Abstract
With extensive perioperative use of dexmedetomidine, there has been rising concern about its side effects, which are most commonly bradycardia and hypotension. However, increased urine output or diuresis is not well recognized with perioperative use of dexmedetomidine. We report a case of intraoperative polyuria induced by dexmedetomidine. The unusual feature of this case is that only a loading dose of dexmedetomidine was administered, but there was a long-lasting and large effect of diuresis. The findings from our case suggest that clinicians should be alert to the risk of diuresis when using dexmedetomidine.
Background
Dexmedetomidine, which is a selective α2-adrenergic receptor agonist, is widely used in clinical practice because of its excellent sedative and analgesic effects. The common side effects of dexmedetomidine include bradycardia and hypotension, 1 while animal studies have shown that dexmedetomidine can also cause diuresis.2,3 We report a case of polyuria after intraoperative dexmedetomidine administration. The distinguishing feature of our case is that only a loading dose was administered, but there was a long-lasting and large effect of diuresis.
Case presentation
A 62-year-old Chinese man was scheduled for combined dissection of the tongue, mandible, and neck, right forearm free flap transplantation, mandibular titanium plate implantation, and tracheotomy. He was a smoker, 170 cm in height, and 51 kg in weight. Except for hypertension, which was treated with losartan potassium, the patient had no other medical history and denied any allergies. Two sessions of chemotherapy (docetaxel injection 120 mg, nedaplatin injection 120 mg) were completed before admission. The patient provided consent for publication of this case report, and the Institutional Review Board of the University of the Chinese Academy Of Sciences (Ningbo No. 2 Hospital) granted approval for the study protocol (YJ-KYSB-NBEY-2019-120-01).
On the day of surgery, the patient received 2 mg of intravenous midazolam as preoperative medication. Standard American Society of Anesthesiologists monitors were applied and a loading dose of dexmedetomidine was intravenously infused at 0.6 µg/kg over 10 minutes before induction. Induction was performed by propofol (80 mg), sufentanil (20 µg), and rocuronium (50 mg) followed by tracheal intubation. An invasive arterial catheter and an internal jugular vein catheter were placed. Anesthesia was maintained with sevoflurane (1% to 2%), propofol (150–200 mg/hour), and intermittent sufentanil 10 µg and rocuronium 10 mg, which were titrated to hemodynamic status and surgical requirements.
Urine output increased 30 minutes after the dexmedetomidine infusion was completed, reaching approximately 1900 mL within 2 hours. Pituitrin 2 U/hour was infused intravenously and changed to 3 U/hour 30 minutes later until the operation was over. Urine output showed a downward trend since the pituitrin treatment, and decreased to 500 ml/hour 2 hours later. Serial urine output is shown in Figure 1.

Intraoperative and postoperative urine output.
The surgery lasted for 14 hours, during which time the patient received 5500 mL of lactated Ringer’s solution, 2000 mL of hydroxyethyl starch (130/0.4), and 375 mL of packed red blood cells. The estimated blood loss was 600 mL and the total urine output was 7150 mL.
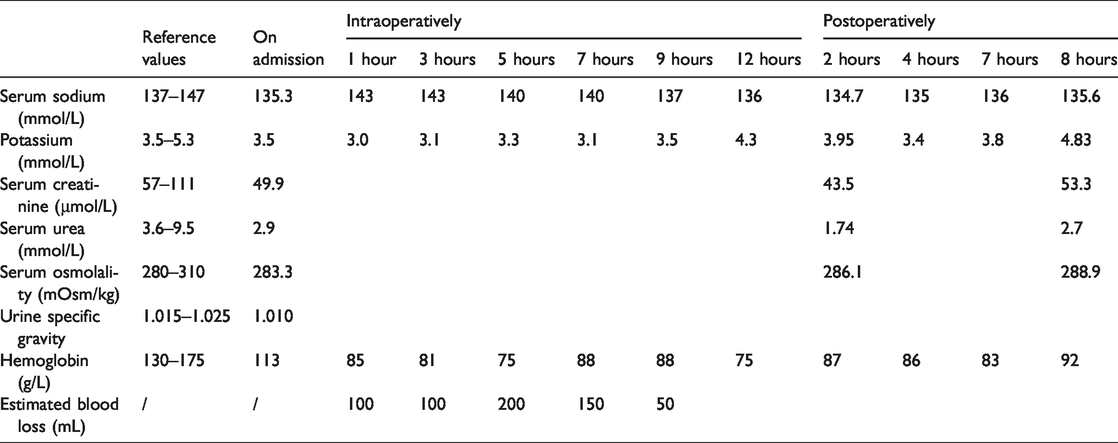
The patient’s serum sodium level was 135 mmol/L 5 days before surgery and rose to 143 mmol/L intraoperatively. The patient’s serum osmolality was within normal limits at 2 hours after surgery (Table 1). The patient was hemodynamically stable throughout the procedure without major episodes of hypotension or tachycardia.
