Abstract
Infantile hepatic haemangioendothelioma (IHH) is the most common benign hepatic tumour in infants. However, experience of managing IHH is lacking and treatments for symptomatic IHH are controversial. Here we report the case of a patient with IHH treated by liver resection. A liver mass was found in a newborn by prenatal ultrasonography. The patient presented with abdominal distention with a tangible mass. Further imaging diagnosis and biopsy were carried out and complete surgical resection of the mass was performed. Histological examination confirmed IHH. The patient recovered uneventfully after surgery, with no additional therapy after discharge and no recurrence during follow-up. We also summarise previously published resected cases of IHH and review the surgical outcomes. Surgical resection and liver transplantation appear to be effective treatments for symptomatic IHH.
Keywords
Introduction
Infantile hepatic haemangioendothelioma (IHH) is the most common benign mesenchymal liver tumour in infants, accounting for 70% of benign tumours and 40% of all paediatric liver neoplasms.1,2 IHH usually occurs within 2 years of birth and is usually diagnosed before the age of 6 months. Symptomatic IHH commonly presents as abdominal masses, hepatomegaly, congestive heart failure (CHF) caused by shunts in the tumour, and coagulation dysfunction, such as Kasabach–Merritt syndrome, as well as other concomitant diseases.3–12 The main diagnostic tools for IHH are currently ultrasonography, computed tomography (CT), magnetic resonance imaging (MRI), and needle biopsy.13–15 Once a diagnosis is established, appropriate interventions can be planned. Asymptomatic IHH is generally monitored by periodic imaging, usually ultrasonography.2,16–18 However, a lack of experience means that treatments for symptomatic IHH are controversial,2,4,16–18 and drug treatments, interventions, and surgery may be chosen depending on the experience of the staff at the specific medical institution.2,16–18 We present a case of IHH treated successfully by liver resection. We also review previous cases reported in PubMed treated by surgery, including liver transplantation, with the aim of summarising the current clinical experience and surgical outcomes of IHH.
Case report
A 7-day-old newborn girl, who was diagnosed 7 days before birth with a liver tumour by antenatal Doppler colour ultrasonography, was admitted to Wuhan Children’s Hospital. The mother was gravida 1 para 1, and the baby was born at 43+3 weeks and weighed 3.1 kg at birth. Her parents noted abdominal distension without fever, vomiting, bloody excrement, or convulsions. There was no relevant family or genetic history. Her vital signs, including body temperature, pulse, respiration rate, and blood pressure, were normal. Physical examination revealed an abdominal bulge and a palpable mass in the right upper quadrant, around 80 mm × 80 mm, hard, fixed, with suspicious tenderness and uncertain boundaries (Figure 1). Routine blood examination and coagulation function were within normal ranges. Tumour markers, including alpha-fetoprotein (AFP, 13,908 IU/mL, normal range: 0–15,770 IU/mL), carbohydrate antigen 199 (24.4 U/mL, normal range: 0–27 U/mL), and carcinoembryonic antigen (2.1 ng/mL, normal range: 0–3.4 ng/mL) were all within normal limits, but carbohydrate antigen 125 was slightly elevated (42 U/mL, normal range: 0–35 U/mL). Liver tests were normal apart from decreased total protein (46.4 g/L, normal range: 63–82 g/L) and albumin (26.4 g/L, normal range: 35–50 g/L). There was no evidence of hepatitis B, A, or C virus infection. An abdominal CT scan revealed an obviously enlarged liver, and single mild round hypodense foci in the right hemi-liver, 61 mm × 72 mm × 79 mm in size, with well-defined margins and slightly uneven density inside, associated with bleeding, degeneration, and necrosis (Figure 2a). The plain CT value was 18 to 36 Hu. During the enhanced phase, the lesion supplied by the hepatic artery was obviously enhanced in the periphery, with a CT value of 120 Hu, and gradually intensified inward in the delayed phase (Figure 2b–2d). The right kidney was shifted downward as a result of tumour compression, with the portal vein to the left and forward and inferior vena cava to the left (Figure 2e–2g). The preoperative diagnosis was hepatic haemangioendothelioma. Chest and head CT scans found no signs of distant metastasis, but the results of preoperative needle biopsy were inconclusive (Figure 3).

