Abstract
Objective
The incidence of digestive tract malignancies (DTMs) is increasing, early diagnosis is limited, and treatment effects are unsatisfactory. DTMs express ghrelin, which might be involved in tumor formation and development; whether serum ghrelin can provide useful guidance remains unknown.
Methods
Sera of healthy individuals were obtained from October 2017 through March 2018; serum samples from patients with gastric (GC), colon (CC), and rectal (RC) cancers were collected during the same period. Serum ghrelin was tested by ELISA and correlated with clinicopathology of patients with DTMs.
Results
Serum ghrelin was higher in patients (GC, 38 patients; CC, 24; RC, 26) than in 69 healthy individuals and decreased significantly after tumor resection. Nutrition Risk Screening 2002 score and neutrophil:lymphocyte ratio affected perioperative serum ghrelin levels. The epithelial cell marker AE1/AE3 (pan keratin) in patients with GC, tumor location in the colon in patients with CC, and age in patients with RC also affected perioperative serum ghrelin.
Conclusions
Serum ghrelin might provide early warning of occurrence and guide prognosis of DTMs. Ghrelin can be used when screening for nutritional risk and inflammation. The clinicopathological influence on serum ghrelin in patients with DTMs is related to tumor location in the digestive tract.
Keywords
Introduction
In recent years, digestive tract malignancies (DTMs) have become a serious public health threat. Data from the National Cancer Center of China in 2019 showed that DTMs are increasing each year, with the incidence of gastric cancer (GC) being 10.79% and that of colorectal cancer (CRC) being 9.74%. 1 Although the 5-year survival rate for DTMs has improved, overall prognosis remains poor; the mortality for GC and CRC is 12.45% and 8.0%, respectively, and these are ranked third and fifth in cancer-related mortality, respectively. Early diagnosis, timely surgery, and effective detection of recurrence are the keys to improving prognosis. However, there are still no sensitive and effective indicators for clinical monitoring.
Ghrelin is a multifunctional brain–gut peptide, mainly produced by the fundus of the stomach, that regulates gastrointestinal motility and energy intake, among other functions. 2 Ghrelin and its receptors are expressed in DTMs,3–5 which suggests that ghrelin may be involved in tumorigenesis and tumor development. Ghrelin might therefore be helpful in the early diagnosis and treatment of DTMs, especially if serum ghrelin can provide early warning of the occurrence and recurrence of DTMs. There is no relevant research in this area to date.
In this study, perioperative serum ghrelin was measured in both healthy individuals and patients with DTMs; the influence of clinicopathology on serum ghrelin was analyzed as well. We preliminarily explored whether ghrelin could provide useful guidance for the early diagnosis and treatment of DTMs.
Patients & methods
Inclusion criteria
The study included healthy individuals with no abnormalities on physical examination and patients with DTMs confirmed by pathology who had not undergone surgical intervention at Peking Union Medical College Hospital (PUMCH, Beijing, China).
Exclusion criteria
Patients with malignancies other than DTMs and those with severe cardiac, pulmonary, or renal dysfunction, as well as patients with obesity (defined as body mass index >28 kg/m2) were excluded.
Ethical review
The study was reviewed by the Ethics Committee of PUMCH (approval number ZS-1581) and conducted in accordance with the Declaration of Helsinki.
Clinicopathology data and acquisition of specimens
For healthy individuals, we coordinated with the physical examination center of PUMCH. We digitally labeled and recorded the participants’ clinical data and collected 5 to 10 mL of serum from each individual. For patients with DTMs, we collected and recorded clinicopathologic data from those who underwent surgery in the third general surgery ward of PUMCH. Patients or their families signed the informed consent forms. Before surgery and 8 to 18 hours after surgery, 5 to 10 mL of serum was collected from each patient. All serum specimens were frozen at −80°C. The study period was from October 2017 through March 2018. The clinicopathological indexes recorded are shown in Table 1 and described here in brief.
Clinicopathological indexes.

