Abstract
Fine motor and manual dexterity deficits are the main causes of significant physical and psychosocial impairments in stroke survivors. Transcranial direct current stimulation (tDCS) is a noninvasive brain stimulation technique used to modulate brain activity and improve clinical outcomes. This study was performed to investigate the efficacy of dual-hemispheric tDCS of the primary motor cortex (M1) in a patient with stroke exhibiting cortical sensation deficits. A double-blind, sham-controlled, single-case study was conducted. The patient underwent 30 sessions of sham tDCS followed by 30 real-stimulation sessions over both M1 cortices. Each session involved 20 minutes of 2-mA stimulation (current density, 0.08 mA/cm2; total charge density, 0.096 C/cm2). Functional measures were assessed using the Action Research Arm Test, grooved pegboard test, nine-hole peg test, and box and block test at several time points. Structural and diffusion tensor imaging data were acquired before (t0) and after (t1) stimulation. Slight improvements following sham tDCS were observed. However, following real stimulation, all results at all time points were clinically significant. Higher fractional anisotropy of the corticospinal tract and regional gray matter density were positively correlated with better recovery of fine motor skills. tDCS intervention induced functional improvement and structural changes in this patient with stroke.
Keywords
Introduction
Persistent upper extremity impairments are frequent consequences of stroke and may lead to profound disabilities and reduced quality of life.1,2 Sensory guidance plays a critical role in motor control, specifically in dexterous hand movements. 3 Effective object grasping and manipulation, hallmarks of human dexterity, are based on three essential characteristics of sensorimotor integration: the capacity to generate independent finger movements, the ability to transform different sensory information concerning properties of the object to be grasped using appropriate hand configuration, and delicate somatosensory control of finger movements.4,5 A lack of sensory deficits is a significantly positive predictor of manual dexterity recovery. 6 However, improving dexterity recovery following a stroke remains a considerable clinical and scientific challenge. 6 Although rehabilitation interventions have shown some positive results, the extent of improvement remains insufficient.
Transcranial direct current stimulation (tDCS), a noninvasive brain stimulation technique, has yielded promising results in cognitive enhancement and sensorimotor retraining. 7 Application of tDCS has improved functional recovery after stroke through modulation of cortical excitability and neural activity. 8 Researchers have proposed that unilateral stroke induces an interhemispheric imbalance in which the contralesional hemisphere becomes more excitable and excessively inhibits the ipsilesional hemisphere. 9 This abnormal interhemispheric inhibition is the hypothetical model underpinning the experimental therapeutic application of tDCS.10,11 Given the interhemispheric competition that occurs following a stroke, 12 functional motor recovery can be achieved by enhancing the neuronal excitability of the affected hemisphere by applying anodal stimulation or inhibiting the activity of the unaffected hemisphere through application of cathodal stimulation.8,13 Several researchers have proposed the use of dual-hemispheric tDCS to rectify interhemispheric imbalances after stroke, which can induce neural plasticity and improve functional recovery.8,13,14 Brain imaging data combined with clinical data might be used to tailor therapeutic interventions and provide prognostic information. 15 Structural brain imaging techniques can be used to assess the structural integrity of white matter tracts following a stroke.16,17 For example, diffusion tensor imaging (DTI) measures have been shown to be potential biomarkers for predicting outcomes in patients with stroke undergoing tDCS. 18 The present case study describes a man who developed a frontoparietal stroke with cortical sensory deficits (stereognosis and graphesthesia) but with preservation of sensory and motor processing. Because the primary motor cortex (M1) is a crucial area in the network of brain regions responsible for receiving and processing sensory information, 19 it is hypothesized that delivering dual-hemispheric tDCS to both M1 cortices would induce neuroplasticity changes and improve both the cortical sensory deficits and consequently the manual dexterity. DTI and T1-weighted imaging were performed to measure the integrity of the corticospinal tract (CST) and the structural changes before and after the tDCS intervention.