Abdominal manifestation in the patient. The patient was laid on the operating table under anaesthesia. A herringbone incision was marked in the upper abdomen. A protuberance with abdominal distention was observed in the right upper quadrant and a mass (about 80 mm diameter) was palpable in the hepatic region.

Computed tomography (CT) scans of patient with infantile hepatic haemangioendothelioma. (a) Plain CT scan showed a large regular lesion with heterogeneous density occupying the right hemi-liver. (b) Enhancement appeared first at the periphery of the tumour in arterial phase on contrasted CT scan, (c) moved slowly into the centre of the mass in venous phase, and (d) faded to the same level as the liver parenchyma in delayed phase. (e) Coronal enhanced CT scan revealed the tumour pushed the inferior vena cava to the left and compressed the right kidney, causing it to shift downwards. (f) Median sagittal section of enhanced CT showed that the tumour was close to the transverse colon and compressed the right kidney downwards. (g) Three-dimensional reconstruction of blood vessels showed the tumour encased by the inferior vena cava, which was pushed to the left. (h) The portal vein was moved to the left and forward due to tumour compression in the venous phase.
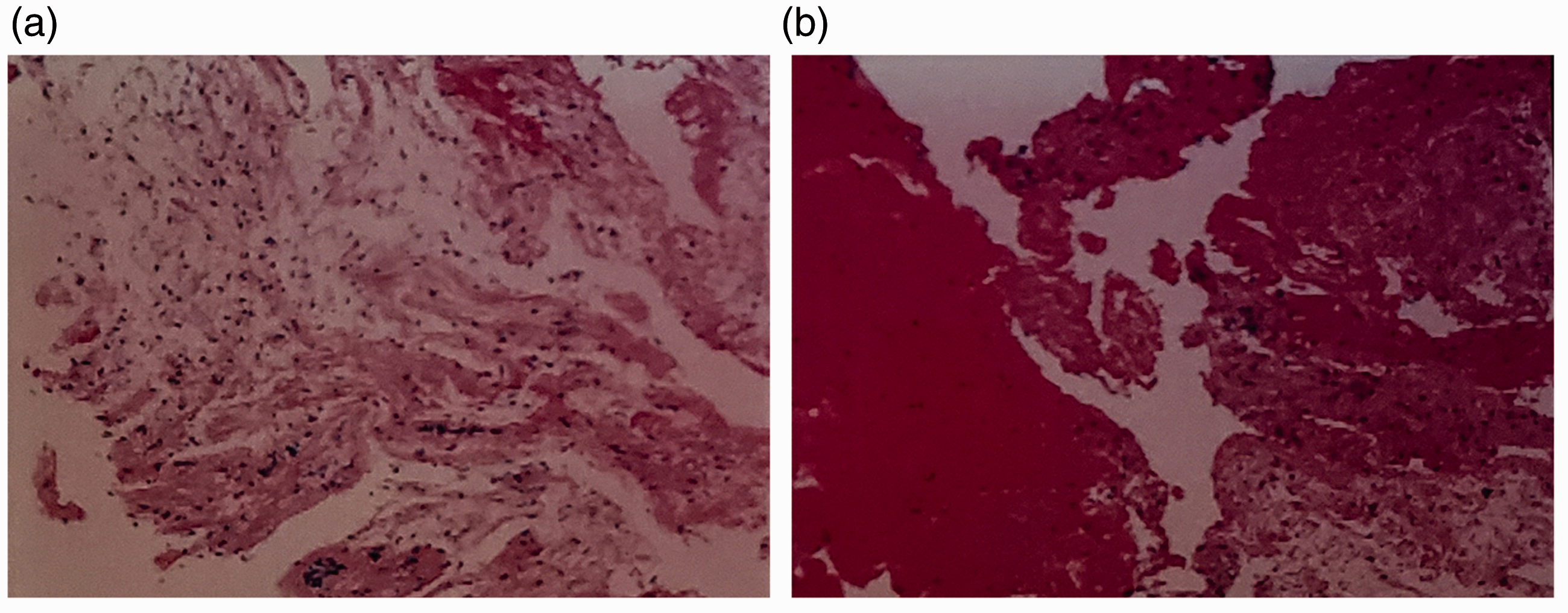
Pathology of tumour biopsy. The patient underwent preoperative ultrasound-guided needle biopsy of the liver tumour. Six whole biopsy tumour tissues were used to make serial slices. (a) The lesion contained few blood vessels, indicating degenerative and necrotic tissues. (b) Some parts of the tissue were filled with blood. Few inflammatory cells were observed. (Haematoxylin and eosin, ×100).
