Abstract
After primary dissemination of Mycobacterium tuberculosis bacillus infection that is localized in liver, disease progression and changes to immune function in the body occur. Various forms of tuberculosis, including granuloma, caseous necrosis, liquefactive necrosis, fibrosis, and calcification, occur that could be presented at different stages, and imaging examination shows findings that are consistent with these stages. Not all liver tuberculosis patients are suitable for liver resection, and preoperative imaging examination and pathological immunohistochemical results could be used to determine whether tuberculosis was active, avoid unnecessary liver resection, and prevent the postoperative spread of tuberculosis. Here, we reported a case of miliary tuberculosis, pelvic tuberculosis, and tuberculous abscess of the thigh muscle in a 51-year-old man after liver lesion resection. The liver lesion was confirmed to be tuberculosis by surgical pathology, which is rare and has not been previously reported. The purpose of this case report is to remind radiologists of the importance of the floral-like enhancement and to estimate whether liver tuberculosis is active. This will help to guide clinicians to determine the timing of surgery, avoid unnecessary liver resection, and avoid hematogenous transmission.
Introduction
Primary involvement of liver tuberculosis (TB) is rare, and it is difficult to diagnose in the absence of a previous history of TB or concurrent pulmonary involvement. It is usually misdiagnosed as a neoplastic liver lesion, which misdirects the treatment protocol, delays proper treatment, and causes unnecessary damage.1,2 Using a combination of ultrasonography, computed tomography (CT), magnetic resonance imaging (MRI), and pathological examination is of great help and significance to diagnose and classify this disease. The pathological staging of this disease could be evaluated, and the treatment measures could be adjusted accordingly to avoid unnecessary surgery. This paper describes the case of a patient with hepatic TB with hematogenous dissemination after hepatic TB surgical incision and abscess drainage, which resulted in miliary TB and lumbar TB, followed by pathological fracture, TB of the hip joint, and TB muscle abscess.
Case report
A 51-year-old male patient who complained of pain in the right upper abdomen and subxiphoid process for 1 week without obvious cause attended our hospital. The pain was accompanied by fever, nausea, fatigue, abdominal distension, belching, and loss of appetite, which was accompanied by dizziness, headache, and body temperature up to 39°C. Since the onset of the disease, the patient’s mental condition was good, his appetite was fair, and he had no obvious abnormal defecation or weight loss. He had no medical history of hepatitis, TBs, or cancer. He had no history of contact with an epidemic area, epidemic situation, or epidemic water, and he did not live in a pastoral area. Physical examination revealed the following characteristics: aspartate aminotransferase (66 U/L [normal range, 8–40 U/L]) was slightly higher than normal and glutamyl transferase was increased (131 U/L [normal range, 11–50 U/L]). Because his liver function was seriously damaged, there was an increase in total bilirubin (83.1 μmol/L [normal range, 3.4–20.5 μmol/L]) and a prolongation of the activation time of partial thrombin (APTT) (45.6 s [normal range, 28.0–43.5 s]). The hepatitis complete set (qualitative), anti-HIV, and syphilis antibody test results were negative. A complete set of tumor markers showed elevated ferritin. Parasite antibody tests and TB antibody test results were negative.
After admission, chest X-ray examination was performed on 24 May 2017, and the result was normal (Figure 1). An abdominal CT examination (Figure 2a–f) was performed on 26 May 2017. To more comprehensively evaluate the features of the lesions, make a preoperative qualitative diagnosis, and allow for better surgical planning, pre-contrast and contrast-enhanced MRI examinations were performed 3 days after the CT examination. CT findings revealed multiple patchy hypodense lesions in the liver, and an irregular hyperdense calcification (Figure 2a–d) was seen at the center of the lesion that showed hyperdense calcified foci in the bone window of the abdomen axial CT (Figure 2f) (T1 hyposignal and T2 hyposignal on MRI [Figure 3a–h]). A contrast-enhanced image showed the hypodense lesions around the calcification, and the periphery of the lesion showed a floral ring delayed enhancement. The main pathological features were fibrous encapsulations that had formed on the periphery of the lesion in the upper segment of the right anterior lobe (VIII) and the lower segment of the right posterior lobe (VI). Because the calcification at the center of the lesion and the fibrous encapsulation around the lesion are characteristic of chronic infectious lesions (calcification and fibrous encapsulation), the lesion was placed in the context of a chronic infection. Thus, we mostly considered the chronic infectious lesions with calcification and enlargement of the hilar lymph node.