AE1/AE3, anti-(pan) cytokeratin monoclonal antibodies; BMI, body mass index; CAP, College of American Pathology; CD, cluster of differentiation; CDX2: caudal-type homeobox transcription factor 2; CEA, carcinoembryonic antigen; CgA, chromogranin A; CK, cytokeratin; D2-40; lymphatic endothelial cell antibody; ERCC, excision repair cross-complementing; HER2, human epidermal growth factor receptor 2; IMP3, insulin-like growth factor 2 mRNA binding protein 3; Ki67, protein indicator of cell proliferation; MLH1, MLH1 mismatch repair protein; MSH2, MSH2 mismatch repair protein; MSH6, MSH6 mismatch repair protein; PMS2, PMS1 homolog mismatch repair protein; MUC, mucin; NLR, neutrophil–lymphocyte ratio; NRS2002, Nutritional Risk Screening 2002; OPNI, Onodera Prognostic Nutrition Index; p16, p16 tumor suppressor gene; Syn, synaptophysin.
Nutrition Risk Screening (NRS2002) is a recommended nutritional risk screening method for inpatients by European Society for Parenteral and Enteral Nutrition (ESPEN). A total NRS2002 score ≥3.0 indicates that the patient is at risk of malnutrition and needs nutritional support. A total NRS2002 score <3.0 indicates the patient can undergo major surgery; nutritional status will be reevaluated weekly. Body mass index (BMI) was also used as an index to measure the nutritional status of human body. The calculation method is weight divided by the square of height. Onodera’s prognostic nutritional index (OPNI) is a scoring system based on the nutritional and immune status of patients. In recent years, OPNI has been reported to be related to the prognosis of various digestive system tumors.
The neutrophil-to-lymphocyte ratio (NLR) is an index of systemic immunity and inflammation, which is related to the prognosis of many diseases such as lung cancer, esophageal cancer, and colorectal cancer. The TNM system was used to assess tumor stage and CAP (College of American Pathologists) grade was used to evaluate the degree of tumor regression after neoadjuvant treatment of gastrointestinal cancer. Several pathology-related indexes were assessed; MLH1, MSH2, MSH6, and PMS2 are mismatch repair genes, and ERCC and HER2 are genes that determine the sensitivity of tumor targeted therapy. Syn and CGA are neuroendocrine markers, and IMP3, CEA, β-catenin, Ki67, and p16 are tumor markers. AE1/AE3, Cdx-2, MUC1, MUC2, MUC5AC, CK7, CK20, calretinin, CD20, CD68, CD34, D2-40, and desmin are indicators related to tumor differentiation. AE1/AE3, also known as pan keratin, is a marker of epithelial cells. Because there are no epithelial cells in normal lymph nodes, AE1/AE3 is helpful to detect micrometastasis of lymph nodes.
Detection of serum ghrelin
After cryopreservation for 3 months, serum ghrelin was tested by ELISA using a rabbit polyclonal antibody kit (Immunoway Biotechnology Company, Beijing, China), Multiskan Spectrum microplate reader and HERAcell CO2 incubator (both from Thermo Fisher Scientific, Beijing, China). Each sample was tested three times and the average value was taken.
Correlation between serum ghrelin and clinicopathology
Serum ghrelin data of patients with DTMs were divided into three groups: preoperative, postoperative, and the group in which serum ghrelin content differed before and after surgery (decline range group). Serum ghrelin in each group was associated with clinicopathologic data to explore the associations of clinicopathology with perioperative serum ghrelin in patients with DTMs.
Statistical methods
SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA), was used for statistical analysis of all results. The normality distribution of the variables was tested using the one-sample Kolmogorov–Smirnov test. Paired sample t-tests, independent samples t-tests, or nonparametric tests were applied depending on whether the results conformed to normal distribution. P-values < 0.05 were considered statistically significant.