An exploratory laparotomy was performed 12 days after admission, after preoperative preparation including electrocardiogram, coloclyster, vitamin supplements, and diet regulation. A herringbone incision was made in the upper abdomen, and a firm, solid-cystic, grey-white mass, 80 mm × 70 mm × 60 mm in size, was found in hepatic segments 5, 6, and 7, very close to the porta hepatis and secondary porta. The tumour grew exogenously downwards and inwardly from the liver parenchyma, compressing the transverse colon, right kidney, portal vein, and inferior vena cava. No enlarged lymph nodes were discovered. The tumour was first isolated from the surrounding tissues with an elastic tape around the first porta hepatis to control the total portal inflow, and division of the hepatic ligamentum teres, falciform ligament, right coronary ligament, and right triangle ligament. The right portal vein, right hepatic artery, right hepatic duct, and right hepatic vein were then dissected, ligated, and divided. The right hemi-liver demarcation line appeared immediately after the right portal vein occlusion. The parenchymal demarcation line and liver resection line, 1 cm from the margin of the tumour, were marked using an electric scalpel. Under first hepatic portal control (Pringle’s manoeuvre), liver parenchymal transection was carried out using an ultrasonic scalpel, the vessels and intrahepatic ducts in the cut surface of the liver parenchyma were ligated and divided, and the tumour was then removed (intact) from the liver (Figure 4). The surface of the parenchymal transection was examined for bleeding and bile leakage, which were stopped using a 4-0 Prolene thread, which was simultaneously used to suture the stumps of the right portal vein and right hepatic vein. Finally, the abdominal cavity was cleaned with saline solution, a drainage tube was placed under the right diaphragm exiting via the right lower abdomen wall, and the incision was closed routinely. The total surgery time was 130 minutes, total blood loss was 20 mL, and the portal triad clamping time was 20 minutes. The patient was transferred to the neonatal intensive care unit after surgery and moved to the general ward 3 days later. The drainage tube was removed on the fifth postoperative day. The patient’s serum alanine aminotransferase (483 U/L), aspartate aminotransferase (196 U/L), and total bilirubin (20.7 µmol/L) levels were elevated on the first day after surgery, but returned to normal on the fifth day. Prophylactic antibiotics and liver-protective agents were administered for 5 days. The patient recovered from the operation without complications and was discharged 7 days after surgery.

Gross inspection of infantile hepatic haemangioendothelioma. (a) The tumour was removed successfully from the liver. (b) The tumour had an intact soft capsule, was reddish brown, and measured 80 mm × 65 mm × 60 mm. After cutting open, the tumour showed a necrotic and scarred centre filled with dark blood clots.
Postoperative histological examination of the tumour confirmed IHH (Figure 5), which verified the CT diagnosis. Immunohistochemical results revealed AFP(−), CD31(+), CD34(+), pan-cytokeratin(+), cytokeratin 7(+), Friend leukaemia integration-1 (+), glypican-3(−), human melanoma black-45(−), Hepatocyte(−), Ki-67(+, about 10%), Spalt-like transcription factor-4(−), vimentin(+), smooth muscle actin(+), and β-catenin(plasma +).

Histological examination of the tumour confirmed hepatic haemangioendothelioma. (a) Tumour tissue included variable-size blood vessels with swollen and hyperplastic vascular endothelium. A thickened basement membrane and hyaline changes of vessels were also observed. (b) The tissue was filled with blood. Tumour cells with sparse mitoses were arranged in a consistent manner. Inflammatory cells were uncommon. (Haematoxylin and eosin, ×100).
