Abstract
Chicken ovalbumin upstream promoter-transcription factor II (COUP-TFII) is an orphan receptor that regulates the expression of genes involved in development and homeostasis. COUP-TFII is also dysregulated in cancer, where it plays important roles in oncogenesis and malignant progression. Recent studies have also investigated altered microRNA-mediated regulation of COUP-TFII in cancer. Although many investigators have studied the expression and clinical significance of COUP-TFII in several cancer types, there remain many controversies regarding its role in these diseases. In this review, we will describe the functions and underlying molecular mechanisms of COUP-TFII in several cancers, especially colorectal, gastric, breast, and prostate cancer; additionally, we will briefly summarize what is known about microRNA-mediated regulation of COUP-TFII.
Keywords
Introduction
Chicken ovalbumin upstream promoter transcription factors (COUP-TFs) belong to the nuclear hormone receptor superfamily. First cloned in 1986,1–3 COUP-TFs are now classified as orphan nuclear receptors because their ligands have not been identified. There are two COUP-TF homologues, COUP-TFI (EAR3 or NR2F1)4,5 and COUP-TFII (ARP-1 or NR2F2).6,7 COUP-TFs have an N-terminal DNA binding domain (DBD) and a C-terminal ligand-binding domain (LBD).
8
COUP-TFI and COUP-TFII proteins share 95% sequence identities, and their DBD and LBD amino acid sequences are highly conserved.
9
COUP-TFs can form homodimers or heterodimers with retinoid X receptor (RXR) and other nuclear receptors to bind to response elements containing imperfect
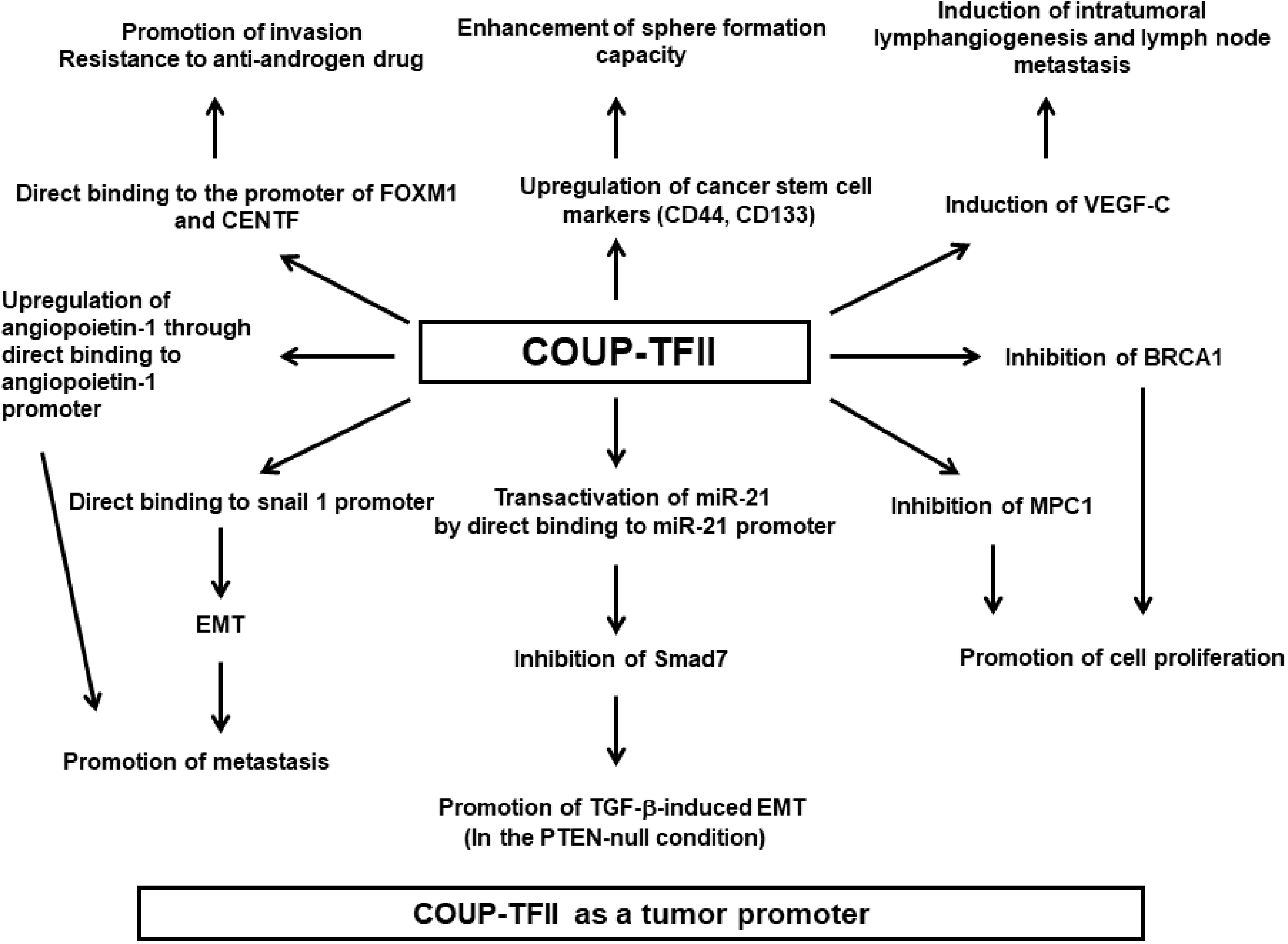
Mechanisms of the tumor promoting activities of COUP-TFII in cancer. EMT: epithelial-to-mesenchymal transition; VEGF-C: vascular endothelial growth factor-C; MPC-1: mitochondrial pyruvate carrier 1; TGF-β: transforming growth factor-β.
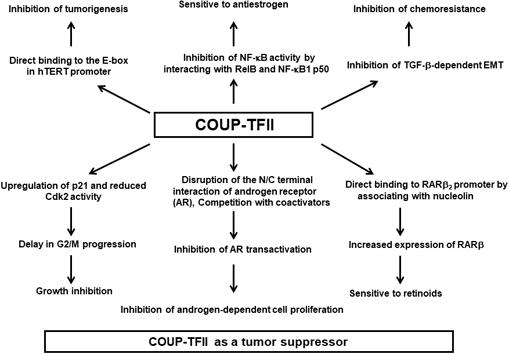
Mechanisms of the tumor suppressive activities of COUP-TFII in cancer. EMT: epithelial-to-mesenchymal transition; CDK2: cyclin dependent kinase 2; hTERT: human telomerase reverse transcriptase; RARβ: retinoic acid receptor β.

