Abstract
Objective
To compare the treatment outcome of venovenous extracorporeal membrane oxygenation (VV-ECMO) versus mechanical ventilation in hypoxemic patients with acute respiratory distress syndrome (ARDS) at a referral center that started offering VV-EMCO support in 2010.
Methods
This retrospective cohort study enrolled adults with severe ARDS (PaO2/FiO2 ratio of <100 with FiO2 of ≥90 or Murray score of ≥3) who were admitted to the intensive care unit of Siriraj Hospital (Bangkok, Thailand) from January 2010 to December 2018. All patients were treated using a low tidal volume (TV) and optimal positive end-expiratory pressure. The primary outcome was hospital mortality.
Results
Sixty-four patients (ECMO, n = 30; mechanical ventilation, n = 34) were recruited. There was no significant difference in the baseline PaO2/FiO2 ratio (67.2 ± 25.7
Conclusion
Although VV-ECMO promoted lower-TV ventilation, it did not improve the in-hospital mortality rate.
Keywords
Introduction
Acute respiratory distress syndrome (ARDS) is defined by acute onset of worsening respiratory symptoms, severe hypoxemia, and bilateral pulmonary infiltration that cannot be explained by cardiac failure or volume overload. 1 Despite advancements in ARDS management strategies, the mortality rate remains high in Thailand and worldwide, especially among patients with refractory hypoxemia.2–4 In recent years, venovenous extracorporeal membrane oxygenation (VV-ECMO) support has been successfully used to improve outcomes among selected patients with severe ARDS.5–7
A few decades ago, VV-ECMO support among patients with severe acute respiratory failure was reportedly associated with poor outcomes. 8 Recent evidence from cohort studies and randomized controlled trials has shown that VV-ECMO support among patients with ARDS who develop refractory hypoxemia is associated with improved outcomes.5–7 However, the current guideline from the American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine does not include a definitive recommendation for or against the use of VV-ECMO in adults with severe ARDS. 9 The reported improvement in ECMO-associated outcomes might be partially explained by improvements in ECMO device technology. However, the more likely reason that outcomes have improved is that physicians’ skills have been enhanced and experience has been gained in ECMO cannula insertion and general management, which has dramatically lowered the rate of complications associated with ECMO support. 10 Studies from the United Kingdom, Australia, and New Zealand showed that transfer of adults with severe ARDS, especially those with influenza A (H1N1) infection, to a high-experience ECMO center for VV-ECMO support was associated with improved survival outcomes. 5 , 6 There is currently a lack of data specific to the outcome of VV-ECMO support versus conventional ARDS management in a recently established VV-ECMO service setting.
Siriraj Hospital (Bangkok, Thailand) is a 2,300-bed university-based teaching hospital that is affiliated with Mahidol University, and it is Thailand’s largest national tertiary referral center. At this center, VV-ECMO support has been available as a rescue therapy for refractory hypoxemia since 2010; however, the case volume since the introduction of VV-ECMO therapy has been less than six cases per year. This low case volume has limited the speed with which the physicians have gained experience in VV-ECMO cannula insertion and long-term ECMO care. The objective of this study was to compare the treatment outcomes of VV-ECMO support versus conventional mechanical ventilation in patients with severe ARDS who developed refractory hypoxemia at Siriraj Hospital from 2010 to 2018.