No additional therapy was given to the patient after discharge from hospital. She was monitored at intervals for the following 19 months (to date) with no sign of recurrence or metastasis.
Discussion
Although IHH is the most common benign hepatic mass in children, its incidence rate is only 5/100,000 and it is therefore rarely seen in clinical practice.4,9–12,18–20 Fewer than 20 cases have been reported in the literature, most of which were case reports.1,4,17,18,21 A lack of clinical experience of managing IHH thus means that its diagnosis and treatment vary among different hospitals.
In addition to clinical manifestations such as abdominal masses, hepatomegaly, and potential concomitant diseases including CHF, Kasabach–Merritt syndrome, and multiple skin or visceral haemangiomas, the diagnosis of IHH depends largely on imaging methods such as ultrasonography, CT, and MRI. Ultrasonography can be used to reveal the location, size, and number of tumours, and their relationship with the major blood vessels and bile duct, and has the advantages of presenting various sonographic views at relatively low cost, in real-time, in a non-invasive manner. 14 Ultrasonography is also widely used to detect and check abnormal masses during prenatal care and postnatal follow-up. IHH mainly displays as a hypoechoic or isoechoic regular mass with a clear boundary, and occasionally as a hyperechoic mass, accompanied by strong-echoic calcification in the centre and a hypoechoic periphery on ultrasound test.14,22,23 Most cases show enlarged arteries and veins, and shunts of artery to veins, or portal vein to veins in the tumour under colour and spectral Doppler.14,24,25 Furthermore, contrast-enhanced ultrasonography demonstrates peripheral enhancement with centripetal filling-in of the lesion.14,26 CT is the main and most sensitive method used to diagnose IHH. 14 IHH presents as a regular and hypodense mass on plain CT, or occasionally as a hyperdense mass due to haemorrhage or calcification. 13 Injection of a contrast agent leads to rim enhancement in the tumour in the arterial phase, gradually spreading to the centre of the tumour during the portal and venous phases, until the entire tumour is intensified in the delayed phase.13,14,27–32 MRI often shows low T1-weighted and high T2-weighted signals, and sometimes variable signals arising from changes in the lesion due to haemorrhage, necrosis, fibrosis, and calcification.13,14,25 The findings on enhanced MRI are identical to those of enhanced CT.13,14 In the present case, prenatal ultrasound imaging 7 days before birth revealed a liver mass, leading to follow-up intervention after birth. MRI was performed pre-admission, and CT scans after admission showed classical images of IHH, as described above, which further confirmed the diagnosis. A biopsy was also carried out in this case. Biopsy can be used to differentiate between IHH and liver malignancies, 15 and although a needle biopsy may cause massive bleeding and can sometimes fail to give definite results (as in the present case), liver biopsy is considered to be an indispensable means of identifying a suspicious diagnosis. The reported efficacy rate of liver biopsy ranges from 97.8% to 99.3%;33–35 however, the diagnostic rate of liver biopsy for IHH ranges from 50% to 100% because the small number of IHH cases means that a single negative result could have an apparently large effect on the overall efficacy of liver biopsy.4,17,18 The uncertain outcome of liver biopsy is mainly attributed to inadequate sampling or unsuitable specimens, as in our case, where the mass contained necrosis and bleeding. Complications of liver biopsy, including intra-abdominal bleeding, bile peritonitis, visceral penetration, pneumothorax, and even death, are rare.33–35 However, because IHH is a vascular tumour, we should be cautious of bleeding during biopsy, and coagulation function, liver function, complete blood count, chest radiography, and abdominal ultrasonography should be performed before liver biopsy and vital signs need to be monitored.
IHH needs to be differentiated from liver mesenchymal haematoma, hepatoblastoma, and hepatic cavernous haemangioma,2,22,33 and ultrasonography, CT, MRI, and biopsy can be used to facilitate these differential diagnoses.
