Abstract
Objective
Periarticular defect coverage remains challenging because multi-vector tension forces affect wound tensile strength. This study presents our experience with keystone design perforator island flap (KDPIF) reconstruction of non-oncological periarticular defects surrounded by the zone of injury and describes the expanding versatility of KDPIF reconstruction for periarticular defects.
Methods
From June 2017 to July 2019, 12 patients aged 8 to 84 years underwent KDPIF reconstruction to cover periarticular defects. All defects resulted from non-oncological causes and were surrounded by the zone of injury. We reviewed the patients’ medical records and clinical photographs to collect and analyze clinical and operative data.
Results
The defect size ranged from 0.8 × 1.2 to 7 × 10 cm2. The flap size ranged from 1.5 × 3 to 15 × 18 cm2. All flaps survived completely. All patients showed favorable functional outcomes without significant limitation in joint range of motion during the follow-up period (range, 4–12 months). The mean observer scar assessment scale summary score and patient scar assessment scale total score were 17.667 ± 5.921 and 20.167 ± 6.478, respectively.
Conclusion
KDPIF reconstruction is a simple and effective surgical option for coverage of non-oncological periarticular defects surrounded by the zone of injury.
Introduction
The human body has many joints that allow for different degrees and types of movement. Periarticular wounds and defects are defined as those located around any joints. Periarticular wound closure should be performed with great care because multi-vector tension forces affect the wound tensile strength.1,2 Coverage solutions for periarticular defects should be able to withstand changes in tensile forces similar to those exerted on normal periarticular tissues. 2 These inherent characteristics of periarticular tissues can make coverage of even a small defect around the joint difficult. Many reconstructive options, including skin grafts, free flaps, and local flaps, are available for coverage of periarticular defects. Skin grafting is a simple and easy reconstructive modality. However, periarticular skin grafts are subjected to movement and shearing forces,2,3 which may result in the disruption of early fibrin bond formation and thus increased graft failure rates.1,2 Furthermore, contour deformities and secondary contractures may detrimentally affect the cosmetic and functional outcomes of periarticular skin grafts.2–4 Free flaps are appropriate options for covering moderate to large defects around the joint; however, their use is limited when skilled microsurgeons are unavailable, the treatment center is unable to perform postoperative microsurgical monitoring and care, and comorbidities that prohibit lengthy operations are present.3,5 Color and texture mismatch are additional disadvantages. 3 Local flaps are the most suitable option for covering small to moderate periarticular defects. Among various local flaps, the keystone design perforator island flap (KDPIF) devised by Behan 6 in 2003 has been applied to various fields of reconstructive surgery in the past decade because of its design simplicity, robust vascular supply, and easy reproducibility.3,7–9 The KDPIF, similar to the perforator-based island flap first described by Kim et al. 10 in 2010, does not require microsurgical perforator dissection, allowing for a safe and rapid operation.2,3,6,7 In numerous studies of KDPIF reconstruction, the causes of defects were usually oncological (after skin and soft tissue tumor removal), 3 and few reports have described periarticular defect coverage.2,4 In this retrospective study, we present our experience with KDPIF reconstruction for treatment of various periarticular defects surrounded by the zone of injury and demonstrate the expanding versatility of KDPIFs in the reconstruction of periarticular defects.
Materials and Methods
The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki, reflected in the approval by the institutional review board of Konyang University Hospital (approval number: KUH 2019-06-022). We obtained written informed consent from all patients.
This study involved patients who underwent KDPIF reconstruction to cover skin and soft tissue defects in periarticular areas from June 2017 to July 2019. We retrospectively reviewed data from the patients’ medical records, including the cause, location, and size of the defect; size and type of the KDPIF; total operative time; flap survival; complications; and follow-up duration. The range of joint movement was checked in all patients at the final follow-up. The scar-specific Patient and Observer Scar Assessment Scale11–13 was used to assess the final appearance of the scar. A single observer estimated the Observer Scar Assessment Scale (OSAS) score, and all patients self-assessed their final scars using the Patient Scar Assessment Scale (PSAS) score. After scoring the items, both the observer and patient rated the overall scar appearance to obtain an objective scar rating and overall patient satisfaction, respectively, based on a 10-point visual analog scale ranging from poor to excellent.
