.71) and clinical efficacy (1.30; 95% CI, 1.21 to 1.41). Subgroup analysis indicated that age may influence heterogeneity. Eleven trials reported adverse effects and there were no significant differences between the control and HUK groups (risk difference, 0.01; 95% CI, –0.02 to 0.04).
Conclusions
HUK ameliorates neurological symptoms in stroke patients with few adverse effects. Further high-quality, large-scale randomized trials are needed to confirm these results.
Keywords
Introduction
Stroke is a leading cause of morbidity and mortality worldwide, and is a main cause of severe long-term disability in older people. Acute ischemic stroke (AIS), caused by the obstruction of a blood vessel that supplies blood to the brain, is the most common type of stroke, and accounts for nearly 80% of all kinds of stroke. 1
Human urinary kallidinogenase (HUK) is a glycoprotein extracted from human urine that triggers positive regulation of the kallikrein–kinin system, thus catalyzing the hydrolysis of low molecular weight kininogens to vasoactive kinins. 2 Numerous laboratory studies have indicated that HUK treatment promotes post-ischemic angiogenesis and cerebral perfusion via the activation of bradykinin B1 and B2 receptors. 3 Furthermore, HUK is approved by the State Food and Drug Administration of China, and has been used clinically in China to treat stroke patients for over 10 years. However, as a relatively newly developed drug, the retail price of HUK is very high compared with traditional therapies. Hence, it is very important to evaluate the clinical efficacy and safety of HUK treatment for AIS patients.
Methods
We followed the PRISMA guidelines to carry out this meta-analysis of randomized controlled trials (RCTs), in which we assessed the efficacy and safety of HUK injection for AIS patients. All analyses were based on previously published studies; thus, no ethical approval or patient consent was required.
Search strategy
We searched the online databases PubMed, Embase, Cochrane Library, Google Scholar, and China National Knowledge Infrastructure (CNKI) for papers published between January 2015 and December 2019, to identify all eligible studies. The search process was conducted independently by two reviewers (P.W. and Y.Z.). The following MeSH terms were used: “human urinary kallidinogenase”, “HUK” and “ischemic stroke”. We also manually searched relevant journals and conference proceedings (Figure 1).
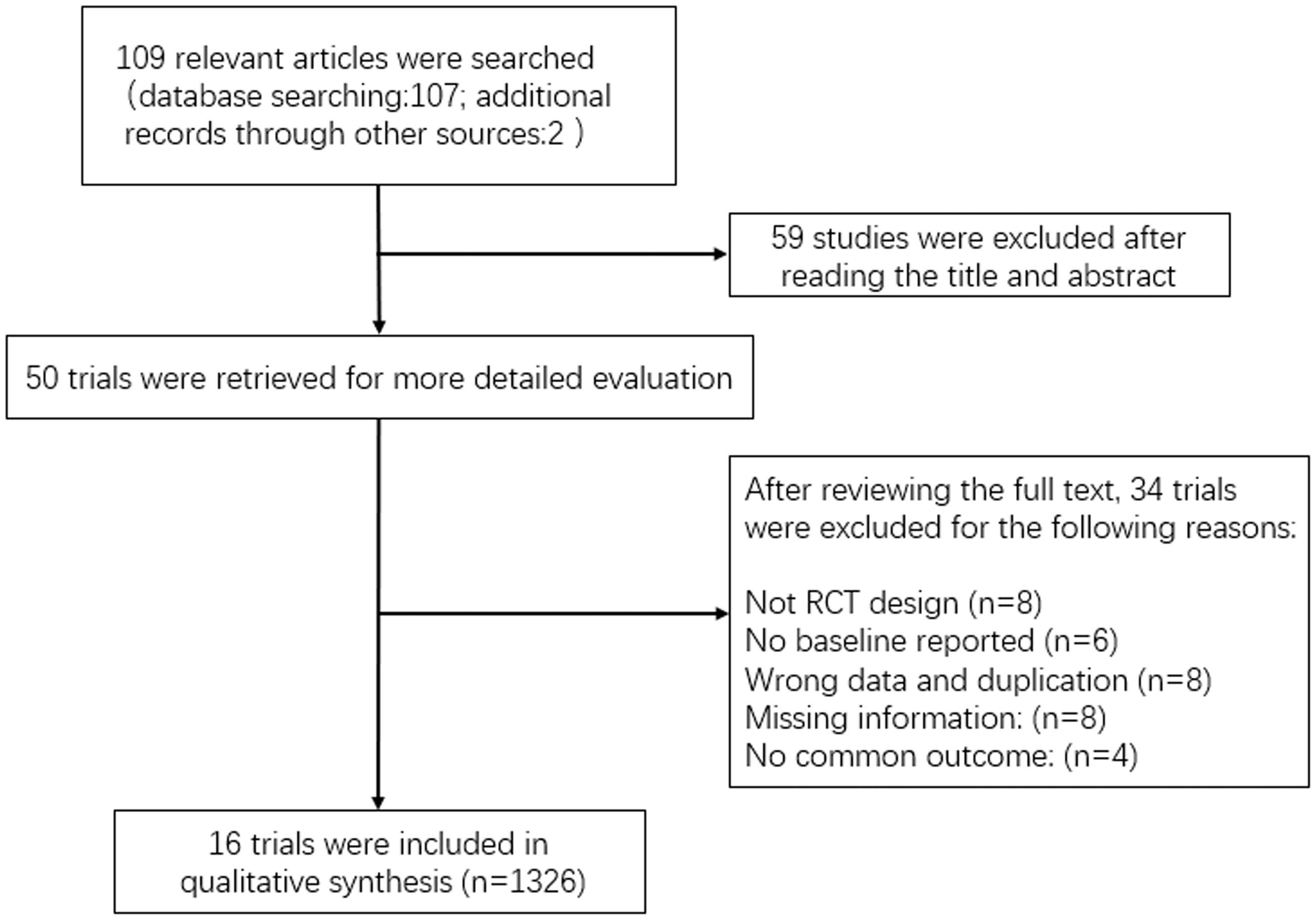
Flow diagram for the literature search and selection.
Eligibility criteria
Original full-text articles and study reports were included in this meta-analysis if they met the following criteria: [a] all study participants, regardless of position, race, region, and sex, were diagnosed with definite AIS from either brain CT or MRI examination; [b] study designs were RCTs comparing HUK therapy with conventional therapy for the recovery of stroke patients; [c] studies had data of clear outcomes after treatments; and [d] studies were published in English or Chinese. The outcomes extracted from the original trials included assessments of neurological improvement in the National Institutes of Health Stroke Scale (NIHSS) and clinical efficacy.
Data extraction