IHH is a benign disease and some cases regress spontaneously. Treatments for IHH vary, including observation, medication, intervention, and surgery (Figure 6), and rely on the experience of different medical centre staff. Most medical centres treat patients with asymptomatic IHH by observation with periodic imaging checks,2,16–18 while symptomatic and unresectable patients receive various drug combinations including steroids, alpha-interferon, beta-blockers, diuretics, and digoxin, as well as chemotherapy, radiation, and interventions such as transcatheter arterial embolisation, transcatheter arterial chemotherapy and embolisation, and hepatic artery ligation.16,17,21,34 Unresectable IHH may transform to resectable IHH through conservative treatment. 34 If the disease continues to progress despite combined therapies, patients will be considered for liver transplantation.2,3,7,16,21,35 Hepatectomy is the first choice of treatment for symptomatic and resectable patients, 17 and surgery is also recommended in patients with a definite diagnosis and high AFP, even in the absence of symptoms, to eliminate liver malignancies.5,11 We summarised previous cases treated by resection, including liver transplantation, reported in PubMed (Table 1). Although IHH usually occurs in infants, two cases were reported in adults.12,36 There were more female than male HH patients, and most resectable masses were singular and occurred in association with various liver syndromes. Surgical outcomes were generally good, and 90% (45/50) of resected cases survived with no recurrence during follow-up. However, three patients died of bleeding during or after surgery, one exhibited disseminated intravascular coagulation and one suffered multiple organ failure. Regarding liver transplantation, including living donor liver transplantation (which is not recommended by some medical centres), 17 only 57.1% (8/14) of patients survived and remained disease-free because of extensive lesions with severe syndromes, recurrence, and the complexity of the surgery.

Algorithm demonstrating management of hepatic haemangioendothelioma (IHH). Asymptomatic and symptomatic IHH relived by medications, intervention, and hepatic artery ligation should be closely followed up. Complete resection is recommended in patients with symptomatic IHH. Liver transplantation is the last choice for patients with progressive IHH.
Previously reported cases of hepatic haemangioendothelioma treated with surgery.

F, female; M, male; CHF, congestive heart failure; PAH, pulmonary artery hypertension; SCN, severe congenital neutropenia; DIC, disseminated intravascular coagulation; CH, cutaneous haemangioma; K-MS, Kasabach–Merritt syndrome; RD, respiratory decompensation; B-CS, Budd–Chiari syndrome; HMH, hepatic mesenchymal hamartoma; LF, liver failure; GI, gastrointestinal; LD, largest diameter; LDLT, living donor liver transplantation; OLTX, orthotopic liver transplantation; MOF, multiple organ failure; IVH, intraventricular haemorrhage; PTLD, posttransplant lymphoproliferative disease; CR, complete resection; TAE, transcatheter arterial embolisation; TACE, transcatheter arterial chemotherapy and embolisation; IR, intensive resection.
The current patient exhibited mild symptoms including abdominal distention, hepatomegaly, and compression of adjacent vital organs, but no CHF or coagulation disorders, and therefore underwent hepatectomy with complete resection, without conservative treatment. She recovered well after surgery, with no sign of recurrence to date. However, surgical treatment remains controversial in symptomatic patients because the disease is benign, and some patients may recover completely with conservative treatment.2,17,18,37–40
IHH is typically classified histologically as type 1 or type 2: most cases are type 1 tumours, and type 2 tumours may develop into angiosarcoma, generally categorised as a borderline tumour with potent malignancy. 18 However, the above classification is controversial and is not described by guidelines, and the prognosis of IHH does not appear to be related to its morphological type.17,18
In summary, IHH is a benign tumour, but surgery, including liver transplantation, can be an effective backup treatment for patients with symptomatic IHH.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics
Ethical approval was obtained from the Ethics Committees of Wuhan Children’s Hospital and Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, China. Written informed consent was obtained from the patient’s guardians for publication of this case report and any accompanying images.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The National Natural Science Foundation of China [grant no. 81803175], Chen Xiao-ping Foundation for the Development of Science and Technology of Hubei Province, China [grant no. CXPJJH11900001-2019345], and the Foundation of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, China [grant no. 2017B001].