Surgical techniques
Preoperative wound preparation
The patients underwent serial debridement followed by negative-pressure wound therapy (NPWT) or conventional wound dressing for at least 1 to 2 weeks. At the time of debridement, the patients were asked to choose whether to undergo NPWT or conventional dressing. For inflammation of surrounding tissues, either empirical antibiotics or intravenous antibiotics were administered in accordance with the microorganism(s) identified by our Department of Infectious Disease. Through these treatments, we achieved sufficient wound preparation and stabilization by reducing edema, controlling infection, promoting perfusion around the wounds, and facilitating growth of healthy granulation tissue formation in the wound bed. 3 We then performed the final defect coverage using a KDPIF.
Flap surgery
The operations were performed under general anesthesia except in one patient (Patient 8), who underwent local anesthesia because she declined general anesthesia and her lesion was relatively small. After the final debridement, the final defect size was measured and adjacent hot spots of perforators around the defect were checked and marked using a hand-held ultrasound Doppler device. The flap was designed according to the defect size with consideration of tissue laxity and relaxed skin tension lines. The flap width was designed to be larger than the defect width. After the skin incision was made along the flap, the fibrous subcutaneous septa and deep fascia were released using a monopolar device until the flap could be moved freely from the surrounding tissues. An island-shaped flap was then created. The basic movement of the KDPIF was achieved by advancement via these procedures (releasing the fibrous subcutaneous septa and deep fascia). 7 Minimal undermining of the flap margin was performed to achieve further flap movement. 7 Through minimal undermining, the KDPIF acquired further flap movement with preservation of the vascularity of the central hot spot of perforators. If superficial veins and sensory nerves were identified during division of the deep fascia and minimal undermining of the flap, we attempted to isolate and preserve them to maintain the flap vascularity and sensation as long as they did not interfere with the flap movement. After achieving hemostasis, flap inset was first performed at the central portion of the flap on the defect side and then on both ends, which were aligned in a V-Y apposition. To further reduce tension during flap inset, an omega (Ω)-variant modification of the KDPIF was used, providing further flap movement via additional rotational movement. 14 After flap inset, the donor sites were closed without skin grafting in all cases, and drains were placed under the flap and in the donor site if needed. Either a mild compressive dressing for smaller flaps or incisional NPWT for larger flaps was applied.
Postoperative management
Postoperatively, complete rest of the joint was maintained with the assistance of an immobilization splint in cases involving the elbow, knee, and distal phalangeal joints and with an elastic bandage fixation in cases involving the glenohumeral joints. The splint remained in place until removal of the skin sutures after 14 days postoperatively. Steri-Strips (3M, Maplewood, MN, USA) were then applied for 2 weeks to prevent wound dehiscence, and passive joint mobilization was performed during this period. After 4 weeks postoperatively, active joint mobilization was performed in all patients. We also recommended that the patients put on pressure garments to encourage the healing process and facilitate movement for 3 months and that they use Mepiform (Mölnlycke Health Care, Gothenburg, Sweden), a self-adherent soft silicone sheeting designed for scar management, for a further 5 months.
Results
In total, 12 patients (5 male and 7 female patients) aged 8 to 84 years were included in this study. Table 1 shows the patients’ characteristics and clinical data. All defects resulted from non-oncological causes. The defect size ranged from 0.8 × 1.2 to 7 × 10 cm2, and the flap size ranged from 1.5 × 3 to 15 × 18 cm2. Eleven defects were covered using only KDPIFs, whereas one defect (Patient 2) was covered using a KDPIF with a Pacman flap. The KDPIF types used were type IIA (n = 4), Ω-variant type IIA (n = 2), type IIA with Sydney Melanoma Unit (SMU) modification 15 (n = 3), and Ω-variant type IIA with SMU modification (n = 3). All flaps fully survived without flap-related complications such as arterial insufficiency, venous congestion, or flap failure. Salvage of the main branches of the superficial veins and sensory nerve was achieved in all cases. One patient (Patient 6) developed marginal maceration, which healed with conservative treatment without further surgery. No other postoperative complications occurred, such as wound infection, hematoma, or seroma. After an average follow-up period of 6.92 months (range, 4–12 months), all patients were fairly satisfied with the final outcomes. At the final follow-up, all patients showed a favorable functional outcome without significant limitation in joint range of motion (ROM) compared with the preoperative state (Table 1). Moreover, no scar contractures were observed in any patients. Tables 2 and 3 show the results of the OSAS and PSAS assessments, respectively. The mean OSAS summary score was 17.667 ± 5.921, and the mean objective scar rating was 4.667 ± 1.303. The mean PSAS total score was 20.167 ± 6.478, and the mean overall patient satisfaction rating was 4.75 ± 1.545.