Results
Enrolled patients with DTMs and their characteristics
In addition to the serum samples from 69 healthy individuals, serum was collected from 88 patients with DTMs, including 38 patients with gastric cancer (GC), 24 patients with colon cancer (CC), and 26 patients with rectal cancer (RC). The last follow-up was in May 2018. The clinicopathological characteristics of the patients with DTMs are listed in Table 2.
Patient characteristics.
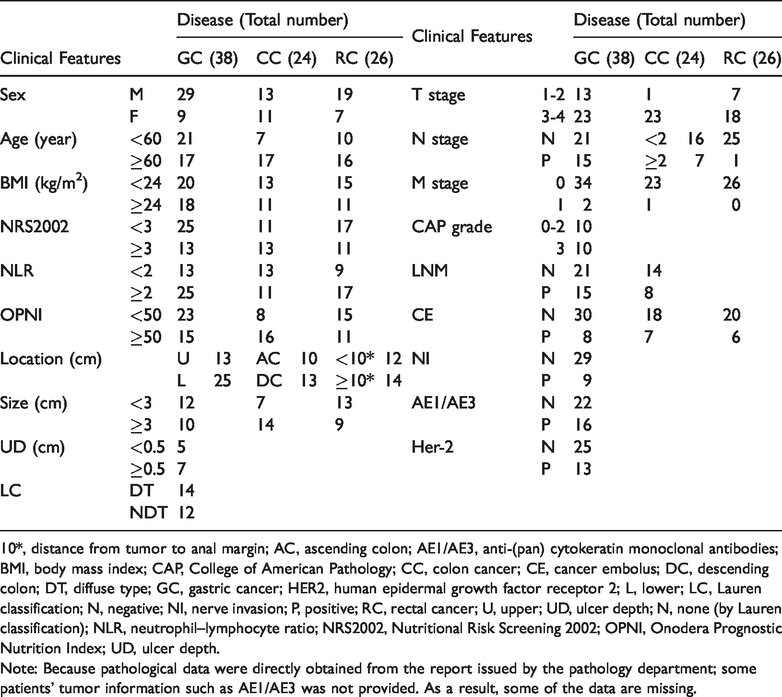
10*, distance from tumor to anal margin; AC, ascending colon; AE1/AE3, anti-(pan) cytokeratin monoclonal antibodies; BMI, body mass index; CAP, College of American Pathology; CC, colon cancer; CE, cancer embolus; DC, descending colon; DT, diffuse type; GC, gastric cancer; HER2, human epidermal growth factor receptor 2; L, lower; LC, Lauren classification; N, negative; NI, nerve invasion; P, positive; RC, rectal cancer; U, upper; UD, ulcer depth; N, none (by Lauren classification); NLR, neutrophil–lymphocyte ratio; NRS2002, Nutritional Risk Screening 2002; OPNI, Onodera Prognostic Nutrition Index; UD, ulcer depth.
Note: Because pathological data were directly obtained from the report issued by the pathology department; some patients’ tumor information such as AE1/AE3 was not provided. As a result, some of the data are missing.
Serum ghrelin levels
Table 3 shows that preoperative serum ghrelin levels in patients with DTMs were significantly higher than those of healthy individuals, being highest in patients with RC and lowest in those with GC (all P = 0.000); thus, serum ghrelin may be correlated with location of DTM. Postoperative serum ghrelin levels in patients with DTMs were significantly lower (P = 0.014) than preoperative levels. These differences were all statistically significant.
Serum ghrelin of healthy individuals and patients with DTMs in perioperative period.

CC, colon cancer; DTMs, digestive tract malignancies; GC, gastric cancer; M, mean; N, normal; Pre, preoperative; Post, postoperative; P1: comparison of serum ghrelin before and after surgery in each group; P2: comparison of the group with DTMs with the healthy group; RC, rectal cancer; SD, standard deviation.