Methods
Study design and population
This single-center retrospective cohort study was conducted in a 15-bed medical intensive care unit (ICU) of the Division of Critical Care, Department of Medicine, Faculty of Medicine Siriraj Hospital from January 2010 to December 2018. The study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) recommendations. 12 We screened all adult patients aged >18 years who were admitted to our ICU to identify those who met the criteria for diagnosis of ARDS according to the 2012 Berlin criteria for the diagnosis of ARDS. 1 Of these, patients who had an indication for ECMO support because of refractory hypoxemia were enrolled. The definition of refractory hypoxemia was a PaO2/FiO2 ratio of <100 with an FiO2 of ≥90% despite optimal mechanical ventilator support and paralytic agent infusion according to the 2013 Extracorporeal Life Support Organization (ELSO) guideline 11 for at least 2 hours. Optimal mechanical ventilator support was defined as a low tidal volume (TV) (6 mL/kg of ideal body weight) with positive end-expiratory pressure (PEEP) titration. A paralytic agent was infused to prevent patient–ventilator dyssynchrony. For patients with an oxygen saturation of <90% or pH of <7.20, the TV could be increased as high as the plateau pressure, but not more than 35 cmH2O. Once the patient met the indication criteria for ECMO, a group discussion involving the ICU attending physician, the VV-ECMO team, and the patient’s family was conducted to decide whether to initiate VV-ECMO. Patients who were considered unsuitable for VV-ECMO support were excluded from the study. If the family members of VV-ECMO candidates accepted the risks associated with VV-ECMO, then VV-ECMO was initiated. If the family members of VV-ECMO candidates declined initiation of VV-ECMO support, treatment was continued using conventional ventilator support. Patients in both groups received a paralytic agent with deep sedation for 72 hours. During VV-ECMO support, the mechanical ventilator was adjusted to maintain a TV of approximately 4 mL/kg of ideal body weight with a low PEEP (5–10 cmH2O) and low FiO2 (≤40%). The VV-ECMO weaning process started when the underlying disease was successfully treated with improvement of lung function (plateau pressure of <25 cmH2O to maintain the TV at 6 mL/kg of ideal body weight).
This study protocol was approved by the Siriraj Institutional Review Board (approval no. Si 203/2018). The requirement to obtain written informed consent was waived because of the retrospective nature of the study.
