Abstract
Primary hepatic neuroendocrine tumors (PHNETs) are a group of extremely rare tumors that are difficult to differentiate from common hepatic malignancies on routine imaging studies. By presenting a case of PHNET, we herein introduce our experience with the diagnosis, differential diagnosis, and management of patients with this rare disease. The patient was preoperatively diagnosed with hepatic hydatidosis but postoperatively diagnosed with a PHNET with multiple liver metastases. He was successfully treated with transcatheter arterial chemoembolization. This case indicates that the clinical diagnosis of PHNET is a medical challenge. Although peptide receptor radionuclide therapy has been suggested as the mainstay of treatment for well-differentiated somatostatin receptor-positive PHNETs, patients with a large tumor burden may also benefit from transcatheter arterial chemoembolization.
Keywords
Introduction
Neuroendocrine tumors (NETs), also known as carcinoid or argyrophilic tumors, are a very rare group of malignant tumors. In a recent report, the age-adjusted annual incidence of NETs was 1.09 per 100,000 persons in 1973, but the incidence increased to 6.98 per 100,000 persons by 2012.1,2 Liver metastases are common in neuroendocrine neoplasms (NENs), although the liver itself is rarely described as the site of the primary tumor.1,3 Until 2019, 150 cases of NENs were reported in the English literature1,5 since the first report of this disease by Edmondson 4 in 1958. NENs do not seem to have a sex predilection, and most patients have no clinical symptoms. As a result, misdiagnosis is common. We herein report a case of a PHNET that was preoperatively misdiagnosed as hepatic hydatidosis.
Case report
A 38-year-old man with slight intermittent upper right quadrant pain visited the Inner Mongolia Autonomous Region People’s Hospital (Hohhot, China) on 1 September 2016. Multiple liver tumors were identified on the initial abdominal ultrasound scan. An enhanced computed tomography (CT) scan of the upper abdomen revealed multiple well-circumscribed, heterogeneous, hypodense masses (largest dimension: 9.6 × 8.4 × 5.0 cm) with significant ring enhancement during the arterial and portal phases as well as multiple enlarged portal and retroperitoneal lymph nodes (Figure 1). Considering this patient’s history of raising dogs, an initial diagnosis of hepatic hydatidosis was made. This diagnosis was supported by a positive Casoni skin test performed at the Jiangsu Parasitic Research Institute 10 days later. The patient then revisited our hospital for treatment. The most common tumor markers were measured, including alpha-fetoprotein (4.05 ng/mL; reference, <7 ng/mL), carbohydrate antigen 19-9 (22.5 U/mL; reference, <34 U/mL), neuron-specific enolase (13.55 µg/L; reference, <17 µg/L), cancer antigen 72-4 (1.85 U/mL; reference, <6 U/mL), and carcinoembryonic antigen (negative). Fluorodeoxyglucose positron emission tomography–CT showed multiple low-density lesions with increased glucose metabolism in the liver (maximum standardized uptake value, 24.5). The largest tumor was a cystic thick-walled lesion of 9.6 × 8.4 × 5.0 cm in size located in the right liver lobe. No lesions with abnormally increased metabolism were identified in other organs (Figure 2). Based on the imaging characteristics, patient’s history, and positive Casoni skin test, a preoperative diagnosis of hepatic hydatidosis was made. The patient underwent a surgical exploration, during which multiple cystic solid tumors of different sizes were found in the liver. The largest lesion (10 × 10 cm) was found in segment VIII of the liver close to the first and second hilum extending to the caudate lobe (Figure 3(a)). Because forced removal of such a lesion may be fatal, only palliative resection (complete removal of the 2- × 2-cm liver mass in segment III) was performed. Histological and immunohistochemical examinations (Figures 4, 5) revealed the presence of the following tumor markers within the tumor cells: synaptophysin, chromogranin A, CK8, CK19, and CD56. The Ki67 index was 3% to 5% (Figures 3(b), 5), indicating nuclear reactivity. These findings suggested a malignant neoplasm that originated from well-differentiated NETs (G2). The patient was followed up for 10 months, and the possibility of metastatic cancers from extrahepatic primary sites was excluded. The patient was finally diagnosed with a PHNET with multiple liver metastases. After the operation, the patient was treated with transcatheter arterial chemoembolization (TACE) consisting of 100 mg of oxaliplatin (perfusion by catheter micropump at 99 mg/hour), a bottle of gelatin sponge particles (300–500 µm), and 10 mL of iodized oil. During the follow-up period (>12 months), the patient underwent three cycles of TACE. At the time of this report, he was disease-free and living a normal life.

Enhanced multidetector computed tomography. (a) Multiple well-circumscribed, heterogeneous, and hypodense liver masses were present. The largest mass (9.6 × 8.4 × 5.0 cm) was seen in the right lobe. All lesions demonstrated significant enhancement in the (b) arterial and (c) portal phases, with no enhancement of the irregular necrotic area within the largest lesion.