Two reviewers independently extracted the data from each study. The information was collected as follows: the trial and authors’ names; publication year; sex and mean age of the participants; relevant scores related to the interventions; outcomes of clinical efficacy for each group; and adverse effects in the two arms.
Quality assessment
The methodological quality of the included studies was evaluated using the Cochrane Collaboration’s risk of bias tool 4 , which assesses the following domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessments, incomplete outcome data, selective reporting, and other potential biases for each included study. Each domain was classified into one of three groups: “low risk,” “unclear,” or “high risk.” In the random sequence generation domain, trials that only mentioned “randomization” or “randomly,” without a description of randomization methods, were allowed. For the sensitivity analysis, merged trials were excluded one by one to see if the synthesis result changed significantly. Minor changes indicated that the synthesized result was stable. If an observable difference appeared, we reassessed the study and make a cautious decision about the position. The publication bias was assessed using a funnel plot.
Data analysis
Results for dichotomous outcomes between HUK injection groups and control groups were expressed as risk ratios (RRs) or risk differences (RDs) with 95% confidence intervals (CIs). Results for continuous outcomes were expressed as mean differences (MDs). (RevMan 5.3 software). Chi-squared tests (level of significance = 0.05) were used to analyze trial heterogeneity and to quantitatively determine the magnitude of heterogeneity in combination with I2. If no significant heterogeneity or low heterogeneity was observed, a fixed-effects model was used. If there was a large amount of statistical heterogeneity, a random effects model was used following exclusion. Subgroup analyses were performed when heterogeneity was very high.
Results
Search results
A total of 109 relevant articles were identified by searching the electronic databases. Of these, 59 articles were excluded after reviewing their titles and abstracts. After further reviewing, 16 studies (
Descriptions of studies
The 16 studies, involving a total of 1326 participants, were all conducted in mainland China (Table 1). The average age of subjects in the included trials ranged from 52.5 to 71.5 years old, and the number of participants in each study ranged from 30 to 200. HUK was administered intravenously each day at a dose of 0.15 PNAU in all the treatment groups. The duration of HUK treatment ranged from 7 to 14 days. There were four studies that had a different duration of HUK treatment from the other studies. Basic treatment included aspirin or clopidogrel to suppress platelet aggregation, atorvastatin to reduce blood lipids, and some traditional Chinese medicine treatments to facilitate circulation. Co-interventions with other medications were only permitted if they were administered equally to both arms of the trial. All studies reported comparable baseline characteristics and assessed neurological deficits before treatment. In addition, all studies used NIHSS scores to measure neurological deficits, and nine reported the clinical efficacy of the different groups after treatment.