Data collection
The patients’ electronic medical charts were reviewed, and the following information was collected and recorded: demographic data, baseline clinical characteristics, severity score, hemodynamic parameters, and respiratory parameters. The mechanical ventilator setting and blood gas analysis results were also recorded, both at the time the patient met the indication criteria for ECMO and then at 24-hour intervals for 4 days. The primary outcome was in-hospital mortality. The secondary outcomes were 28-day mortality, mechanical ventilator-dependent days, and vasopressor-dependent days. The complications associated with mechanical ventilator support and VV-ECMO support were recorded to assess treatment safety.
Statistical analysis
SPSS Statistics (SPSS, Inc., Chicago, IL, USA) was used to perform all data analyses. Continuous variables are expressed as mean ± standard deviation, and categorical variables are presented as number and percentage. An independent
Results
In total, 107 patients with severe ARDS were screened. Of these, 67 patients met the inclusion criteria for this study. However, three patients were excluded because a decision was made for them to receive palliative care (Figure 1). Therefore, 64 patients were finally enrolled. VV-ECMO was initiated in 30 patients, and 34 patients received conventional ARDS management. The patients’ baseline characteristics, including age, sex, disease severity, lung injury score, and causes of ARDS, were not significantly different between the two groups (Table 1). However, the VV-ECMO group contained a significantly higher proportion of patients with underlying hypertension (
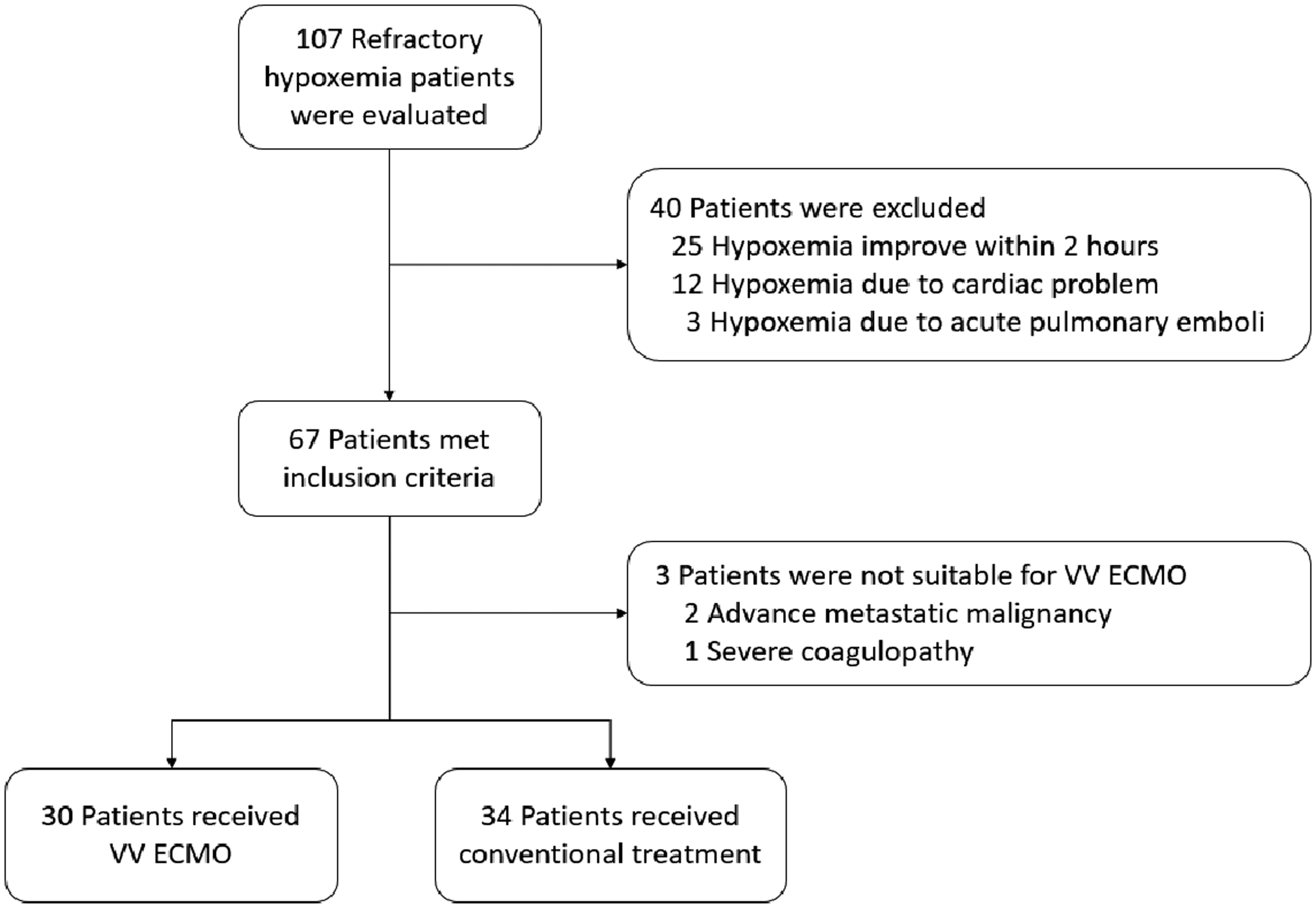
Flow diagram demonstrating the screening and enrollment of patients. VV-ECMO, venovenous extracorporeal membrane oxygenation.
Patients’ baseline characteristics and respiratory parameters on the day on which ECMO initiation criteria were satisfied.
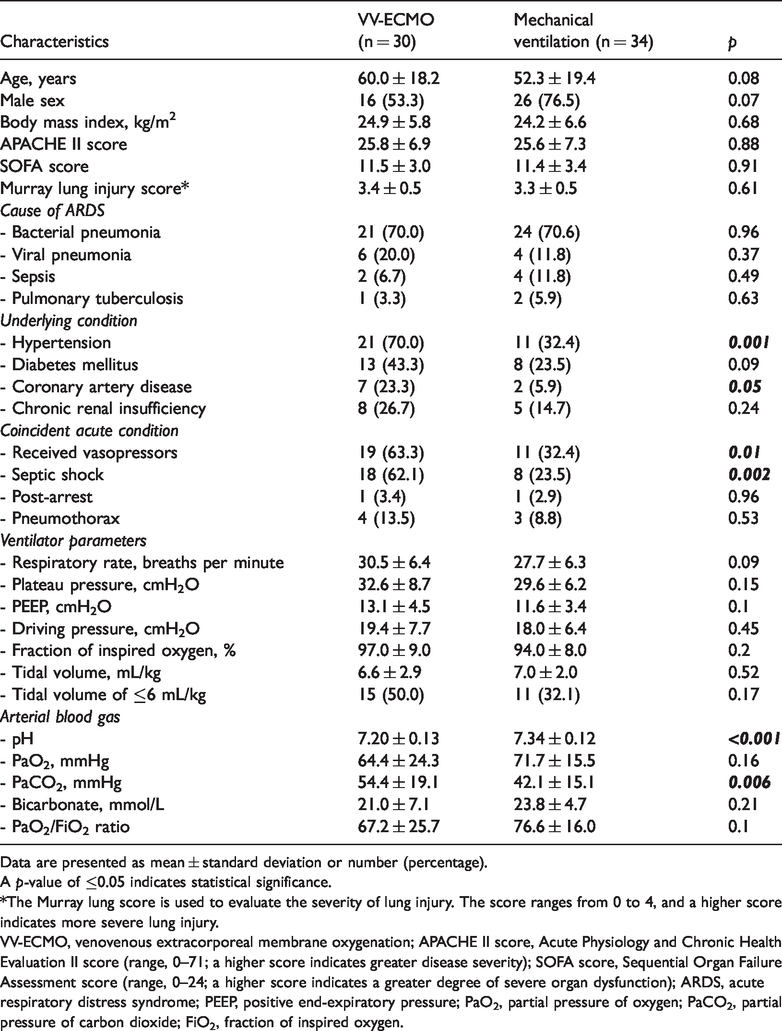
Data are presented as mean ± standard deviation or number (percentage).
A
*The Murray lung score is used to evaluate the severity of lung injury. The score ranges from 0 to 4, and a higher score indicates more severe lung injury.
VV-ECMO, venovenous extracorporeal membrane oxygenation; APACHE II score, Acute Physiology and Chronic Health Evaluation II score (range, 0–71; a higher score indicates greater disease severity); SOFA score, Sequential Organ Failure Assessment score (range, 0–24; a higher score indicates a greater degree of severe organ dysfunction); ARDS, acute respiratory distress syndrome; PEEP, positive end-expiratory pressure; PaO2, partial pressure of oxygen; PaCO2, partial pressure of carbon dioxide; FiO2, fraction of inspired oxygen.
