Abstract
Breast cancer gene 1 and 2 (BRCA1 and BRCA2) are human tumor suppressor genes. BRCA mutations increase the risk for breast, ovarian, and gastric cancer. However, double heterozygosity for BRCA1 and BRCA2 mutations in gastric cancer have not been reported and their clinical significance is unclear. In this study, a 52-year-old Chinese male patient with gastric cancer was chosen for analysis. A tumor tissue biopsy and blood sample were collected, and next-generation sequencing-based deep panel sequencing was performed on the IlluminaNextSeq-500 platform. Comprehensive genomic alterations of 450 cancer-related genes and 47 tumor susceptibility genes were analyzed. Here, we report for the first time a case of gastric cancer that carried both BRCA1 S1841Vfs*2 and BRCA2 Q1886* heterozygous mutations. Unfortunately, the patient was resistant to olaparib treatment. Further RNA analysis revealed that only the wild-type alleles of BRCA1 and BRCA2 were expressed, although genetic BRCA1 and BRCA2 mutations were present in the patient. This genetic finding may explain the mechanism for primary resistance to olaparib observed in the BRCA1/2-mutated patient.
Introduction
Breast cancer gene 1 and 2 (BRCA1 and BRCA2) are common tumor susceptibility loci, and individuals with germline mutations in either of these two genes have a higher risk of early onset breast and ovarian cancer. 1 Generally, hereditary cancers caused by germline mutations of BRCA are found with a single mutation in BRCA1 or BRCA2; simultaneous mutations in both genes are rare. 2 In a breast cancer pedigree from Spain, Caldes et al. 3 reported inherited heterozygous mutations of BRCA1 A1708E and BRCA2 2098*. Nakamura et al. 4 reported a case of familial breast cancer carrying BRCA1 L63* and BRCA2 5804del4 mutations.
Germline mutations in the BRCA1 and BRCA2 genes are associated with a high risk of specific cancer types, including breast and ovarian cancer. 5 A previous study suggested that both BRCA1 and BRCA2 proteins are independent predictors of favorable prognosis in gastric cancer. 5 Additionally, in The Cancer Genome Atlas (TCGA) of gastric cancer, BRCA1/2 mutations have not been detected. 6 Cases of gastric cancer that carry both BRCA1 and BRCA2 mutations have not been previously reported. In this study, we report a gastric cancer case with both BRCA1 and BRCA2 mutations and further analyze the mechanism of resistance to olaparib treatment.
Case presentation
This study was approved by the ethics review committee of Shanxi Province Tumor Hospital, and written informed consent was obtained from the patient. A 52-year-old Chinese male with a family history of cancer sought evaluation in our hospital for upper abdominal discomfort. A computed tomography (CT) scan revealed irregular thickening of the stomach, enlargement of the lymph nodes, and multiple pulmonary nodules. Gastroscopy confirmed a lesion in the gastric cardia, and pathological examination suggested a stage IV poorly differentiated adenocarcinoma. The patient received tegafur (S-1), oxaliplatin, and paclitaxel liposome for 6 cycles as first-line therapy and obtained stable disease (SD); however, he subsequently progressed after oral S-1 maintenance for 3 cycles. The patient was then switched to FOLFIRI therapy (leutamic acid, leucovorin, and fluorouracil) for 6 cycles, but progressed after 5 months of treatment. Subsequently, the patient received combination therapy of cisplatin and pemetrexed for another 2 cycles before switching to apatinib. Three months after the initiation of apatinib, a CT scan revealed disease progression with primary progression of the gastric lesion and widespread metastases to different organs, including the spleen, liver, left lung, and pleura.
After multiple lines of systemic therapies had failed, a next generation sequencing (NGS)-based comprehensive genomic profiling analysis was performed on a primary gastric biopsy specimen and matched normal blood cells that were obtained at the time of the latest disease progression. NGS results indicated that the patient harbored somatic alterations of BRAF G596R and EGFR amplification, and two germline mutations of BRCA1 S1841Vfs*2 (c.5521delA) and BRCA2 Q1886* (c.5656C>T). Considering the genomic alterations, the patient was given olaparib treatment. One month later, progressive disease was noted. Specifically, both primary and metastatic lesions as well as new nodules were detected (Figure 1). The patient discontinued olaparib treatment and was switched to cetuximab in combination with FOLFIRI; however, 3 weeks later the disease progressed again. Finally, the patient began palliative treatment.