MicroRNAs regulate COUP-TFII expression through direct and indirect activities. Several microRNAs downregulate COUP-TFII expression based on published literature.26,88–94 For example, miR-27b expression is decreased in gastric cancer, and its re-expression inhibits the proliferation, migration, and invasion of gastric cancer cells in vitro and metastasis to the liver in vivo by downregulating COUP-TFII. 88 MiR-382 expression is decreased in colorectal and prostate cancer, and studies have shown that miR-382 inhibits the proliferation, migration, and invasion of colorectal and prostate cancer.89,90 MiR-101 and miR-27a have decreased expression in prostate cancer, where they function to inhibit metastasis by downregulating COUP-TFII. 26 MiR-302 regulates stem cell differentiation by directly targeting COUP-TFII;91,92 for example, miR-302a promotes osteoblastic differentiation by inhibiting COUP-TFII. 93 Finally, miR-194 promotes osteogenesis and inhibits adipogenesis by regulating COUP-TFII. 94
Tumor promoting effect and underlying molecular mechanisms of COUP-TFII
Tumor promoting effect of COUP-TFII in colorectal cancer (CRC)
It has been reported that COUP-TFII increases the expression of angiopoietin 1, hepatocyte nuclear factor 1 homeobox A (HNF1A), VEGFR2 (kinase insert domain receptor), and hepatocyte nuclear factor 4α (HNF4A). 8 Bao et al. 14 demonstrated that COUP-TFII regulates metastasis in colorectal adenocarcinoma cells by directly binding to the SNAI1 (Snail 1) promoter. They also showed that COUP-TFII may be a poor prognostic factor in CRC patients. 14 Snail 1 is a member of the Snail family of transcription factors, which are involved in epithelial-to-mesenchymal transition (EMT). EMT promotes tumor metastasis by enhancing the invasive ability of cells, negatively regulating E-cadherin, and promoting immunosuppression.15–17 Additionally, COUP-TFII is upregulated by transactivation of microRNA-21 (miR-21) in CRC cells, which promotes TGF-β-induced EMT by inhibiting Smad7. 18 COUP-TFII activates miR-21 expression by directly binding to the miR-21 promoter region. 18 It has been reported that miR-21 is upregulated in several cancers, suggesting it plays an important role in tumorigenesis. 19 Wang et al. 20 showed that COUP-TFII was positively expressed in 65.2% of CRC tissues compared with only 15.5% of paired non-CRC tissues. They demonstrated that COUP-TFII expression was significantly correlated with TNM stage and lymph node metastasis, but was negatively correlated with Smad4 expression. Finally, they showed that patients with higher COUP-TFII levels had reduced disease-free survival and overall survival. 20
Tumor promoting effect of COUP-TFII in prostate cancer
Most studies have shown that COUP-TFII promotes the progression of prostate cancer cells. To investigate the clinical relevance of COUP-TFII in human prostate cancer, Qin et al. 21 examined COUP-TFII expression by immunohistochemical staining in a tumor tissue microarray (TMA) comprising 407 patient specimens. They also examined correlations between COUP-TFII expression and clinicopathological characteristics. They observed that COUP-TFII expression was higher in prostate cancer tissues compared with the adjacent normal prostate epithelium and suggested that COUP-TFII expression in prostate tumor cells could serve as a predictor to stratify risk of recurrence after prostatectomy. Using genetically engineered mice models, they found that COUP-FTII has a crucial role in driving the full malignant progression of PTEN-null prostate tumorigenesis by inhibiting TGF-β negative feedback signaling. 21 In their report, COUP-TFII-overexpressing mice (COUP-TFIIOE/+) exhibited normal prostate histology, suggesting COUP-TFII alone could not initiate tumorigenenesis. 21 Wang et al. 22 demonstrated that COUP-TFII promoted prostate cancer growth by inhibiting mitochondrial pyruvate carrier 1 (MPC1). The MPC genes MPC1 and MPC2 were recently identified to control pyruvate transport through the inner mitochondrial membrane.23,24 MPC1 was also found to be downregulated in several cancers. Co-expression of MPC1 and MPC2 inhibited colon cancer cell growth. 25 COUP-TFII promotes the invasion of prostate cancer cells by upregulating FOXM1 and CENPF by directly binding to their promoters. 26 Additionally, COUP-TFII expression contributes to resistance to the anti-androgen drug enzalutamide. 