With respect to the severity of ARDS and the degree of hypoxemia at baseline, there was no significant difference in the Murray lung injury score between the VV-ECMO and mechanical ventilation groups (3.4 ± 0.5
At 48 hours after patients met the ELSO criteria for VV-ECMO initiation, patients who underwent VV-ECMO cannula insertion were ventilated with a significantly lower plateau pressure (
Therapeutic management and patient outcomes.

Data are presented as number (percentage) or mean ± standard deviation.
A
*ARDS management that patients received after meeting the criteria for ECMO initiation.
**Evaluated at 48 hours after patients met the criteria for ECMO initiation.
VV-ECMO, venovenous extracorporeal membrane oxygenation; ARDS, acute respiratory distress syndrome; PaO2, partial pressure of oxygen; PaCO2, partial pressure of carbon dioxide.
Significantly more patients in the VV-ECMO group than mechanical ventilation group required vasopressor therapy (
Complications associated with VV-EMCO (n = 30).

A
*
VV-ECMO, venovenous extracorporeal membrane oxygenation.
Cox regression analysis of factors independently predictive of hospital mortality among patients with severe ARDS who developed refractory hypoxemia.

A
*The Murray lung score is used to evaluate the severity of lung injury. The score ranges from 0 to 4, and a higher score indicates more severe lung injury.
ARDS, acute respiratory distress syndrome; HR, hazard ratio; CI, confidence interval; PEEP, positive end-expiratory pressure; PaO2, partial pressure of oxygen; FiO2, fraction of inspired oxygen; PaCO2, partial pressure of carbon dioxide; VV-ECMO, venovenous extracorporeal membrane oxygenation.
Discussion
In this retrospective cohort study, VV-ECMO support among patients with severe ARDS who developed refractory hypoxemia was associated with a lower plateau pressure, lower driving pressure, lower PEEP, and lower FiO2 and promoted low-TV-strategy compliance. However, neither the in-hospital mortality rate nor the number of ventilator-dependent days was significantly different from those in patients who received conventional mechanical ventilator support. Additionally, the in-hospital mortality rate was significantly higher among patients with than without VV-ECMO-related complications.