Case report
Patient information
A 48-year-old left-handed man developed an ischemic right frontoparietal lesion 2 years prior to the study. The patient fully recovered with the exception of persistent stereognosis and graphesthesia in the left (dominant) hand. Sensory examination was performed using the Nottingham Sensory Assessment. 20 The patient demonstrated “normal” tactile sensation in all modalities as shown by a score of 2, which indicates normal sensation. However, he demonstrated “impaired” higher cortical sensation as shown by a score of 1, indicating the presence of stereognosis and graphesthesia. The presence of graphesthesia was also assessed by asking the patient to identify characters (letters or numbers) being traced onto his palm or fingertip while his eyes were closed. Manual muscle testing revealed a muscle strength score of 4+/5 in the upper extremity muscle groups. A neuropsychological assessment revealed no cognitive dysfunction.
The patient provided written informed consent, and the study was approved by the Institutional Review Board of Imam Abdulrahman Bin Faisal University. This was a double-blind, sham-controlled crossover study in which the patient underwent 30 sessions of sham stimulation (5 consecutive days of intervention per week for 6 weeks). Following a 2-week washout period, the patient underwent another 30 sessions of real stimulation. He received only tDCS with no accompanying physical therapy or training during application of the brain stimulation and received no treatment throughout the follow-up period.
Interventions
A Magstim current stimulator (The Magstim Company Ltd., Whitland, Carmarthenshire, Wales, UK) was used to administer tDCS. Using the International 10–20 system, 21 the anode was placed over the ipsilesional M1 (C4) while the cathode was placed over the contralesional M1 (C3) using 5- × 5-cm saline-soaked electrodes. Each session included 20 minutes of stimulation with a set intensity of 2 mA, yielding a current density of 0.08 mA/cm2 and total charge density of 0.096 C/cm2, which fall within the tDCS safety guidelines. 22 Sham tDCS was ramped up only during the first 30 seconds of the session to ensure blinding of the procedure. 23
Functional outcome measures
The Action Research Arm Test (ARAT) was used to evaluate the functional ability of the upper extremity. The ARAT assesses the ability to handle objects differing in size, weight, texture, and shape. The grooved pegboard test (GPT) was used to evaluate fine motor speed and dexterity. The score is based on the number of pegs inserted and the time it takes to accomplish the task; thus, shorter times with higher insertion rates are associated with better test outcomes. The nine-hole peg test (9HPT) was used to measure fine manual dexterity and speed, and the box and block test (BBT) was used to assess gross manual dexterity. The functional outcome scores following sham stimulation were recorded before tDCS at baseline (t0) and immediately after tDCS (t1), while the scores following real stimulation were recorded at t0, t1, 1 month after tDCS (t2), and 3 months after tDCS (t3). The procedure was conducted by an independent assessor to ensure blinding. No formal statistical tests were used because each outcome was represented by a single score. However, the minimal clinically important difference (MCID) was used as a valid change in the score; this parameter assesses meaningful differences that are not due to chance. 30 Within-condition data comparisons were performed between the post-tDCS and pre-tDCS measurements (t1–t0 comparison) for both the sham and real stimulations. Between-condition data comparisons were performed between the post-tDCS measurements for the real and sham stimulations [post-real (t1) vs. post-sham (t1) comparison].
Imaging data acquisition
High-resolution three-dimensional T1-weighted images and DTI data were acquired at t0 and t2 using a 1.5T Siemens Aera scanner (Siemens, Munich, Germany) with an eight-channel head coil. T1 structural data were acquired using a 1-mm isotropic magnetization-prepared rapid acquisition gradient-echo sequence with the following parameters: repetition time (TR) = 2200 ms; echo time (TE) = 2.9 ms; flip angle = 9°; field of view (FOV) = 256 × 256 mm2; matrix size = 256 × 256; number of slices = 126; and acquisition time = 9.2 min.
Diffusion tensor images were also acquired using a 2-mm isotropic single-shot echo-planar imaging sequence with the following parameters: TR/TE = 8200/81 ms; matrix size = 256 × 256; FOV = 240 × 240 mm2; flip angle = 90°; and excitation number = 3. Images were collected along 25 orthogonal diffusion gradient directions, with a b-value of 0 and 1000 s/mm2.