Association of clinicopathology with perioperative serum ghrelin
Table 4 shows the influence of clinicopathologic findings on the serum ghrelin levels of patients with DTMs. For patients with DTMs, postoperative serum ghrelin was influenced by the NRS2002 score and NLR; the nutrition risk group (NRS2002 score ≥3) was significantly higher than that in the non-nutritional risk group (NRS2002 score <3) (P = 0.03). Serum ghrelin was significantly greater in the high-inflammation group than in the low-inflammation group (P = 0.023). For patients with GC, perioperative serum ghrelin was related to the NRS2002 score, cancer embolus, and AE1/AE3 positivity. Postoperative serum ghrelin in the nutrition risk group (NRS2002 score ≥3) was significantly higher than that in the non-nutritional risk group (NRS2002 score <3) (P = 0.034). The cancer embolus and AE1/AE3-positive groups had significantly lower serum ghrelin than the corresponding negative groups (P = 0.028 and P = 0.05, respectively). For patients with CC, tumor location affected the postoperative serum ghrelin level. Serum ghrelin was significantly higher in patients with carcinoma in the descending colon than in patients with carcinoma in the ascending colon (P = 0.05). In patients with RC, serum ghrelin was related to age and NRS2002 score: the preoperative ghrelin level was significantly higher in those >60 years than in those <60 years (P = 0.043). Postoperative serum ghrelin in the nutrition risk group (NRS2002 score ≥3) was significantly higher than that in the non-nutritional risk group (NRS2002 score <3) (P = 0.047).
Influence of clinicopathologic findings on serum ghrelin in patients with DTMs.

AE1/AE3, anti-(pan) cytokeratin monoclonal antibodies; BMI, body mass index; CAP, College of American Pathology; CC, colon cancer; CE, cancer embolus; DTMs, digestive tract malignancies; GC, gastric cancer; HER2, human epidermal growth factor receptor 2; LC, Lauren classification; NI, nerve invasion; NLR, neutrophil–lymphocyte ratio; NRS2002, Nutritional Risk Screening 2002; OPNI, Onodera Prognostic Nutrition Index; Pre, preoperative; Post, postoperative; RC, rectal cancer; UD, ulcer depth. Note: See Tables 1S, 2S, 3S, and 4S in the appendix for detailed data.
Discussion
DTMs are the most common epithelial-derived tumors and their incidence remains high. In the current situation, the rate of early diagnosis is low, overall treatment is not effective, and prognosis remains poor. 1 The number of new cases of GC and CRC in China in 2015 was 430,000 (29.31/100,000) and 388,000 (28.2/100,000), respectively; there were 291,000 (21.16/100,000) and 187,000 (13.61/100,000) deaths, respectively. The 5-year survival rates for early and advanced GC were 95% and 40%, respectively. The detection rate of early GC is close to 80% in Japan but only 5% to 10% in China. 6 Although there are no exact data on the early detection rate of CRC in China, it is much lower in South Korea (21.2%) and Japan (19.2%). The 5-year survival rate of patients with CRC in China is 54.6%, which is still lower than that of developed countries such as Europe, the United States, and Japan. 7 , 8 We are still lacking simple, convenient, and effective clinical indicators that permit early detection, warning of recurrence, as well as guidance for treatment.
Ghrelin, a brain–gut peptide containing 28 amino acids, is the first endogenous ligand of the growth hormone secretagogue receptor. It was first reported in rat stomach by Kojima in 1999. 9 Ghrelin is secreted mainly by the fundus of the stomach, and its receptors are ubiquitous throughout the body.10–15 In addition to promoting growth hormone secretion, ghrelin is involved in feeding, energy metabolism, cardiovascular regulation, and immune regulation.16–18 Research on ghrelin and cancer have gradually accelerated; ghrelin may be involved in the biological processes of tumor proliferation, invasion, and metastasis through the PI3K/AKT/mTOR, Ras/Raf/ERK, and other pathways,19–22 which might highlight the important research value and clinical significance of ghrelin in cancer screening, treatment, and prognosis. Ghrelin may be a therapeutic target or a new prognostic indicator for DTMs.