VV-ECMO is reportedly a successful lifesaving treatment for refractory hypoxemia caused by severe ARDS. The ECMO to Rescue Lung Injury in Severe ARDS (EOLIA) trial was a multicenter randomized controlled trial that randomly assigned patients with severe ARDS to receive either ECMO support (ECMO group) or conventional ARDS treatment (control group).
13
Patients in the control group received standard ARDS treatment, which included a low-TV strategy, optimal PEEP, neuromuscular blockade, prone position, and recruitment maneuver. Thirty-five of 125 patients (28%) in that group developed refractory hypoxemia despite receiving comprehensive ARDS treatment. Those patients had evidence of cardiovascular failure (as determined by an increasing serum lactate level) that required a higher dose of inotropes, the presence of severe right ventricular failure, and cardiac arrest before rescue ECMO was initiated. Fifteen of those 35 (43%) patients survived to 60 days.
13
Improving oxygenation by VV-ECMO prevents cardiovascular collapse and cardiac arrest due to severe hypoxemia, and it allows adequate time for the patient’s lungs to recover from ARDS, especially if the cause is reversible. Data from cohort studies conducted during the 2009 influenza H1N1 worldwide pandemic era showed that patients with refractory hypoxemic ARDS were successfully treated by VV-ECMO support until their recovery from ARDS.
6
The results from a 2009 matched cohort study showed that adults with suspected or confirmed influenza H1N1 infection-associated respiratory failure who were referred to a VV-ECMO center had significantly lower hospital mortality than non-referred patients.
5
Moreover, most of the VV-ECMO-referred patients (86.3%) received VV-ECMO support. When a propensity score-matching model was performed, the hospital mortality rate was 23.7% for VV-ECMO-referred patients versus 52.5% for non-VV-ECMO-referred patients (
In addition to improving oxygenation, VV-ECMO also improves patient ventilation. This technique allows patients to be ventilated under a very low TV together with a low FiO2. The TV can be set to <4 mL/kg of ideal body weight, which dramatically decreases the peak inspiratory pressure. The present study showed that at 48 hours after patients met the criteria for VV-ECMO initiation, patients who received VV-ECMO were ventilated with a significantly lower plateau pressure, PEEP, driving pressure, FiO2, and TV than patients who did not receive ECMO support (Table 2). This finding corresponds with a recently reported multicenter study involving patients from 23 international ICUs; the study showed that the concept of lung rest has been accepted by physicians who care for patients with severe ARDS worldwide. 14 Evidence from another multicenter cohort study revealed that a lower driving pressure, which is the difference between the inspiratory pressure and PEEP, after VV-ECMO initiation was an independent predictor of the hospital survival outcome. 15 The combination of a lower driving pressure and lower TV prevents volutrauma and barotrauma, both of which are important mechanisms of ventilator-induced lung injury. Other preventable types of lung injury include atelectrauma, which can be prevented by using a higher PEEP. Although data from a meta-analysis did not confirm the benefit of a higher PEEP over a lower PEEP in patients with ARDS who underwent mechanical ventilation without VV-ECMO support, 16 evidence from a multicenter retrospective observational study suggested that a lower PEEP during VV-ECMO support was associated with a higher mortality rate. The mean PEEP of VV-ECMO survivors and non-survivors was 12.7 ± 2.9 and 11.0 ± 2.7 cmH2O, respectively. 16 Recovery from ARDS may be promoted and hastened via a reduction in the ongoing process of lung injury during mechanical ventilation. In our patients undergoing ECMO and similar to previous studies, we were able to optimize prevention of volutrauma and barotrauma by decreasing the TV and driving pressure to a low level. However, even with a relatively low PEEP (8.9 ± 2.6 cmH2O) after VV-ECMO support, atelectrauma could not be fully prevented.