Data preprocessing
DTI data were first preprocessed and adjusted for motion artifacts and eddy current distortions using the Diffusion Toolbox of the FMRIB Software Library, version 4.19 (FSL library of imaging analysis tools: www.fmrib.ox.ac.uk/fsl/). 24 Image registration and brain mask extraction were performed using the Brain Extraction Tool. 25 The diffusion tensor and fractional anisotropy (FA) maps were calculated using MedINRIA software. 26 Tractography was performed using the Diffusion Toolkit and TrackVis. 27 Using the interpolated streamline algorithm with a step length of 0.5 mm, fiber tracking was performed with a threshold angle of 30° and threshold FA of 0.15. The threshold for statistical significance was set at p ≤ 0.05. A one-sample t-test was used to compare each DTI measure between the ipsilesional and contralesional CST.
Voxel-based morphometry analysis was performed on the three-dimensional T1-weighted images (pre- and post-stimulation) using Statistical Parametric Mapping (SPM12) software (http://www.fil.ion.ucl.ac.uk/spm/software/spm12/) in a MATLAB environment (MathWorks, Natick, MA, USA). First, a binary lesion (infarction) mask was incorporated before the segmentation step to prevent misclassification of the tissue type during affine transformation and nonlinear adjustments. 12 The Diffeomorphic Anatomical Registration Through Exponentiated Lie Algebra (DARTEL) algorithm 13 was used to normalize the images to the Montreal Neurological Institute (MNI) space. The signal change in regional gray matter density (rGMD) between the pre- and post-stimulation images was computed at each voxel, including only voxels with rGMD values of >0.10 in both the pre- and post-stimulation magnetic resonance images. The pre-rGMD images were subtracted from the post-rGMD images, resulting in maps representing the change in rGMD between the pre- and post-magnetic resonance images.
Results of functional outcome measures
The ARAT score increased by 2 points following sham stimulation (t1–t0 comparison) and showed a dramatic increase of 27 points following real stimulation (t1–t0 comparison) with a continuous improvement at time points t2 (28 points) and t3 (29 points); this improvement reflected better arm motor function. The length of time required to perform the GPT decreased by 10 seconds following sham stimulation (t1–t0 comparison) and showed a further decrease of 75 seconds following real stimulation (t1–t0 comparison) with a continuous reduction at time points t2 (80 seconds) and t3 (85 seconds). This reduction in time taken to perform the GPT indicated better fine motor control. The number of displaced cubes in the BBT increased by two cubes per minute following sham stimulation (t1–t0 comparison) and by eight cubes per minute following real stimulation (t1–t0 comparison) with a continuous increase at time points t2 (48 cubes/minute) and t3 (67 cubes/minute). These higher numbers of displaced cubes indicated better gross dexterity. The time taken to perform the 9HPT decreased after the sham and real stimulations. In addition, the Nottingham Sensory Assessment was used to assess cortical sensation deficits. The patient showed no difference (“impaired” sensation, score of 1) after sham stimulation compared with baseline. However, he showed “normal” sensation (score of 2) after real stimulation, indicating better tactile sensation. The difference between the post- and pre-sham stimulation measurements across all functional outcome measures did not reach the MCID values. 30 Therefore, a follow-up assessment for the sham stimulation was not performed. However, in the real stimulation (t1–t0 comparison), the change in scores was greater than the MCID value for each outcome measure. In the ARAT, the difference (score of 27) was greater than the MCID value of 5.7. 31 The change in the GPT result (75 seconds) was greater than the MCID value of 6.5 seconds. 32 Similarly, in the 9HPT, the change (32 seconds) was higher than the MCID value of 30 seconds. 32 Finally, the change in the BBT (eight cubes/minute) was greater than the MCID value of five cubes/minute. 32 Comparable between-condition data [post-real (t1) vs. post-sham (t1) comparison] showed that real stimulation elicited greater outcome effects than did sham stimulation according to the MCID values. The change in scores was greater than the MCID value for each outcome measure, as shown in Figure 1.