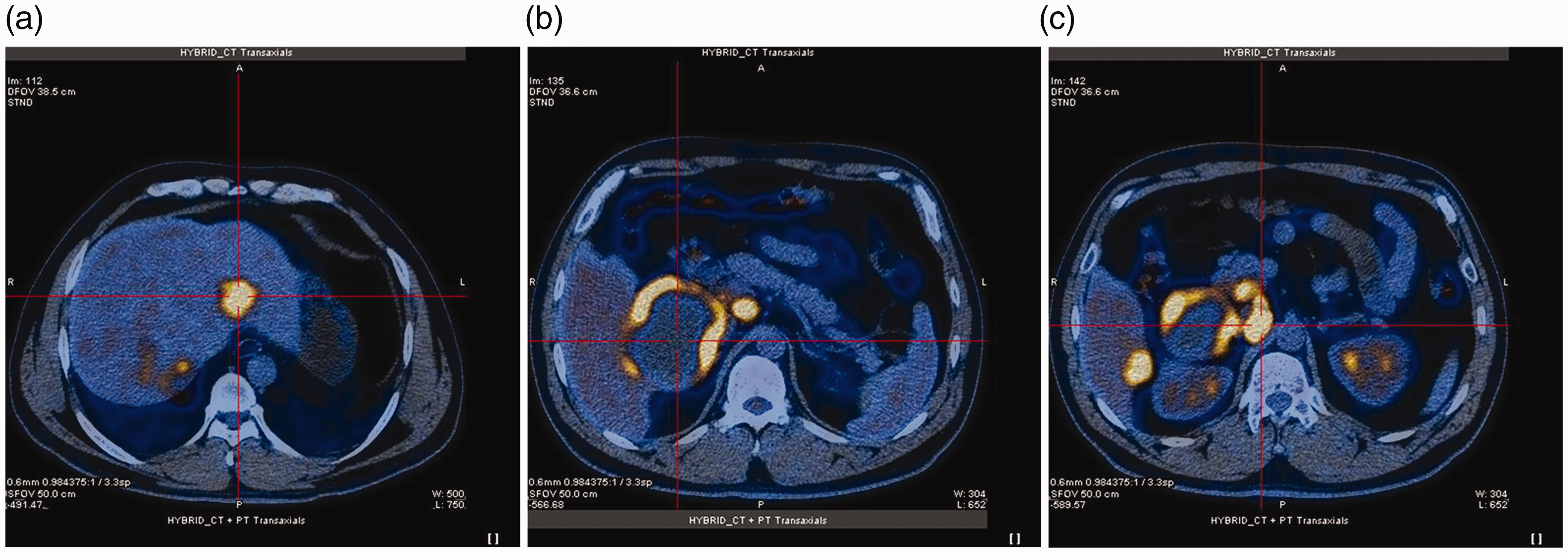
Positron emission tomography–computed tomography images of the liver. (a–c) Multiple low-density nodules with increased glucose metabolism. The largest nodule was seen in the right liver lobe (9.6 × 8.4 × 5.0 cm).

Intraoperative view of the tumor and expression of Ki-67 in tumor tissues. (a) A large soft tumor (10 × 10 cm) was seen in the right liver. The mass was connected to the caudate lobe. (b) Immunostaining for Ki-67.

Histologic examination of tumor tissues by hematoxylin and eosin staining.

Expression of the neuroendocrine tumor markers CK8, CK18, CK19, synaptophysin, chromogranin A, and CD56.
Discussion
NETs are a group of heterogeneous tumors derived from neurons with endocrine functions and neuroendocrine cells that are capable of producing functional peptide hormones. Perhaps the earliest research on NETs can be traced back to the work of Oberndorfer, who presented a discussion of gastroenteropancreatic NETs in the 19th century. 6
Based on the latest World Health Organization classification of gastrointestinal, hepatobiliary, and pancreatic NENs, NENs are divided into two differentiation categories: well-differentiated NETs and poorly differentiated neuroendocrine carcinomas. The former is further divided into three different grades according to the number of mitoses per 2 mm2 tumor area and the Ki-67 index: G1, G2, and G3. Based on this classification, the patient in our case was diagnosed with NET-G2.
Data from the Surveillance, Epidemiology, and End Results Program 9 show that the incidence of NET has been increasing during the past 30 years. This rise may be attributed to the increasing awareness of NET and the advances in diagnostic technologies. However, PHNETs are extremely rare, and their origin is unclear. Several sources of PHNETs have been proposed, including from neuroendocrine cells in the hepatic capillary bile, an ectopic pancreas, the adrenal glands, and neuroendocrine differentiation of malignant stem cells. 10 The pathogenesis and mechanisms of how PHNETs occur have not been identified. Understanding the molecular basis of PHNETs at the genome level will be very important for the development of effective therapeutic approaches.