In this study, we found significant differences in preoperative serum ghrelin levels of patients with DTMs. Patients with RC had the highest serum ghrelin levels and those with GC had the lowest. However, Huang et al. 23 detected serum ghrelin in 58 patients with GC and 20 patients with CRC; serum ghrelin was not correlated with tumor location in that study. In addition, ghrelin secretion gradually decreased from the stomach to the colon and rectum under physiological conditions. 24 The results of our study are in contrast to those of previous studies, which might be related to the higher secretion of ghrelin in CRC tissues than in GC tissues. Verification of the expression of ghrelin in GC and CRC is necessary.
Preoperative ghrelin levels in the serum of patients with GC, CC, and RC were significantly higher than those in healthy individuals, and the levels decreased dramatically after surgery, indicating a degree of correlation between serum ghrelin and tumor tissue in patients with DTMs. Tumor tissue may secrete ghrelin or promote ghrelin secretion. The results of the study by An et al. 3 also showed significant differences: preoperative serum ghrelin was 10 times higher in patients with GC than in healthy individuals in that study. However, some studies have shown no significant difference between these groups. The study by An et al. showed that ghrelin was lower in tumors than in adjacent normal tissue, and ghrelin levels were negatively correlated with the degree of differentiation. Therefore, it is possible that there is no difference in serum ghrelin levels between cancer patients and the healthy population. In our study, we found a correlation between serum ghrelin and DTMs, indicating that serum ghrelin might have value as an early-warning marker of cancer occurrence and as a means to gauge prognosis. Some studies have shown that serum ghrelin levels in patients with other malignancies (such as lung cancer) 25 as well as inflammatory diseases (such as gastric ulcer 26 and pancreatitis 27 ) were higher than those in the healthy population. Serum ghrelin has many influential factors; it has multiple sources through the whole body and a wide range of functions. Therefore, the predictive effect of ghrelin on the occurrence and recurrence of DTMs is neither absolute nor specific and can only be used as a reference index.
Ghrelin plays an important role in regulating gastrointestinal motility, appetite, and energy intake. Oral administration of active ghrelin receptor agonists can increase gastrointestinal motility. 28 Ghrelin can improve anorexia–cachexia status and prolong the survival time of patients with malignancy. 29 , 30 Because of such issues as physiological or psychological stress, trauma, poor appetite, and restriction of energy intake, nutrition-related complications commonly occur after surgery. We found that postoperative serum ghrelin was positively correlated with NRS2002 score in patients with DTMs. Tan et al. 31 also found that serum ghrelin was positively correlated with the NRS2002 score and negatively with nutritional indicators in hemodialysis patients, which was consistent with the literature. 32 Serum ghrelin was low in obese and overenergetic patients, but high in patients with anorexia and malnutrition. 33 , 34 Increased serum ghrelin in the nutritional risk group might be a compensatory response to insufficient energy intake and malnutrition. Therefore, ghrelin could be a useful reference index in screening for nutritional risk, correcting malnutrition, and improving nutritional status clinically.
Research confirms that another important function of ghrelin is to inhibit inflammation; inflammation is generally accompanied by increased ghrelin synthesis and secretion. In an inflammatory state, various cytokines increase ghrelin secretion; meanwhile, ghrelin decreases proinflammatory factors, increases anti-inflammatory factors, and alleviates inflammatory damage to the body. In this study, we found that serum ghrelin in the high-inflammation group (NLR >2) was significantly higher than in the low-inflammation group (P = 0.03). In other words, serum ghrelin in patients with DTMs was positively correlated with inflammation. This finding is consistent with the literature. The NLR is an indicator of systemic inflammation. 34 , 35 The increase in lymphocytes and decrease in neutrophils coincides with improvement in systemic inflammation. 36 , 37 Systemic inflammation is usually accompanied by increased tissue consumption, increased catabolism, and reduced appetite. Therefore, ghrelin not only inhibits inflammation but can also increase appetite and energy intake, as well as improve the nutritional status of patients. Tsubouchi et al. 38 found that ghrelin not only alleviated cachexia in a lung cancer rat model but also regulated systemic inflammation. These studies provide useful guidance for the application of ghrelin in treating malignancy.