Characteristics of included studies.
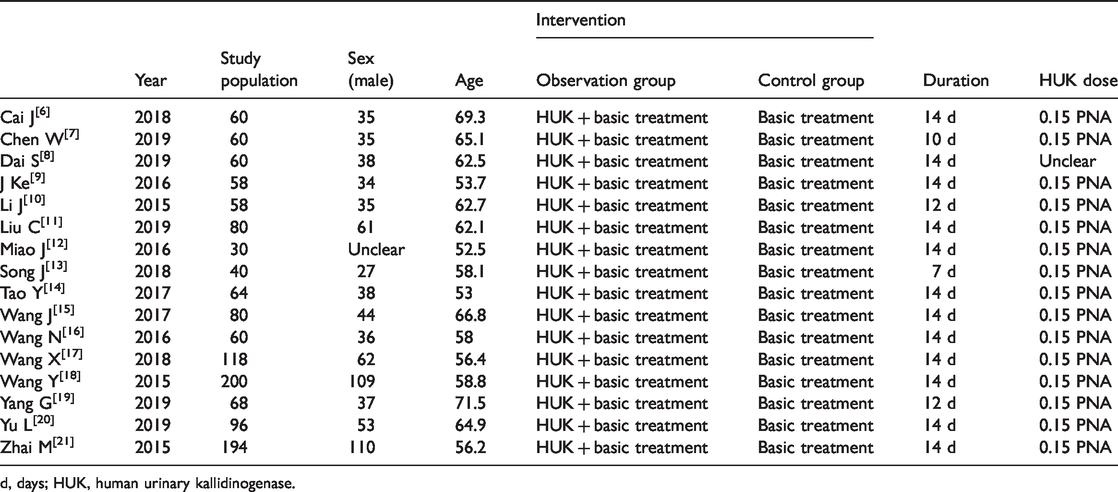
d, days; HUK, human urinary kallidinogenase.
Effects of interventions
Dichotomous variables on the clinical efficacy of neurological deficits were available from nine trials with 676 participants. A fixed effects model was used because the trials had no significant heterogeneity (

Forest plot comparing clinical efficacy of neurological deficits at the end of treatment.

Forest plot comparing National Institutes of Health Stroke Scale (NIHSS) scores. The subgroup analysis was based on average age.
Adverse effects
The HUK groups were associated with nearly the same number of adverse events as the control groups, and there was no statistical difference between the two groups (11 studies; RD, 0.01; 95% CI, –0.02 to 0.04;
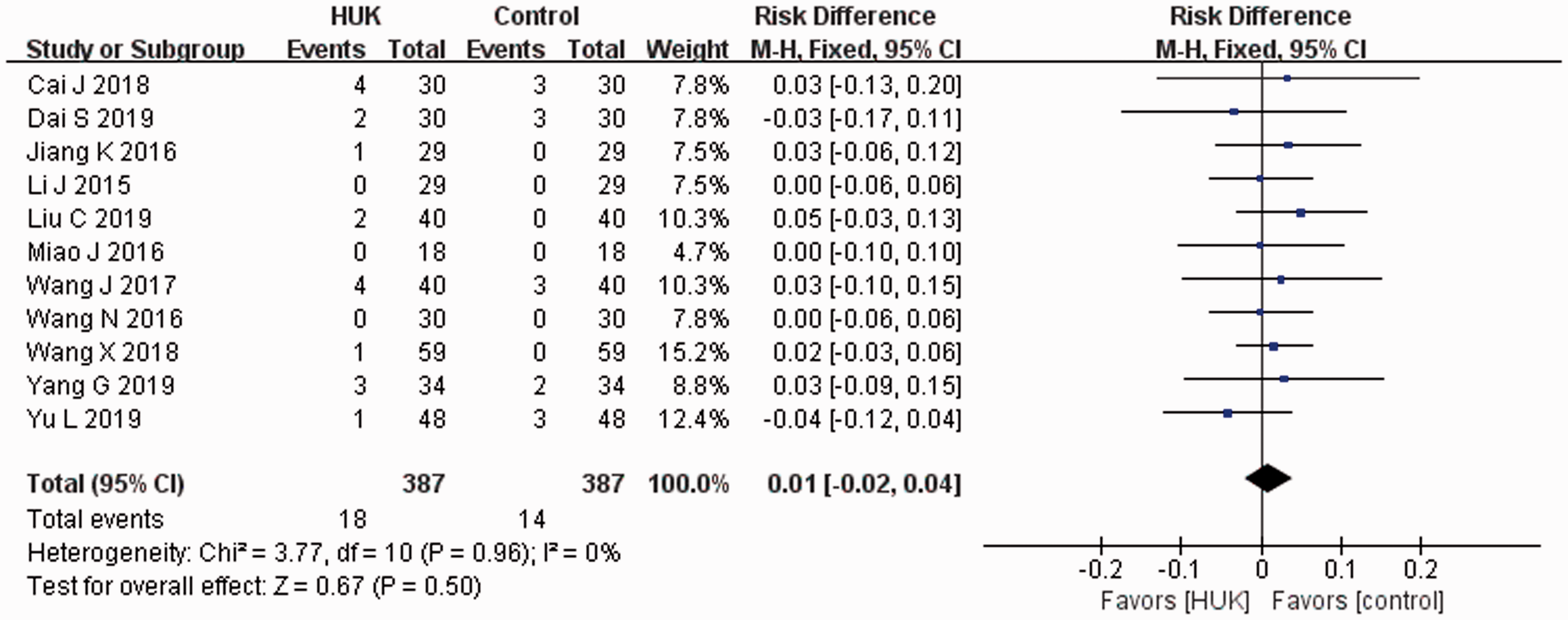
Forest plot comparing adverse effects during treatment.
Sensitivity and risk of bias
The merged studies were excluded one by one. Except for the studies by Ke et al. 8 and Yu et al., 19 there were no significant differences in the outcome profiles between the HKU and control groups after excluding each of these studies. All of the included trials claimed randomization, but no study mentioned allocation concealment. Only one study (Zhai 20 ) reported the use of a single-blind procedure. Selective reporting