Laboratory values preoperatively, intraoperatively, and postoperatively.
The patient was transferred to the intensive care unit after the operation with no diuresis or abnormal electrolyte and plasma osmotic pressure. He was discharged on postoperative day 18 without further related complications.
Discussion
In our case, we concluded that polyuria accidentally occurred during the intraoperative period. Common causes of intraoperative polyuria include diuretic therapy, poorly controlled hyperglycemia, and nephrogenic or central diabetes insipidus. 4 However, none of these situations applied to our patient. Because he had no previous history of diuresis, we consider that perioperative medication-related polyuria was the principal diagnosis. We reviewed his medical history and searched Lexicomp Online, and found that losartan, docetaxel, and nedaplatin are not associated with polyuria.
A literature review showed a total of eight case reports on polyuria possibly induced by dexmedetomidine2,5–11 (Table 2). Coincidently, in four of the right cases, the patients underwent the same type of surgery, spinal fusion surgery.2,5,6,8 An interesting hypothesis is an association between the type of surgery and polyuria. However, there have been no reports on oral and maxillofacial surgery (as in our case) and polyuria. Therefore, the effect of the type of surgery on polyuria requires further research.
Comparison of previous literature on polyuria induced by dexmedetomidine.

Additionally, our patient was only provided a loading dose without continuous infusion. However, the single dose of dexmedetomidine still resulted in a urine output of more than 500 mL/hour for up to 5 hours, which considerably exceeded the 2-hour time frame in the previously reported cases. This finding suggested that dexmedetomidine use could be a “trigger point” of a diuretic effect, despite the dose or infusion rate. Kirschen et al.
8
showed that there was no dose-dependent relationship between the dexmedetomidine infusion rate and urine output (
Possible mechanisms of dexmedetomidine causing polyuria have been investigated in animal research.3,12–15 Existing approaches inducing polyuria are as follows. (1) In the central approach, dexmedetomidine inhibits arginine vasopressin (AVP) release from the isolated hypothalamic paraventricular nucleus and paraventricular magnocellular neurons. (2) In the peripheral approach, dexmedetomidine directly acts on the renal tubules, attenuating the effect of AVP on the renal tubules by interference in release of renin or atrial natriuretic peptide. (3) With regard to other approaches, dexmedetomidine acts peripherally via imidazoline receptors, leading to increased sodium and free water excretion in a non-AVP dependent manner at sites outside of the collecting duct. Based on the above mechanism, dexmedetomidine-induced polyuria should be accompanied by increased serum sodium levels, lowered urine specific gravity, and higher serum osmolarity. However, the serum sodium level and serum osmolality of this patient were within the normal range. Additionally, intraoperative urine gravity was not obtained in our patient because dexmedetomidine was not identified as a plausible cause until the end of our patient’s clinical course. We speculate that the unexpected condition of our patient was due to the large and rapid concomitant infusion of lactated Ringer’s solution that was administered during the period of diuresis.
Pituitrin is an extract of bovine posterior pituitary containing oxytocin and vasopressin. To some extent, the antidiuretic effect of pituitrin is the same as that of AVP. AVP binds to vasopressin-2 receptors on the basal membrane of the renal collecting tubular cell, which leads to an increase in permeability of tubular fluid by increasing the amount of aquaporin-2 water channels. 16 This in turn leads to water retention, dilutional hyponatremia, and urinary concentration because of the higher intramedullary osmolality compared with that of tubular fluid. In our case, pituitrin appeared to be effective for treating polyuria because urine output dropped from 850 mL/hour to below 500 mL/hour, accompanied by a decrease in serum sodium concentrations.
Propofol and sevoflurane can lead to polyuria. In our case, propofol was used as sedation in the intensive care unit after surgery and no polyuria was detected at this time. Therefore, we consider that propofol was not the cause of polyuria. Sevoflurane can cause a transient impaired aquaporin-2 response and increased intrinsic AVP, 18 but polyuria associated with sevoflurane is rare.17,19 Because performing specific tests to rule out sevoflurane are not possible, sevoflurane and dexmedetomidine are possible contributors to polyuria. Therefore, there is the possibility that sevoflurane could have been involved in the polyuria in our patient.
Generally, despite findings in animal research, clinical studies are still insufficient on dexmedetomidine-induced diuresis, which is rarely reported. We speculate that, in some cases, diuresis caused by dexmedetomidine is not substantial enough to be noticed, or polyuria could vary because of individual differences and heterogeneity of patients. Clinicians should be alert to the potential risk of diuresis when using dexmedetomidine.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by a grant from the Medical Scientific Research Foundation of Zhejiang Province (2018KY157). We declare that the funders had no role in the protocol design and collection, analysis, interpretation of data, or writing of the manuscript.