Patient data.
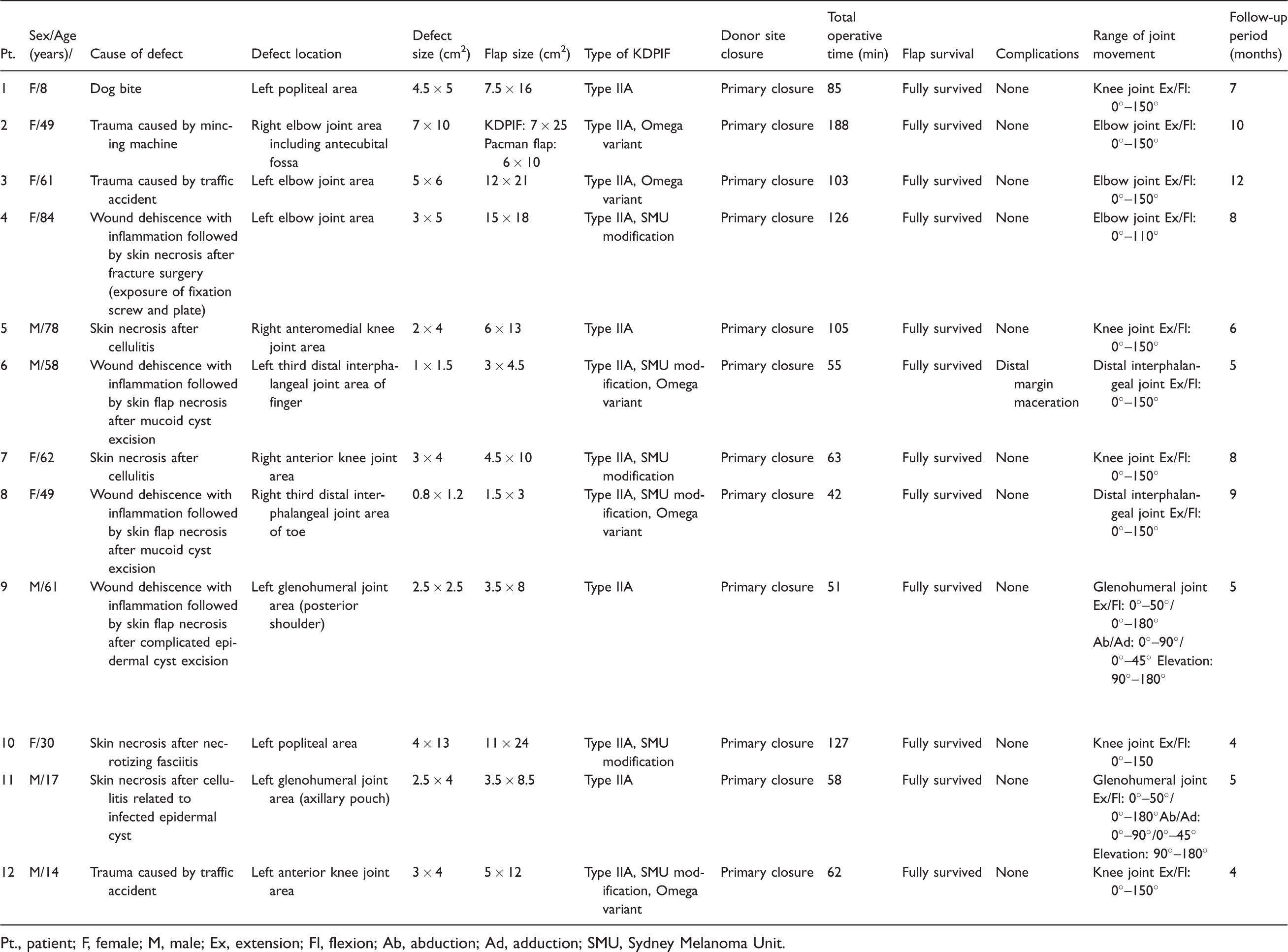
Pt., patient; F, female; M, male; Ex, extension; Fl, flexion; Ab, abduction; Ad, adduction; SMU, Sydney Melanoma Unit.