was suspected in some of the trials. In general, the majority of the studies were deemed to have a relatively high risk of bias based on the Cochrane Collaboration’s risk of bias tool (Figure 5). We also used a funnel plot to investigate potential publication bias, which revealed some potential publication bias in this meta-analysis (Figure 6). The two points that were relatively far from the dotted line were from the studies by Ke et al. 9 and Yu et al. 19
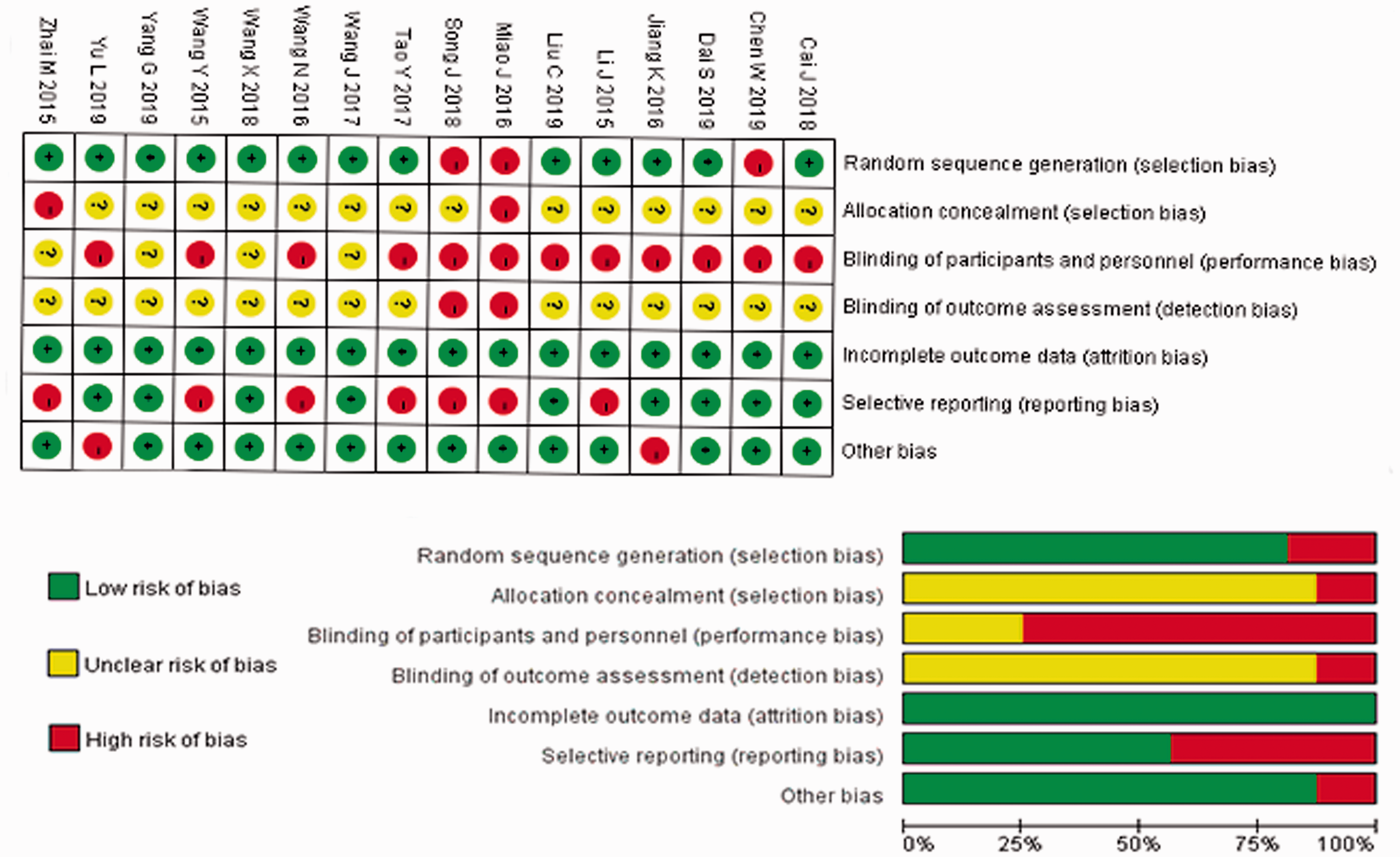
Risk of bias for all included trials. Low, unclear, and high risks are represented with the following symbols, respectively: “+”, “?”, and “−”.
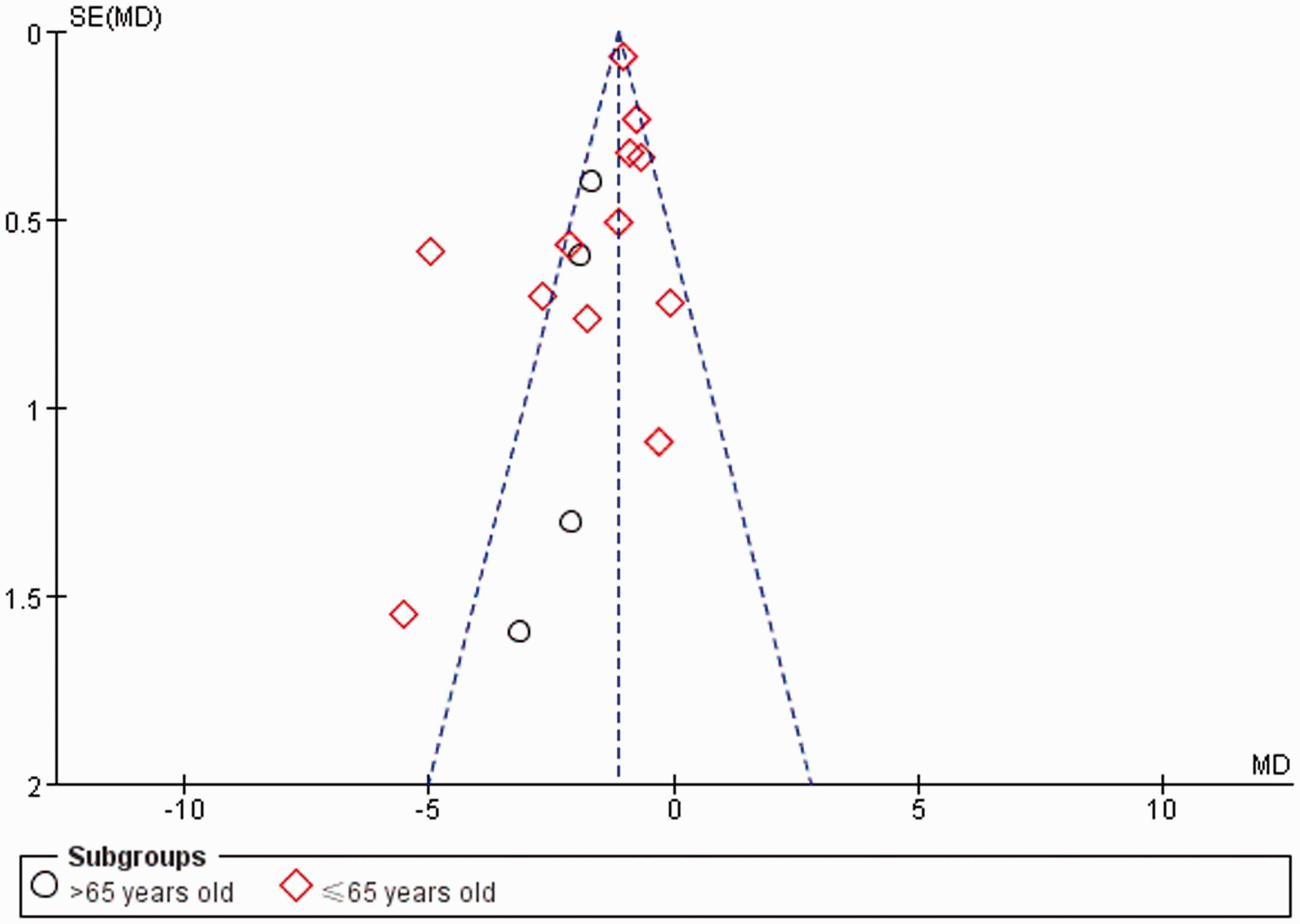
Funnel plot comparing publication bias in all included trials.
Discussion
The aim of this meta-analysis was to assess the curative effects and safety of HUK in the treatment of AIS. Our results revealed that HUK led to marked efficacy of recovery from neurological deficits after stroke, with few adverse effects. Both the NIHSS scores and clinical efficacy rates suggested that patients in the HUK groups had better outcomes after treatment compared with the control groups.
HUK is a relatively new first