In a recent whole-exome sequencing analysis involving 22 samples of PHNETs from 26 hospitals, 11 mutation of SETD1B was identified as a potential cause of and a diagnostic or prognostic molecular marker for PHNETs. Clinically, PHNETs are distinctive from NENs of other organs because the vast majority of PHNETs do not produce the clinical manifestations of increased neuroendocrine hormones. This is likely due to insufficient neuroendocrine hormone production or quality defects in the produced hormones. Hence, these hormones cannot play their biological roles in the target organs. 8 Histologically, the cancer cells of PHNETs are small, polygonal or cubic, and form a tubular, lamellar, or nest-like pattern; they also often show increased mitosis and prominent nuclear pleomorphism. Special neuroendocrine granules are present in the cytoplasm. When examined under the electron microscope, these granules are round or oval, enveloped with membranes, and different in size. Chromogranin A and synaptophysin are considered specific histological markers for the diagnosis of NENs. Clinically, 5-hydroxyindoleacetic acid in 24-hour urine specimens and the serum level of chromogranin A are commonly used diagnostic markers. 1 Unfortunately, because of the rarity of PHNETs, these diagnostic markers are rarely included in routine preoperative biochemistry panels.
Imaging studies are an important part of the diagnostic workup for hepatic cancers. PHNETs usually have a rich blood supply, but this lacks specificity in imaging studies. On CT scans, PHNETs mostly present as uneven low-density lumps with areas of necrosis and liquefaction. Dynamic enhancement scans may show uneven enhancement in the early stage, but this can gradually change to isodense and low-density lesions in the late stages. The enhancement mode of PHNETs is similar to that of hepatic hemangioma, making the differential diagnosis between these two types of cancer difficult. The diagnostic accuracy for PHNETs by other diagnostic approaches such as needle biopsy are reportedly suboptimal.8,12 The low diagnostic accuracy clearly contributes to the misdiagnosis of PHNETs as other common hepatic tumors such as hepatocellular carcinoma, intrahepatic cholangiocarcinoma, or hepatic hemangioma. 13 At present, postoperative analyses of the tumor tissues by histological and immunohistochemical evaluations are the essential methods for a definite diagnosis.3,5,14,15
No treatment guideline for PHNETs has yet been established, but surgical resection (wedge resection or lobectomy) is the treatment of choice and can provide a complete cure. The success rate for surgical excision of PHNETs reportedly exceeds 70% with a 5-year survival rate of 78%. 12 In patients with unresectable disease, various palliative options are available, including systemic chemotherapy using fluorouracil, hepatic artery embolization, octreotide therapy, and liver transplantation. Notably, however, peptide receptor radionuclide therapy (PRRT) is mainly used for well-differentiated and somatostatin receptor-positive NETs. PRRT is less effective for poorly differentiated neuroendocrine carcinomas, and PRRT is ineffective but may cause liver toxicity for multiple NETs with a large tumor burden. 16
In fact, TACE has been reported to be an effective approach for the treatment of patients with extrahepatic NETs with intrahepatic metastasis.12,14,17 For example, treatment of 20 patients with hepatic metastases by TACE led to a significant radiological response and symptom improvement in 90% of the patients. 15 In our current case, because the patient had multiple PHNETs in more than two lobes of the liver, surgical resection was not considered a safe option. Therefore, TACE was performed. After 12 months of follow-up, the patient’s condition was stable. This suggest that TACE with oxaliplatin is an effective treatment for patients with PHNETs.
Patients with PHNETs generally have a poor prognosis, but these patients may have a better prognosis than patients with other primary liver cancers. The factors affecting the survival rate of patients with PHNETs have been rarely reported. We believe that a variety of factors, such as the pathological type, tumor size, presence or absence of tumor metastasis, and type of treatment, may be among the key determinants of the patient’s prognosis.
Conclusion
Accurate diagnosis of PHNETs is a clinical challenge and requires preoperative analysis of the patient’s history, blood biochemistry, and imaging studies as well as postoperative analysis of tumor tissues, long-term follow-up, and exclusion of extrahepatic primary tumor sources. The preoperative misdiagnosis of liver hydatidosis is a lesson to surgeons when dealing with this type of hepatic tumor. Importantly, we demonstrated the effectiveness of TACE as an alternative treatment approach for patients with PHNETs.
Footnotes
Ethics and consent statements
This was a retrospective study of one patient. The study was conducted in accordance with the declaration of Helsinki, and all examinations described in this article were performed for medical purposes only. The patient’s information was kept strictly confidential during the study. The patient provided written informed consent. The study was approved by the Ethics Review Committee of Inner Mongolia Autonomous Region People’s Hospital.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