Chest X-ray showed a normal distribution and shape of the lung, normal texture, no obvious exudation, and no nodule or mass opacity.

(a) Axial CT revealed patchy hypodensity lesions in the upper segment of the right anterior lobe of the liver (VIII) and irregular calcification was seen in the center. (b,c,d) Axial contrast-enhanced CT showed hypodensity around the calcification, and the edge of the lesion showed a floral ring (floral-like) enhancement. (e) During the balance period of CT dynamic enhancement, a filling defect (red arrow) was seen in the small branch surrounding the lesion that was further separated from the right branch of the portal vein. (f) The bone window of an abdominal axial CT showed a hyperdensity calcification foci (red arrows) in the upper segment of the right anterior lobe of the liver (VIII).

(a) Axial T1-weighted image showed that the lesion was slightly hyposignal, (b) axial T2-weighted image showed that the lesion was slightly hypersignal in the upper segment of the right anterior lobe (VIII) of the liver, with a range of about 3.8 × 4.2 × 4.7 cm, and T1 hyposignal and T2 hyposignal calcification was seen in the center of the lesion. (c–h) Contrast-enhanced MRI showed a mild floral ring (floral-like) enhancement at the lesion edge. (i, j) Diffusion-weighted imaging (DWI) revealed a lesion was bright on DWI and dark using the apparent diffusion coefficient (ADC).
Interferon-gamma release (IGRA) tests were not widely used in our region, and thus, this patient did not undergo IGRA tests. Because this was considered to be a chronic infectious disease that had middle-to-late stage lesions, the possibility of TB was considered. TB antibody (IgG antibody) is usually produced in the mid-to-late stage of TB, so TB antibody testing was appropriate. Additionally, the antibody was recognized as a conventional auxiliary index for diagnosis of TB. Thus, TB antibody was tested on 8 June 2017, but it was negative. Then, on 16 June 2017, to understand the pathological changes in the tumor and to make a qualitative diagnosis, the doctors decided to operate on the patient and remove parts of the abscess wall during the operation and send it for frozen pathology. On the same day, the patient underwent liver lobectomy (the right lobe of the liver was surgically removed via an anterior-approach hepatectomy) and liver abscess incision. During surgery, the patient was diagnosed with a TB hepatic abscess and it was then drained. After leaving the hospital on 4 July 2017, the patient underwent symptomatic treatment including antimicrobials to prevent infection; vitamin A, vitamin B, vitamin C, glycyrrhizin, and tiopronin to protect the liver; and an acid inhibitor to inhibit acid-fast bacilli.
The patient was reexamined on 9 January 2018, and chest CT showed multiple fine nodules in both lungs (Figure 4a) and bilateral pleural thickening and adhesions. Thus, infection and TB were considered. MRI showed a slight reduction in the previous lesion (VI, VIII), but a new lesion appeared, which was a T1 hyposignal and a T2 hypersignal lesion in the lower segment of the right anterior lobe of the liver (V), which was bright on diffusion-weighted imaging (DWI) and dark using the apparent diffusion coefficient (ADC). The contrast-enhanced image showed a ring enhancement at the lesion edge. On 13 March 2018, abdominal CT showed multiple hypodense lesions in the liver, and multiple spot-like calcifications in the lesions (Figure 4b), which suggested TB. On 29 March 2018, a pelvic CT was performed, and these results showed multiple areas of patchy and moth-eaten patterns of bone destruction in the lumbosacral coccyx vertebral body and accessories, as well as in the pelvis, bilateral femoral head, and neck (Figure 4c). CT bone window images of the pelvis showed multiple focal osteolytic lesions in the vertebra, sacrum, bilateral ilium, pubis, ischium, and femur (Figure 4d–f), which was considered to be caused by TB. The shape of the L4 vertebral body became flat and the continuity of the marginal bone cortex was interrupted, and a pathological fracture was considered. On 30 March 2018, a plain MRI scan of both hip joints showed narrowing of the joint space, uneven signals in the bilateral ilium, pubis, ischium, and femur, and multiple patchy T1 hyposignal and T2 hypersignal lesions inside both hip joints. Fluid was also seen inside the hip joint. Obvious contrast enhancement was visible (Figure 4g–k), considering that the pelvis was filled with multiple focal osteolysis lesions, and no bone repair was observed. This was considered to be secondary osteolysis and TB. Soft tissue edema was observed in the right pelvic floor muscle, right hip, and right thigh, and patchy mixed signal lesions with unclear boundaries and ring enhancement were observed in the right lateral femoral muscle area (Figure 4l–m). These were considered to be TB abscesses. The pathological examination showed granulomatous inflammation with necrosis, and acid-fast staining revealed acid-fast bacilli. Thus, a diagnosis of liver TB was considered (Figure 5a–e).