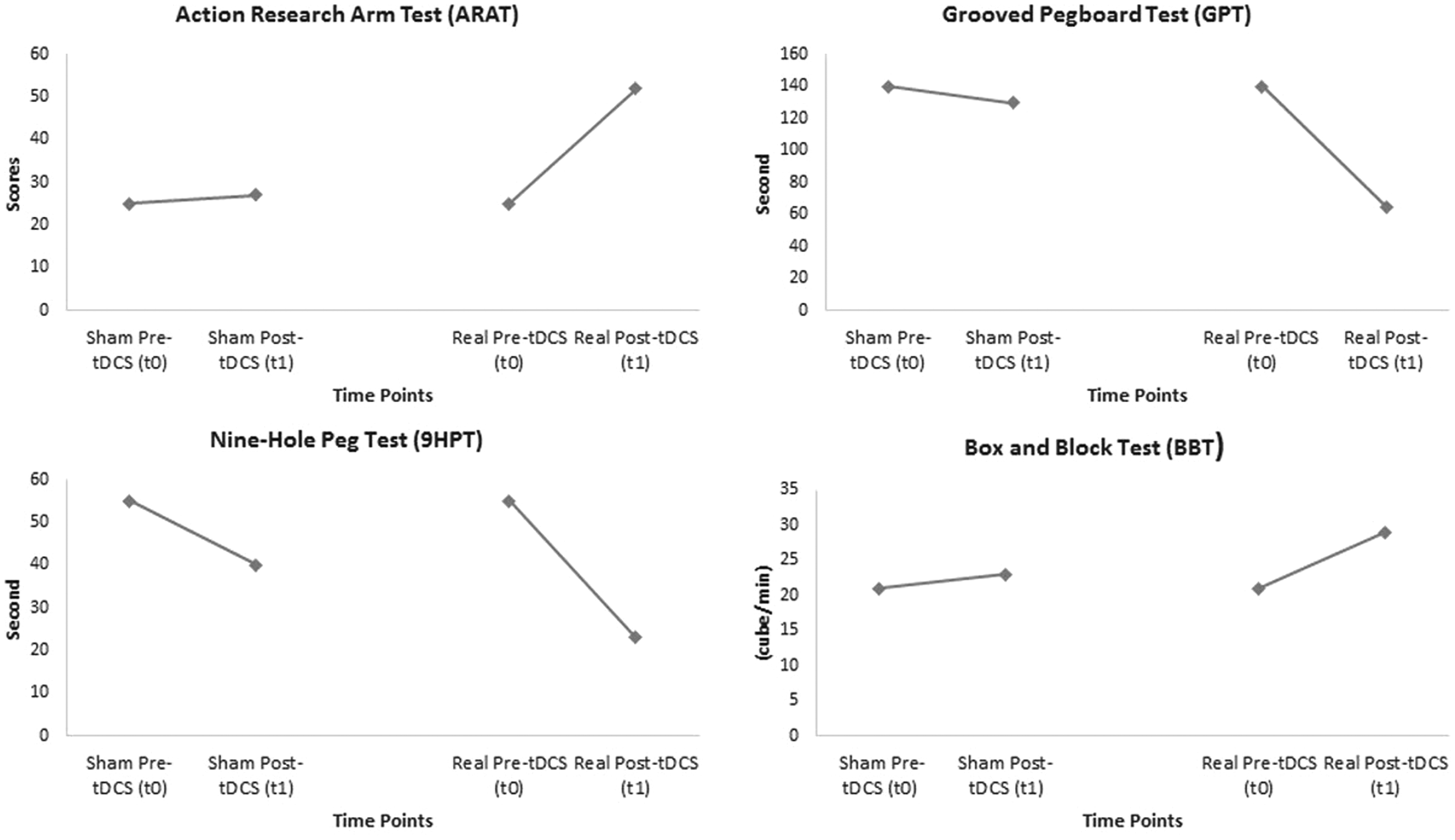
Graphical representation of outcome measures across different time points following sham stimulation, real stimulation and the difference between real and sham stimulation. tDCS, transcranial direct current stimulation.
Results of imaging data
DTI analysis and tractography were performed to assess CST integrity. The FA index of the ipsilesional portion of the CST and of the corresponding region in the contralesional hemisphere was calculated across two time points (t0 and t1 data). Voxel-wise analysis showed significantly increased mean FA values in the ipsilesional region of the CST (t0, 0.19 ± 0.11; t1, 0.21 ± 0.14; t1 vs. t0: t = 7.32, p < 0.001) but not on the contralesional side (t0, 0.81 ± 0.14; t1, 0.74 ± 0.16).