Although VV-ECMO is considered a lifesaving procedure, it can be complicated by several adverse events. According to a meta-analysis of nine observational studies involving 545 patients undergoing VV-ECMO, bleeding problems were the leading complication with an incidence of 24.9%. 15 Intracranial hemorrhage was reported in 2.8% of patients. There was a significantly higher proportion of bleeding complications among non-survivors than among survivors in the univariate analysis; however, neither bleeding nor intracranial hemorrhage was identified as an independent predictor of death among patients undergoing VV-ECMO in the multivariate analysis. In the present study, 10 of 30 patients undergoing VV-ECMO developed complications associated with VV-ECMO. Intracranial hemorrhage was the leading complication (4 of 10 patients), followed by gastrointestinal bleeding that required blood transfusion (3 of 10 patients). Similar to the above-mentioned report, we found the hospital mortality rate to be significantly higher among patients undergoing ECMO with than without complications (Table 3).
In the present study, the in-hospital mortality rate of patients with ARDS who developed refractory hypoxemia and underwent VV-ECMO support at our center was higher than that in previously reported Western studies conducted at high-volume and experienced ECMO centers. This can be explained by the higher proportion of patients with septic shock in our population. To improve ECMO survival outcomes, the mechanical ventilator setting during ECMO support needs to be adjusted. The TV should be set at a low level of 4 to 6 mL/kg of ideal body weight. This lower tidal volume will reduce the plateau pressure and driving pressure. The PEEP should be set at a higher level to prevent atelectrauma. Esophageal pressure monitoring may be helpful to identify the optimal PEEP. Another strategy to potentially improve ECMO outcomes is to prevent complications associated with ECMO support. The most common complication is bleeding (including intracranial hemorrhage); therefore, anticoagulant management should be thoroughly controlled with close monitoring of the activated partial thromboplastin time, which should be 1.5 to 2.0 times the normal value. Prompt platelet transfusion and fibrinogen supplementation should be provided to maintain the platelet count at >100,000/µL and fibrinogen level at >150 mg/dL according to the ELSO 2014 recommendations. 18
Limitations
This study has certain limitations. First, because of the retrospective observational nature of this study, there was an inevitable bias in patient selection to receive VV-ECMO support or not. All physicians on the VV-ECMO team, attending physicians, and patient’s family members who were involved in the ECMO decision conference knew about the patient’s condition. The decision to initiate ECMO may have been based on the presence of more severe hypoxemia. Furthermore, patients whose hypoxemia seemed to be improved by conventional treatment or who had other health problems may have been considered for continuation of conventional treatment. Considering the patients’ hemodynamic status, patients in the ECMO group were likely to have a more severe condition at baseline because they received vasopressors at a significantly higher proportion than patients in the mechanical ventilation group. As such, the comparison outcome between ECMO versus conventional treatment should be interpreted in the context of this selection bias. Second, the number of patients enrolled in this study was small because the rarity of ARDS with refractory hypoxemia. Our small sample size may have limited the statistical power of our study to identify all significant differences and associations. Finally, this was a single-center study, which may limit the generalizability of our findings to other centers or healthcare settings.
Conclusion
This study has demonstrated that VV-ECMO support in patients with ARDS who develop refractory hypoxemia promotes “lung resting” by reducing the TV, peak inspiratory pressure, and driving pressure. This could limit the progression of ventilator-induced lung injury and allow adequate time for lung recovery from ARDS. However, the in-hospital mortality rate among patients undergoing VV-ECMO was not significantly different from that among patients receiving conventional ARDS treatment. Further improvement in the prevention of ECMO-associated complications and optimal mechanical ventilator setting adjustment may lead to more favorable ECMO outcomes.
Footnotes
Acknowledgement
The authors gratefully acknowledge Mr. Kevin P. Jones for editing a draft of this manuscript.
Authors’ contributions
All authors meet the ICMJE authorship criteria. Surat Tongyoo developed the study protocol, performed the data analysis, and drafted the manuscript. Siwalai Sucher developed the study protocol and performed the data collection and analysis. Chairat Permpikul, Preecha Thomrongpairoj, Akekarin Poompichet, Ranistha Ratanarat, and Nitipatana Chierakul contributed to the study design and data acquisition. All authors read and approved the final manuscript.
Declaration of conflicting interest
All authors declare no personal or professional conflicts of interest and no financial support from the companies that produce and/or distribute the drugs, devices, or materials described in this report.
Funding
This study was funded by a grant from Siriraj Critical Care Research Funding.