OSAS scores.

Data are presented as mean ± standard deviation.
OSAS, Observer Scar Assessment Scale.
PSAS scores.
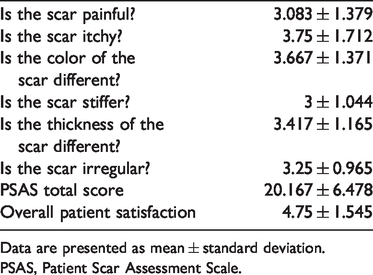
Data are presented as mean ± standard deviation.
PSAS, Patient Scar Assessment Scale.
Case presentations
Patient 1 (Figure 1)
An 8-year-old girl was admitted to our department for repair of a skin and soft tissue defect of the left popliteal fossa area caused by a dog bite. We performed serial debridement, NPWT, and intravenous antibiotic treatment for 2 weeks to achieve wound preparation and stabilization. The final post-debridement defect size was 4.5 × 5 cm2. We covered the defect with a 7.5- × 16-cm2 type IIA KDPIF from the lower posterior thigh. Flap inset and donor site closure were performed without tension or drain placement. Incisional NPWT was applied for wound dressing. The flap fully survived without flap-related complications or other postoperative complications. The patient showed a favorable functional outcome without contracture or joint ROM limitation after a 7-month follow-up.
Patient 2 (Figure 2)
A 49-year-old woman was admitted for repair of a skin and soft tissue defect of the right elbow joint area, including the antecubital fossa, caused by a mincing machine. We performed serial debridement, NPWT, and intravenous antibiotic treatment for 2 weeks to achieve wound preparation and stabilization. The final post-debridement defect size was 7 × 10 cm2. We covered the defect with an Ω-variant type IIA KDPIF (7 × 25 cm2) from the upper arm to cover half of the defect, followed by a Pacman flap (6 × 10 cm2) from the forearm to cover the other half of the defect. Flap inset and donor site closure were performed without tension or drain placement. Incisional NPWT was applied. Both flaps fully survived without flap-related complications or other postoperative complications. The patient was satisfied with the final outcome and showed a favorable functional outcome without contracture or joint ROM limitation after a 10-month follow-up.
Patient 6 (Figure 3)
A 58-year-old man was admitted for treatment of skin necrosis of the left third distal interphalangeal joint of the finger resulting from wound dehiscence with inflammation after mucoid cyst excision in another hospital. We performed serial debridement, conventional dressing, and intravenous antibiotic treatment for 1 week to achieve wound preparation and stabilization. The final post-debridement defect size was 1 × 1.5 cm2. We covered the defect with an Ω-variant type IIA KDPIF (3 × 4.5 cm2) with SMU modification from the radial side of the defect. Flap inset and donor site closure were performed without tension. Mild compressive dressing was applied using foam dressing material. The flap survived well, but marginal maceration occurred at the distal flap margin; however, the maceration completely healed with conservative treatment. No other postoperative complications occurred. The patient showed a favorable functional outcome without joint ROM limitation after a 5-month follow-up.
Patient 7 (Figure 4)
A 62-year-old woman was admitted for treatment of skin necrosis of the right anterior knee area resulting from cellulitis. We performed serial debridement, NPWT, and intravenous antibiotic treatment for 2 weeks to achieve wound preparation and stabilization. The final post-debridement defect size was 3 × 4 cm2. We covered the defect with a 4.5- × 10-cm2 type IIA KDPIF with SMU modification from the lower side of the defect. Flap inset and donor site closure were performed without tension or drain placement. Incisional NPWT was applied. The flap completely survived without flap-related complications or other postoperative complications. The patient showed a favorable functional outcome without contracture or joint ROM limitation after an 8-month follow-up.