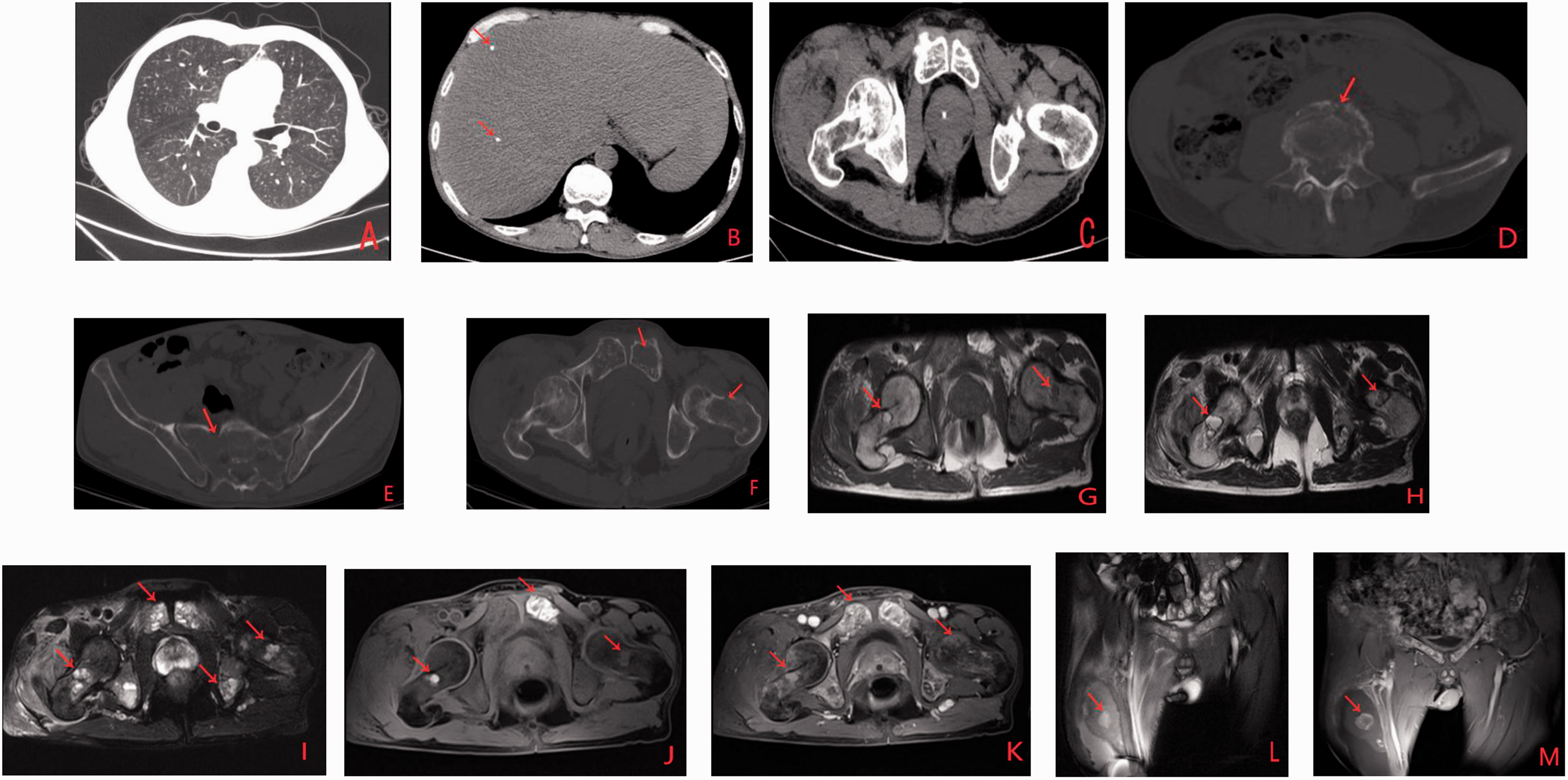
(a) Chest axial CT showed multiple fine nodules in both lungs. (b) Abdominal axial CT showed multiple spot-like calcifications. (c) The pelvic axial CT showed multiple areas of patchy and moth-eaten pattern bone destruction. (d–f) CT bone window images of the pelvis showed multiple focal osteolytic lesions (red arrows) in the vertebrae, sacrum, bilateral ilium, pubis, ischium, and femur. (g–i) Axial T1- and T2-weighted MRI, pressure fat T2-weighted MRI showed the interarticular space was narrowed and multiple focal osteolytic lesions were seen in bilateral ilium, pubis, ischium, and femur. (j–k) Axial contrast-enhanced MRI showed obvious enhancement. (l) Coronary T2-weighted MRI showed that soft tissue edema was found in the right pelvic floor muscle, right hip, and right thigh, and a TB abscess was found in the right lateral femoral muscle area. (m) Coronary contrast-enhanced MRI showed ring enhancement.