26 FOXM1 is a Forkhead box-containing transcription factor that is frequently overexpressed in several cancers, including prostate cancer.27,28 Increased FOXM1 expression has been shown to contribute to cell proliferation, genomic instability, angiogenesis, metastasis, and drug resistance in cancer.28,29 CENPF is a structural protein of the kinetochore and a known target of FOXM1. Increased CENPF expression plays a crucial role in prostate cancer development.30,31 Recent reports have also found tumor promoting effects of COUP-TFII in prostate cancer, although these reports did not provide mechanistic details. For example, Lilis et al. 32 demonstrated that p-mTOR expression is correlated with COUP-TFII expression in lymphatic vessel endothelial cells and prostate adenocarcinoma cells, suggesting that p-mTOR and COUP-TFII could play an important role in lymph node metastasis. Finally, Wang et al. 33 showed that COUP-TFII is upregulated in prostate cancer spheroids and castration-relapsed VCaP-CRPC xenografts. They also showed that COUP-TFII overexpression increased cancer stem cell markers such as CD44 and CD133 and enhanced sphere formation capacity. 33
Tumor promoting effect of COUP-TFII in breast cancer
Nagasaki et al. 34 have reported that COUP-TFII positivity is detected in the nuclei of invasive breast carcinoma tissues and that its expression is correlated with clinical stage, lymph node status, histologic grade, and ERα status. They also showed that vascular endothelial growth factor-C (VEGF-C) mRNA expression was decreased in COUP-TFII-knockdown cell lines, indicating that COUP-TFII might enhance lymph node metastasis by inducing VEGF-C; 34 which is involved in lymphangiogenesis. 35 It has been shown that VEGF-C expression in breast tumor cells induces intratumoral lymphangiogenesis and enhances lymph node metastasis. 36 Qin et al. 37 reported that COUP-TFII upregulates angiopoietin-1 transcription in pericytes by directly binding to its promoter, which promoted tumor growth and metastasis in a spontaneous mammary gland tumor model.
Tumor promoting effect of COUP-TFII in other cancers
Several studies have shown that COUP-TFII can enhance cell proliferation and tumorigenesis in other cancers. Xiao et al. 38 showed that MPC1 expression is decreased by COUP-TFII in the human glioblastoma cell line U87. They also demonstrated decreasing COUP-TFII expression with a COUP-TFII-directed siRNA inhibited the proliferation of glioblastoma cells in vitro and in vivo by increasing MPC1 expression. To investigate the biological functions of COUP-TFII in renal cell carcinoma (RCC), Zheng et al. 39 performed immunohistochemical staining in RCC tumor tissues and mechanistic experiments. They reported that RCC tumor tissues had much higher COUP-TFII levels than adjacent normal tissues and that COUP-TFII siRNA knockdown inhibited cell proliferation and increased apoptosis in RCC cell lines. 39 In vivo experiments in nude mice showed that the growth of COUP-TFII knockdown RCC xenograft tumors was much slower than that of control tumors. They also reported that COUP-TFII might promote RCC progression by inhibiting BRCA1 expression because they observed that COUP-TFII knockdown resulted in increased BRCA1 expression. Silencing BRCA1 via siRNA leads to increased cell proliferation. Thus, they suggested that the effect of COUP-TFII on BRCA1 expression might be indirect. 39 BRCA1 is a tumor suppressor in breast and ovarian cancer. Previous studies have shown that BRCA1 may have tumor-suppressive functions through cell cycle control and DNA damage repair.40–43 Additionally, it has been reported that germline BRCA1 mutations may contribute to RCC development and that BRCA1 may function as a tumor suppressor in RCC. 44
Pancreatic ductal adenocarcinoma (PDAC) is a very aggressive cancer with frequent distant metastasis and recurrence. 45 Polvani et al. 45 showed that COUP-TFII is widely expressed in pancreatic cancer and that its expression is associated with the presence of lymph node and distant metastases as well as clinical stage. They observed that patients with COUP-TFII-positive PDAC had shorter survival times, indicating that COUP-TFII could be an indicator of poor prognosis. In their study, knocking down COUP-TFII by shRNA inhibited cell proliferation, invasion, and tumor growth in vivo. They suggested that COUP-TFII actions were mediated by VEGF-C. Qin et al. 46 have reported that loss of COUP-TFII can inhibit tumor angiogenesis, metastasis, and lymphangiogenesis. They suggested that loss of COUP-TFII inhibited VEGF/VEGFR-2 signaling via increasing VEGFR-1 expression. 46 They found that COUP-TFII could directly bind to the VEGFR-1 promoter to negatively regulate its expression. 46
Finally, Navab et al. 47 demonstrated stable COUP-TFII expression among 549 lung cancer specimens, and showed that COUP-TFII increased the motility and invasion of lung cancer cells by activating focal adhesion kinase (FAK) and upregulating MMP-2 and urokinase-type plasminogen activator levels.