Discussion
This study presents our experience of KDPIF reconstruction in 12 cases of non-oncological periarticular defects surrounded by the zone of injury. We obtained good outcomes and attribute the flap survival to our sufficient wound preparation and flap design, with consideration of the characteristics of the surrounding tissues. Covering periarticular defects is necessary to provide thin, pliable, and durable tissues through flap reconstruction to endure constant motion with multi-vector tension forces, shear, and external pressure.2,16,17 Pedicled perforator flaps (PPFs), as a good modality for achieving the ideal reconstructive goal (replacement of “like with like” tissues), have recently been used to cover various body defects. 3 Many previous studies have involved PPF reconstruction of periarticular defects, such as defects around the elbow joint, antecubital fossa, knee joint, and popliteal fossa.16–19 These studies showed that using PPFs is superior to using conventional locoregional flaps and provides an alternative to free flaps in the reconstruction of small- to moderate-sized periarticular defects.3,16–19 PPFs are easier and less risky than free flaps because PPFs do not require microsurgical anastomosis.3,10 However, microsurgical availability is critical in PPF reconstruction because perforator dissection is usually required and venous supercharging is often used.3,10 The KDPIF, which does not require microsurgical perforator dissection, can be used as a good alternative to the PPF in some circumstances, such as in hospital systems that do not have appropriate conditions for readily performing microsurgery, similar to our hospital. 3 In the present study, we used KDPIFs for periarticular defect coverage; therefore, microsurgical techniques were not required in any patients.
All flaps survived with minimal complications in the present study. KDPIF reconstruction has expanded from simple to complex defects throughout the body because of its evident advantages, including its simple defect-adaptive design, easy reproducibility, high level of safety, and short procedural time.2–4,6–9 Relative contraindications for KDPIF reconstruction include traumatic, irradiated, and inflammatory defects.3,9 These defects differ from oncological defects in that the surrounding tissues have a tendency to be under the zone of injury and are usually accompanied by wound infection.3,9 Thus, there is a high probability that the laxity of the flap is reduced, and wound healing complications occur at a high rate in the reconstruction of these defects.9,20 In 2017, our study group introduced KDPIF reconstruction of traumatic pretibial defects in patients with comorbidities. 3 We believe that sufficient wound preparation and stabilization before flap coverage should be among the crucial factors for successful KDPIF reconstruction, especially when the cause of the defect is non-oncological; this has not been mentioned in other similar studies of periarticular reconstruction with the KDPIF.2,4,21 In these studies, the defects mainly developed after wide excision of skin and soft tissue malignancies2,21 or, in one study, release of scar contracture. 4 In contrast, the defects in our cases resulted from non-oncological causes such as trauma, inflammation, and infection. To our knowledge, the present study may be the first case series of KDPIF reconstruction for non-oncological periarticular defects surrounded by the zone of injury.
In the present study, we carefully considered the surrounding tissue laxity and relaxed skin tension lines and designed the flap at the edge of the defect that exhibited greater tissue laxity. Periarticular defects should be covered with minimal wound tension because of the multi-vector tension forces in joint motion.1,2 Thus, the KDPIF is considered a good option for periarticular defects because it meets these reconstructive requirements. Given that the recruitment of tissue laxity is the principal biomechanical consideration in KDPIF reconstruction, a primary defect without surrounding tissue laxity is exchanged for a secondary defect with sufficient laxity in all margins to enable primary closure.8,20,22 V-Y advancement flaps at either end of the KDPIF facilitate the recruitment of laxity, and skin tension is redistributed perpendicular to the direction of maximal wound tension.3,6–8,20,22 Therefore, we believe that the KDPIF should be designed on the edge of the defect that has greater tissue laxity because this facilitates distribution of the tension required for closure throughout the periphery. Through this recruitment and redistribution of tissue laxity, the KDPIF can cover the periarticular defect with sufficient wound tension reduction, allowing the defect to endure changes in tensile forces by joint motion. Therefore, all flaps in our patients were well maintained without any wound problems, including wound dehiscence, breakage, or thinning, from re-starting joint motion (active joint movement after 4 weeks postoperatively) to the end of the follow-up period. Moreover, no patients showed significant joint ROM limitation compared with the preoperative state.