(a–e) Histopathological examination of the liver specimen showed granulomatous inflammation with caseous necrosis, lymphocytes, multinucleate giant cells, and epithelioid cells. (a, b: lymphocyte; c: epithelioid cell; d: caseous necrosis; e: multinucleate giant cell).
Additional tests were performed and are described below. Immunohistochemistry test results were as follows: CD68 (+), CD163 (+), CD34 (vascular +), Ki67 (+), and LcA (+). CD68 expression is a marker of bone marrow and nerve tissue macrophages in lymphoid tissue. Ki67 (+) results indicate that the tumor shows rapid proliferation and a high degree of malignancy, and it is associated with a poor prognosis. CD34 is a marker of vascular endothelial cells, which indicates vascular invasion. LcA is a lymph tissue-derived marker and a first-line marker antibody of lymphoreticular cells. The final diagnosis was that the hematogenous dissemination after hepatic TB surgery had resulted in miliary TB and lumbar TB, followed by pathological fracture, TB of the hip joint and TB muscle abscess.
Discussion
TB infection is one of the most important causes of death and morbidity in the world, especially in tropical regions. About 15% of people infected with HIV were co-infected with TB, and the TB is distributed both inside and outside the lungs, making it one of the leading causes of death in this population. In 2015, there were an estimated 10.4 million new cases of TB 3 with the highest incidence in developing countries. Although most of the cases were the source of transmission, about half of the patients denied a previous history of TB infection, and more than half showed normal findings on chest radiography.4–10 The ratio of men to women in this disease is 2:1, but isolated hepatic TB was more common between 40 and 60 years of age.11–13
The most common clinical symptoms and signs were right upper abdominal pain, upper abdominal tenderness, low-grade fever, night sweats, weight-loss and fatigue, abdominal masses, hepatomegaly, and jaundice. Abnormal liver function and positive tuberculin test results were found when laboratory tests were performed.14,15 Pathological manifestations included granuloma, caseous necrosis, liquefactive necrosis, fibrosis, and calcification. Based on pathological classification, hepatic TB could be divided into the following types: parenchymal, serohepatic, and TB cholangitis. 14 The parenchymal type is further divided into miliary TB, primary (isolated) hepatic TB, nodular TB, TB hepatic abscess, and mixed TB.7,14 Among them, isolated hepatic TB is rare.
Miliary TB is the most common hepatic TB subtype, and it is found in 80% to 100% of pulmonary TB patients at autopsy. It presents as multiple or diffuse miliary micronodular lesions in the liver (CT diameter, 0.5–1.0 cm), and it is pathologically characterized by small and isolated yellow-white nodules in the liver, non-specific inflammatory responses, focal Kupffer cell proliferation, caseous necrosis, and epithelioid nodules. A plain CT image showed diffuse spotty calcification in the liver. Nodular TB could present as nodular structures, which were named for their ball-like shape and which are similar to TB bulbs that measure 1 to 4 cm in diameter and often become calcified. In different pathological stages, gray-scale two-dimensional ultrasound examination of nodules might show many different manifestations, including hypoechoic, hyperechoic, mixed heterogeneous, anechoic, and strong echo with sound shadow. 4 Lesions of this subtype are rare, and they were associated with miliary or micronodular lesions. The center of the lesion showed caseous necrosis, and granulation tissue would gradually appear during the progression of the lesion, with fibrous capsule formed at the edge. TB can maintain a long-term static state, and sometimes it can also be in a nodular liver abscess that is in a liquefied form. Our patient’s presentation was similar to this type.