As Murphy et al. 39 noted in 2017, serum ghrelin is associated with the risk of CRC, and ghrelin levels may change throughout carcinogenesis. Two years later, Sundkvist et al. 40 validated Murphy’s research. They used fasting plasma samples from 60 patients with CRC in the Västerbotten Intervention Programme and matched them with the control group 5 years before diagnosis of CRC. The results showed that serum ghrelin remained stable for 5 years without any fluctuation and was not associated with the development of CRC; the authors concluded that serum ghrelin may not be a useful marker to predict the risk of CRC. This study also showed that there was no correlation between perioperative serum ghrelin in patients with DTMs and clinicopathological data such as sex, BMI, OPNI, lymph node metastasis, tumor size, ulcer depth, CAP grade, Lauren classification, cancer embolus, nerve invasion, or HER2 positivity (P > 0.05). An’s group noted similar results. 3 Whether ghrelin can be used as a marker for the early diagnosis of DTMs needs to be verified in studies with larger sample sizes.
The small sample size, the lack of data on perioperative changes in ghrelin in patients with benign digestive tract diseases as a comparison, and the short follow-up time are limitations of this study. In a follow-up study, we should rectify these shortcomings and further verify the role of ghrelin in the diagnosis and treatment of DTMs.
Conclusions
As a versatile brain–gut peptide, ghrelin and its relationship with tumors is under increasing scrutiny. However, this line of research is still in its infancy; the results of similar research studies are not always consistent and are sometimes in opposition. In this study, ghrelin may have a role in the diagnosis and treatment of DTMs. Given the diversity of ghrelin’s physiological functions, and that serum ghrelin is susceptible to other factors, assessing ghrelin in DTM tissues in vivo and DTM cells in vitro, rather than in serum (to exclude interfering factors), might yield more convincing evidence of ghrelin as a marker for disease.
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520920441 - Supplemental material for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies
Supplemental material, sj-pdf-1-imr-10.1177_0300060520920441 for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies by Changzhen Zhu, Yuqin Liu, Weiming Kang, Zimu Zhang, Ziyang Zeng and Dong Liu in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_0300060520920441 - Supplemental material for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies
Supplemental material, sj-pdf-2-imr-10.1177_0300060520920441 for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies by Changzhen Zhu, Yuqin Liu, Weiming Kang, Zimu Zhang, Ziyang Zeng and Dong Liu in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_0300060520920441 - Supplemental material for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies
Supplemental material, sj-pdf-3-imr-10.1177_0300060520920441 for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies by Changzhen Zhu, Yuqin Liu, Weiming Kang, Zimu Zhang, Ziyang Zeng and Dong Liu in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_0300060520920441 - Supplemental material for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies
Supplemental material, sj-pdf-4-imr-10.1177_0300060520920441 for Exploration of the role of serum ghrelin in the diagnosis and treatment of digestive tract malignancies by Changzhen Zhu, Yuqin Liu, Weiming Kang, Zimu Zhang, Ziyang Zeng and Dong Liu in Journal of International Medical Research
Footnotes
Acknowledgement
Authors’ contributions
Weiming Kang and Yuqin Liu designed the study. Changzhen Zhu, assisted by Zimu Zhang, Ziyang Zeng, and Dong Liu, collected serum and clinicopathological data, tested the serum ghrelin perioperatively, and analyzed the influence of clinicopathology on perioperative serum ghrelin in patients with digestive tract malignancies. Changzhen Zhu was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors acknowledge funding from Wu Jieping Medical Fund (2019.06-2021.06; No. 320.6750.19020, Correlation between ghrelin and clinicopathological features and prognosis of gastrointestinal stromal tumors) and from the 2019 Chinese Society of Clinical Oncology (CSCO)-Roche Solid Cancer Research Fund (No. Y-HS2019-043, Molecular mechanisms of miR-30b/KRAS/GSTπ pathway-mediated chemotherapeutic resistance in gastric cancer).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