Tumor suppressive effect and underlying molecular mechanisms of COUP-TFII
Tumor suppressive effect of COUP-TFII in gastric cancer
Ding et al. 48 showed that COUP-TFII expression was reduced in gastric carcinoma tissues and gastric carcinoma cell lines compared with normal gastric mucosa tissues and normal gastric mucosa cells (GES-1), respectively. They also demonstrated that overexpressing COUP-TFII inhibited the proliferation, migration, and invasion of gastric cancer cells in vitro and inhibited the growth and hepatic metastasis of gastric cancer in vivo; however, they did not provide the underlying mechanisms for these activities of COUP-TFII in gastric cancer. 48
Tumor suppressive effect of COUP-TFII in CRC
Shin et al. 49 reported that COUP-TFII may be a marker of good prognosis in CRC patients by evaluating the correlation between COUP-TFII expression and 3-year overall survival among 95 primary CRC patients. Yun et al. 50 have reported that absence of COUP-TFII or LXR expression are prognostic factors for poor survival, although they did not clarify the potential mechanism.
Tumor suppressive effect of COUP-TFII in prostate cancer
In contrast to the previously described reports, Song et al. 51 reported that COUP-TFII plays tumor suppressive roles by negatively regulating androgen receptor (AR) expression in prostate cancer cells. AR consists of three functional domains: the N-terminal activating domain, the middle DBD, and the C-terminal LBD. The N-terminal activating domain has been reported to directly interact with the C-terminal LBD in a ligand-dependent manner, which is required for full AR activation. 52 COUP-TFII overexpression can inhibit the androgen-dependent proliferation of prostate cancer cells, which was evidenced by decreased colony formation and decreased [3H]-thymidine incorporation. 51 COUP-TFII also interacts with AR in vitro and in vivo and binds to the DBD and the LBD of AR, disrupting the N-/C-terminal intermolecular interaction of AR. Additionally, COUP-TFII competes with coactivators such as ARA70, SRC-1, and GRIP to regulate AR transactivation and inhibit the recruitment of AR to androgen response element-containing promoters. 51 ARA70 and SRC-1 strongly interact with the AR LBD via the FXXLF motif within coactivators and connects the AR DBL/LBD complex.53–55 GRIP can also bind to both the DBD and LBD of AR and stabilize the DBD/LBD complex. 56 Breakdown of the AR DBD/LBD/coactivator complex leads to inhibition of AR transactivation.54–56 Song et al. 51 were the first to suggest that COUP-TFII could act as a potent AR corepressor.