We believe that the type and modification of KDPIF to be applied should be determined intraoperatively, taking laxity, elasticity, and movement of surrounding tissues into account. The original classifications of KDPIF established by Behan 6 are as follows: type I (skin incision only) (Figure 5(a)), type II [(A, division of the deep fascia) (Figure 5(b)); (B, A with skin graft to the secondary defect) (Figure 5(c))], type III (double opposing keystone flaps) (Figure 5(d)), and type IV (keystone flap with undermining of approximately 50% of the flap subfascially) (Figure 5(e)). Additionally, the Ω-variant KDPIF and SMU modification are the two representative modifications.14,15 The Ω-variant KDPIF provides further flap movement via additional rotational movement and further reduction of tension without sacrificing healthy tissues in wound closure (Figure 5(f)).3,14 SMU modification involves maintenance of a skin bridge along the greater arc of the KDPIF, allowing additional vascularity, preserving the subdermal lymphatics to reduce the risk of pin cushioning, and reducing the time spent suturing the wounds (Figure 5(g)).9,15 We believe that the Ω-variation and SMU modification are especially valuable for covering non-oncologic defects, such as traumatic defects, which have reduced laxity of the surrounding tissues and a high rate of wound healing complications. These two modifications can provide the KDPIF with additional movement and vascularity, allowing traumatic defects to be effectively covered without complications. For cases requiring further flap movement and tension reduction, we used the Ω-variant KDPIF. For cases requiring further vascularity and stabilization, we used the SMU modification. In the present study, we used the Ω-variant KDPIF and SMU modifications in five and six cases, respectively. A previous study showed that using multiple perforator flaps can allow for large defect reconstruction and closure, achieving better donor site closure and tension-free reconstruction. 23 Thus, we consider that a type III KDPIF or a KDPIF combined with another local flap can be useful for extensive periarticular defects or defects that are difficult to cover with one flap. In Patient 2, we used an Ω-variant type IIA KDPIF to cover half of the defect and a Pacman flap to cover the other half of the defect; consequently, the patient showed a favorable functional outcome with no contracture or joint ROM limitation during the follow-up period.





Schematic illustration of Behan’s classification and representative modifications of the keystone design perforator island flap (KDPIF). (a) Type I KDPIF (skin incision only). (b) Type IIA KDPIF (division of the deep fascia along the outer curvilinear line). (c) Type IIB KDPIF (division of the deep fascia and skin graft to the secondary defect). (d) Type III KDPIF (opposing keystone flaps designed to create a double-keystone flap). (e) Type IV KDPIF (keystone flap with undermining of up to 50% of the flap subfascially). (f) The Ω-variant KDPIF (defect closure in fish-mouth fashion). (g) Sydney Melanoma Unit modification (maintenance of a skin bridge along the greater arc of the KDPIF).
Although we successfully covered all non-oncological periarticular defects surrounded by the zone of injury with KDPIFs, our study has some limitations. The present study was a retrospective clinical review with a non-randomized design, a relatively small sample size, and no comparison group, which unavoidably resulted in selection and confounding bias. However, our study is meaningful because it involved a larger number of patients (12 vs. 10 patients in a previous study) and involved a greater variety of periarticular lesions (including the interphalangeal joint area) compared with other similar published studies, definitively demonstrating the expanding versatility of KDPIFs in periarticular defect reconstruction. Future prospective large-scale studies with a comparison group, such as patients with skin grafts and other local flaps, are required to ensure outcome validity. Heterotopic ossification, which is the formation of ectopic mature lamellar bone within the soft tissues, should never be overlooked when fasciocutaneous flaps or muscle-based flaps are used to cover fracture sites associated with combat-related or other high-energy extremity trauma. 24 In particular, periarticular heterotopic ossification can lead to a markedly decreased ROM, loss of function, and increased morbidity and can adversely impact the patient’s rehabilitation, recovery, activities of daily living, and quality of life. 24 Fortunately, no periarticular heterotopic ossification developed in our patient who underwent KDPIF coverage over a fracture site (Patient 4).
We successfully reconstructed non-oncologic periarticular defects surrounded by the zone of injury using KDPIFs. We consider that the inherent characteristics of the KDPIF technique, such as recruitment of tissue laxity, redistribution of wound tension, and minimal flap undermining, can not only guarantee reliable reconstruction but can also achieve the ideal reconstructive goal (replacement of “like with like” tissue) in periarticular defects. KDPIF reconstruction may be a good alternative to conventional and perforator flaps for covering non-oncological periarticular defects surrounded by the zone of injury, with favorable outcomes when following meticulous wound preparation techniques.