Although a second-level voxel-based morphometry analysis could not be carried out for this single patient, a higher GMD was found in the ipsilesional M1 (MNI: x = 36, y = −19, z = 55) after stimulation than before stimulation. Notably, a higher ipsilesional hemisphere GMD in the primary somatosensory cortex (S1) (MNI: x = 19, y = −43, z = 61) was detected after stimulation than before stimulation. A density increase in the premotor area (MNI: x = 18, y = −9, z = 60) was also observed (Figure 2).

Regional gray matter density across the primary motor cortex (M1), primary somatosensory cortex (S1), and premotor area (PMA) before and after real transcranial direct current stimulation (tDCS).
Discussion
This study was performed to investigate the efficacy of bilaterally applied dual-hemispheric tDCS, particularly across the M1 cortices, on the manual dexterity of a patient with stroke who developed cortical sensation deficits. The findings suggest that dual-hemispheric tDCS might have effects on cortical sensation (specifically concerning stereognosis and graphesthesia) 13 and that these effects are associated with improved hand dexterity. Although all functional outcomes assessed fine hand movement, tactile sensory signals are essential for skilled and dexterous manipulation. 36 A potential explanation for the progressive improvement over 3 months may be related to gradual acquisition of fine motor skills by persistent hands-on interactions. Although the present functional recovery results were obtained from only a single patient, the MCID 30 was detectable over time for each functional outcome measure used, highlighting the importance of these findings.
The imaging data clearly illustrated increased FA values in the ipsilesional CST following real stimulation. This finding is in line with previous studies that demonstrated the FA of CST integrity to be a predictive measure of upper limb recovery after stroke.18,34 Changes in cortical structural integrity were associated with improved manual dexterity, which demonstrates a strong correlation between cortical sensation and dexterity measures.15,16 Moreover, better manual dexterity was associated with an increase in GMD in the ipsilesional hemisphere after real stimulation. This increase in GMD in the ipsilesional M1 was not surprising because the stimulation targeted this area. However, increased GMD was also found in the ipsilesional S1 and premotor area, which is consistent with the findings of previous studies. 17 Increased GMD in the S1 following M1 stimulation is primarily mediated by direct density intracortical projections from the M1. 35 A possible explanation for this is that repeated dual-hemispheric tDCS induces synaptic plasticity that contributes to the process of sensorimotor reintegration, which in the context of functional improvement, as seen in this case, is reflected in improved fine motor movement. 35
In summary, multiple sessions of dual-hemispheric tDCS specifically targeting the M1 cortices improved cortical sensation more than the implementation of sham stimulation. This indicates that the M1 is a critical node in sensory integration processing. 19 High-order sensory integration was enhanced as evidenced by increased FA values and changes in rGMD. Improved functional outcomes were accompanied by plasticity changes involving the structural reorganization of the gray and white matter architecture. Because of a lack of follow-up data related to sham stimulation, it is very difficult to conclude that the after-effects of real stimulation in terms of functional recovery are better than those of sham stimulation.
An intrinsic limitation of this study is the fact that the functional and imaging findings were only drawn from the data of a single patient; therefore, the results cannot be generalized. Thus, group studies and further research are needed to investigate the functional, neuroanatomical, and neurophysiological substrates of cortical sensory modalities.
Footnotes
Acknowledgements
The author thanks Mr. Abdullah Alghazwani and Mr. Abdullah Althebiani, from the Department of Physical Therapy, College of Applied Medical Sciences, Imam Abdulrahman Bin Faisal University, Dammam, Saudi Arabia, for their great help in the data collection and assessment. Special thanks must also go to Dr. Bander Al-Dhafery, a consultant from the Department of Radiology, College of Medicine, Imam Abdulrahman Bin Faisal University, Dammam, Saudi Arabia, for his great help in acquiring and reading the imaging data.
Declaration of conflicting interest
The author declares that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