Tumor suppressive effect of COUP-TFII in breast cancer
Several reports have shown that COUP-TFII expression has tumor suppressive effects in breast cancer, such as growth inhibition, inhibition of motility, and increased sensitivity to anticancer agents. Nakshatri et al. 57 found that COUP-TFII expression is reduced in 30% of breast cancer cells and that COUP-TFII overexpression leads to growth inhibition. They also found that COUP-TFII induced a delay in G2/M progression via upregulating p21 and reducing CDK2 activity. 57 Tamoxifen (TAM) is a widely used drug to treat and prevent estrogen receptor (ER)-positive breast cancer. 58 TAM and its active metabolite, 4-hydroxytamoxifen (4-OHT), are antiestrogens that inhibit breast cancer cell proliferation via competitively inhibiting ER and blocking the transcription of ER target genes. 58 However, most breast cancers acquire TAM resistance. 58 Riggs et al. 59 demonstrated that COUP-TFII is reduced in TAM-resistant breast cancer cells and that siRNA-mediated COUP-TFII knockdown causes TAM-sensitive cells to acquire TAM resistance. Their data showed that COUP-TFII/ERα interactions were correlated with antiestrogenic responses. Finally, they showed that reducing COUP-TFII levels could contribute to acquired TAM resistance by reducing interactions between COUP-TFII, 4-OHT, and ERα. 59
Retinoids, such as all-trans retinoic acid (atRA) and 9-cis RA, as well as retinoic acid receptor β (RARβ) have been shown to have with tumor suppressive activities, such as reduced cell proliferation and inflammation, and enhanced apoptosis. 60 RARβ expression is decreased in breast cancer, and restoring RARβ2 expression increases the sensitivity to tumor growth inhibition by retinoids. 61 COUP-TFII is required for atRA- or 9-cis RA-induced RARβ2 expression in breast cancer and can bind to the RARβ2 promoter.62,63 Consistent with a previous report, 62 Litchfield et al. 64 demonstrated by chromatin immunoprecipitation that atRA increases the COUP-TFII/RARβ2 promoter interaction. They also showed that COUP-TFII expression was correlated with ERα expression and inversely correlated with tumor grade in ERα-positive invasive ductal carcinoma, 64 suggesting that COUP-TFII might be important in differentiated ERα-expressing, retinoid-responsive, epithelial breast cancer cells. In this context, reduced COUP-TFII expression results in tumor progression and resistance to RA. Interestingly, they also demonstrated that nucleolin was associated with COUP-TFII and acted as a coactivator for COUP-TFII-mediated RARβ2 transcription in breast cancer cells. 64
Nuclear factor-kappaB (NF-κB) is a transcription factor related to immune and inflammatory responses. Recent studies have demonstrated that NF-κB activation is involved in acquired endocrine-resistant breast cancer.65–67 NF-κB target genes play key roles in oncogenic transformation, including apoptosis resistance,68,69 invasion, 70 and EMT. 71 Litchfield et al. 72 demonstrated that COUP-TFII inhibits TNFα-induced NF-κB activity in LCC9 endocrine-resistant breast cancer cells, which have higher basal and TNFα-induced NF-κB activity than the parental MCF-7 cells (endocrine-sensitive). Inhibiting NF-κB activity by overexpressing COUP-TFII leads to a downregulation of interleukin (IL)-6, intercellular adhesion molecule 1 (ICAM1), and TNFα-induced protein 3 (TNFAIP3). 72 They also showed for the first time that COUP-TFII expression was negatively associated with the NF-κB subunit genes NF-κB2, REL, RELA, and RELB, as well as the NF-κB target genes ICAM1, IL-6, and TNFAIP3 in microarray data from 298 ERα-positive breast tumors from patients treated with TAM for 5 years. 72 COUP-TFII also interacts with the NF-κB subunits RelB and NF-κB1 p50 to inhibit NF-κB binding to the promoter of its target genes. 72 Altogether, these data suggest that inhibiting NF-κB target gene expression by COUP-TFII may contribute to endocrine sensitivity. 72 Additionally, Al-Rayyan et al. 73 showed that decreased COUP-TFII expression in endocrine-resistant breast cancer cells might be caused by hypermethylation of COUP-TFII in the first exon and that 5-aza-2-deoxycytidine can reduce the methylation. Finally, Zhang et al. 74 demonstrated that high COUP-TFII expression is correlated with improved OS and distant metastasis-free survival, which might be caused by COUP-TFII-mediated inhibition of TGF-β-dependent EMT and chemoresistance.