CT results for our patient showed an isolated lesion in the upper segment of the right anterior lobe of the liver (VIII), with irregular calcification at the center and a floral ring enhancement around the lesion, which suggested fusion of miliary nodules. Pathologically, the calcification at the center of the lesion corresponds to the recovery stage of TB, while the small peripheral lesions correspond to the newly developed granulation tissue. Thus, the enhancement can be seen in the early stage, indicating an abundant blood supply. The edge was covered by the fibrous envelope, so that the enhancement continued in the later stage. Mixed TB, 14 which is the typical CT findings of this type of hepatic TB, might comprise multiple and diverse dense lesions, indicating nodular granuloma, liquefactive necrosis, fibrosis, or calcification in hepatic TB at different pathological stages. In this case, hepatic TB included the miliary type, nodular abscess type, and mixed type, and there were different pathological stages. A plain CT image showed a hyperdense area at the center of the lesion and pathology indicated that calcification had developed by caseous necrosis in the corresponding area. The rim of the lesion showed circular hypodensity, which was suggested to correspond to granulation tissue in pathology. There were also multiple spotty calcifications in the liver. Contrast enhancement showed that the central hypodensity of the lesion was not enhanced and the granulation tissue around the lesions was slightly enhanced, and there was a floral ring enhancement around the lesion, which suggested fusion of the miliary nodules. During the balance period of CT dynamic enhancement, a filling defect was seen in the small branch surrounding the lesion that was further separated from the right branch of the portal vein (Figure 2e). The TB lesion was suggested to have invaded into the blood, which means that TB was active and spread easily. A plain MRI image showed hyposignal lesions in the center of the lesion, with a peripheral hyposignal rim on T1-weighted image; hyposignal, isosignal, or hypersignal at the center of the lesion; and a peripheral hypersignal rim on T2-weighted images. Contrast enhancement showed floral-like enhancement around the lesion.
Immunohistochemistry showed CD68 (+), CD34 (vascular +), LcA (+), and Ki67(+), which suggest invasion of nerve, bone marrow, blood vessels, and lymphoid tissues, with rapid tumor cell, high malignancy, and poor prognosis. This might also be a sign of an active TB lesion. Based on the initial imaging findings, this case was considered an infectious lesion. As the disease progressed, it was confirmed to be hepatic TB after pathological examination, and it had developed into a hepatic TB abscess. Because of the surgical incision and abscess drainage, the hematogenous dissemination after hepatic TB surgery resulted in miliary TB and lumbar TB, followed by pathological fracture, TB of hip joint, and TB muscle abscess over the following few months.
If the imaging examination (CT or MRI) combined with image-guided fine needle aspiration biopsy could clarify the diagnosis of TB, then unnecessary surgery could be avoided and conservative anti-TB drug treatment could be used. 1 However, the patient finally chose to be transferred to another hospital for treatment, so this patient did not receive systematic anti-TB treatment at our hospital. If similar cases are encountered in the future, multiple imaging examinations could be combined for comprehensive consideration to determine the stage of TB (with special attention paid to the floral-like enhancement). The corresponding treatment measures could then be implemented to avoid unnecessary surgical injury and postoperative complications or the situations of systemic TB dissemination.
Conclusion
Preoperative imaging examination and the floral-like enhancement might reflect active liver TB lesions, and systemic hematogenous dissemination by unnecessary surgery could be avoided. Multiple imaging examinations combined with fine needle biopsy are of great value in diagnosing hepatic TB. Because imaging findings of liver TB lesions vary, they are easily confused with other liver space-occupying lesions. Most of these patients underwent surgical resection, which causes unnecessary damage. With the development of medical technology, CT, MRI, and immunohistochemical testing could be used to provide more effective diagnostic information, which could reduce unnecessary damage and thereby avoid postoperative systemic spread of TB foci.
Footnotes
Confidentiality statement
We have de-identified all data to ensure patient confidentiality.
Consent
The written consent was obtained from the patient in this case.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics
This is a clinical case report on a patient’s subjective request for admission to hospital for diagnosis and treatment. It is not a prospective study or retrospective work, so ethics approval was not necessary and this case report was not approved by the hospital ethics committee.