We searched electronic databases to find research about the clinical use of HUK. Interestingly, all of the trials that we found showed that early HUK injections for AIS patients led to better neurological recovery (RR, 1.30; 95% CI, 1.21 to 1.41; I2 = 0%). This finding was confirmed by the outcomes measured by NIHSS scores (MD, –1.65; 95% CI, −2.12 to −1.71; I2 = 80%). We then performed a subgroup analysis according to average age. The older group showed more benefits and less heterogeneity in NIHSS scores compared with the younger group (MD, –1.84 vs. –1.58; I2 = 0% vs. 84%), which suggests that HUK may have more potential value in treating older people with AIS. The studies by Ke et al.
8
and Yu et al.
19
both had high NIHSS scores before and after treatment; this may be the result of different subjective judgments for scoring. By removing these two trials from the analysis, the heterogeneity dropped significantly (χ2 = 15.04,
Some limitations should be noted when referring to the results of this meta-analysis. First, all 16 trials were conducted in China, leading to a limitation of general applicability. Second, the general methodological quality of the included trials was not optimal, particularly with respect to the lack of placebo-controlled and double-blind trials. Further high-quality RCTs of the clinical efficacy of HUK for AIS are needed. Aside from professional knowledge and statistical approaches, the price of HUK may be another limitation impeding high-quality RCT trials in large populations. However, HUK was enrolled in the directory of Chinese healthcare insurance in 2019, so more multi-center, double-blind, high-quality RCTs may emerge in the near future.
Conclusion
Based on the current evidence, HUK ameliorates neurological deficits in stroke patients with few adverse effects. Age may be a factor influencing the heterogeneity of its effects. Further high-quality, large-scale randomized trials are needed to confirm these results.
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520943452 - Supplemental material for Efficacy and safety of human urinary kallidinogenase for acute ischemic stroke: a meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_0300060520943452 for Efficacy and safety of human urinary kallidinogenase for acute ischemic stroke: a meta-analysis by Yuanxiang Huang, Binglei Wang, Yue Zhang, Peize Wang and Xiangjian Zhang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_0300060520943452 - Supplemental material for Efficacy and safety of human urinary kallidinogenase for acute ischemic stroke: a meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_0300060520943452 for Efficacy and safety of human urinary kallidinogenase for acute ischemic stroke: a meta-analysis by Yuanxiang Huang, Binglei Wang, Yue Zhang, Peize Wang and Xiangjian Zhang in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