Tumor suppressive effect of COUP-TFII in other cancers
There are several reports that demonstrate tumor suppressive effects of COUP-TFII in other cancers. Lee et al. 75 reported that COUP-TFII is decreased in ovarian cancer by microarray analysis. Hawkins et al. 76 also showed that COUP-TFII expression is significantly decreased in ovarian cancer; however, they did not find a relationship between COUP-TFII expression and clinical outcomes. Their study showed that COUP-TFII knockdown enhanced proliferation and apoptosis in TP53 wild-type ovarian cancer cell lines derived from the epithelial compartment of ovarian cancers via never in mitosis gene a-related kinase 2 (NEK2). 76 However, COUP-TFII knockdown did not affect the proliferation of TP53-mutant ovarian cancer TOV-112D cells. 76 NEK2 can mediate the ability of Ras to promote centrosome amplification and genomic instability in mammary epithelial cells. 77
Telomerase is an important enzyme in cellular immortalization and tumorigenesis that it is composed of two major subunits in humans: the RNA template and reverse transcriptase subunits, which are encoded by hTER and hTERT genes, respectively.78–82 It has been reported that COUP-TFII can inhibit hTERT transcription via binding the E-box close to its initial start site as well as two other binding sites in the hTERT promoter, 82 indicating that COUP-TFII can act as tumor suppressor.
Regulation of COUP-TFII by miRNAs
MiRNAs are small single-stranded noncoding RNAs that are important post-transcriptional negative regulators that completely or partially bind to complementary sites in the 3′-untranslated region (3′-UTR) of target mRNAs. Recent studies have focused on the regulatory role of miRNAs in tumor growth, metastasis, and progression.83–85 Tumorigenesis and metastasis are regulated by various transcription factors. Abnormal expression of miRNAs is related to tumor proliferation and invasion by inhibiting their target genes.86,87 Thus, identifying specific miRNAs and their targets that are involved in tumorigenesis and metastasis could provide clues for the diagnosis, treatment, and prevention of cancer.
Several reports have shown that COUP-TFII expression is regulated by miRNAs, reinforcing the role of COUP-TFII in cancer (Figure 3). Feng et al. 88 demonstrated that miRNA-27b is downregulated and its downregulation is correlated with increased lymphatic metastasis in gastric cancer. They showed that miR-27b inhibited proliferation, migration, and invasion of gastric cancer cells in vitro as well as metastasis to the liver in vivo. They also found that miR-27b activity was mediated by downregulating COUP-TFII via its interaction with the 3′-UTR of COUP-TFII. 88 Recent studies have demonstrated that miR-382 is significantly downregulated in CRC and prostate cancer, and that miR-382 overexpression can inhibit cell proliferation, migration, and invasion via targeting COUP-TFII.89,90 Lin et al. 26 showed that miR-101 and miR-27a can inhibit the metastasis of prostate cancer by inhibiting COUP-TFII.
Finally, miR-302 regulates stem cell differentiation via directly targeting COUP-TFII.91,92 Kang et al. 93 showed that miR-302a promotes osteoblastic differentiation by inhibiting COUP-TFII. MiR-194 promotes osteogenesis and inhibits adipogenesis by regulating COUP-TFII. 94 Thus, studies on the regulation of COUP-TFII by miRNAs suggest that miRNAs are an attractive therapeutic modality for various diseases, including cancer.
Conclusions
COUP-TFII is a transcription factor related to the regulation of tumorigenesis and an intriguing molecule through which we have gained a further understanding of cancer biology. Although many studies on the role of COUP-TFII in cancer have been performed, there are still controversies about its dual effects in tumor promotion and tumor suppression. However, its dual actions can be explained, in part, by cell type-specific effects, the presence of specific mutations in oncogenes or tumor suppressor genes, and its complicated mechanisms such as direct activation, indirect activation, direct repression, and trans-repression. Additionally, there is increasing evidence that COUP-TFII is regulated by miRNAs, suggesting that miRNAs represent a novel therapeutic option for cancer. Therefore, further studies into the molecular interactions between COUP-TFII and its transcriptional partners and identifying its ligand are warranted to further understand the role of COUP-TFII in cancer and develop new treatments.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by a grant (NRF-2017R1A2B4011428) of the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning. It was also supported by a grant (2016R1A5A2007009) of the National Research Foundation of Korea (NRF) funded by the Korean Government (MSIP).