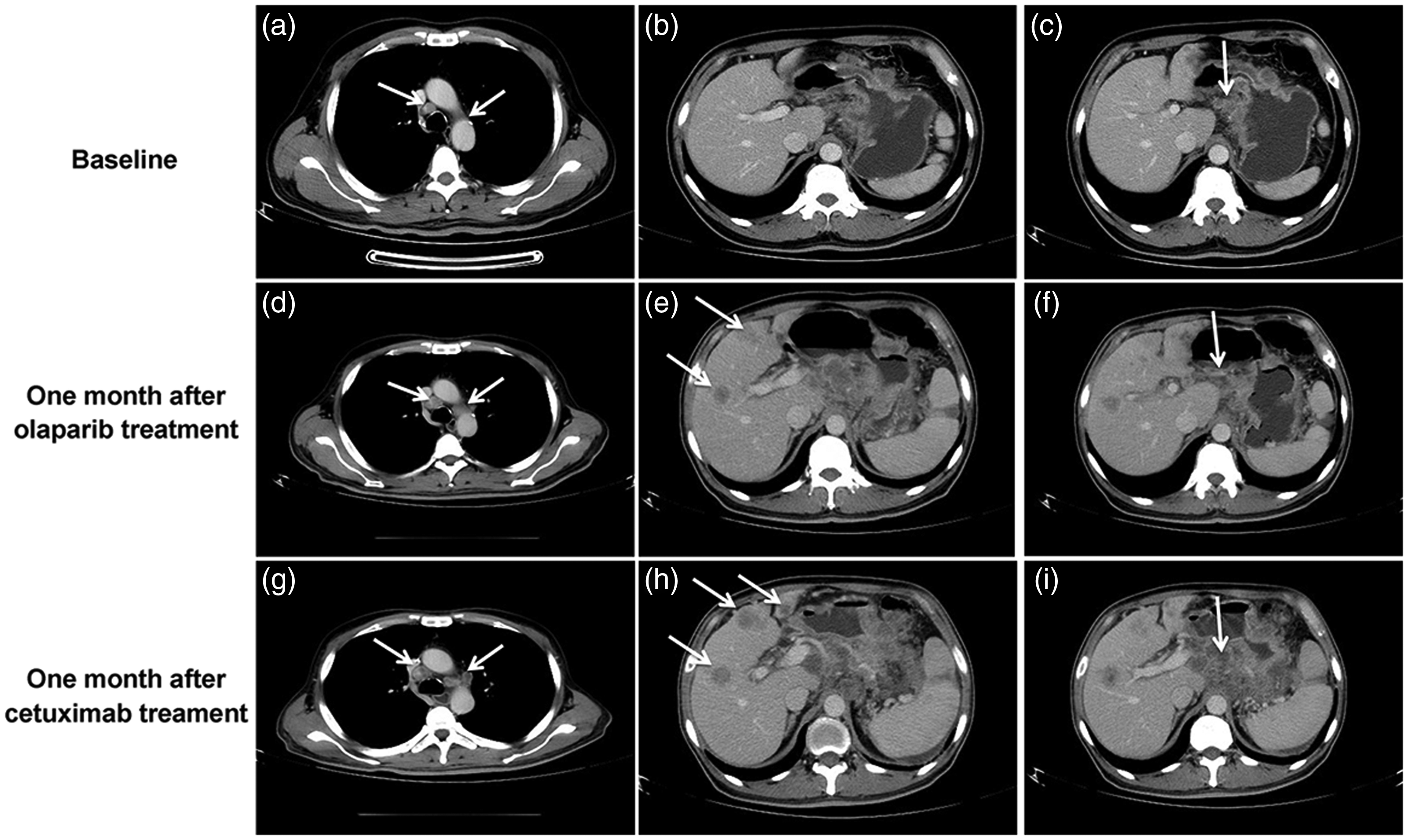
Changes in the solid nodules of the 52-year-old male patient with gastric cancer.
Due to the failed response to olaparib therapy, we collected peripheral blood from the patient for further expression analysis. Polymerase chain reaction (PCR) and Sanger sequencing for BRCA1 S1841Vfs*2 and BRCA2 Q1886* were performed. Although the patient carried both BRCA1 S1841Vfs*2 and BRCA2 Q1886* mutations, the results showed that only the wild-type alleles were expressed in the patient (Figure 2), which indicated that wild-type allele specific expression of these two loci may be the mechanism of olaparib resistance.

Sanger sequencing analysis of the mRNA expression of BRCA1 S1841 (c.5521A) (a) and BRCA2 Q1886 (c.5656C) (b) in the patient’s peripheral blood cells.
Based on our finding of BRCA1 and BRCA2 germline mutations in this patient, saliva DNA samples were collected for analysis of BRCA1 from all available relatives using Sanger sequencing (Figure 3). We found that the patient’s mother did not harbor either of the two mutations, but one of his sisters harbored a BRCA2 Q1886* mutation. The genotype of the patient’s deceased father could not be determined. It is possible that BRCA1 S1841Vfs*2 was a de novo mutation derived from our patient because the BRCA1 mutation was detected only in our patient and not in other members of his family.

Family pedigree of the patient.
Discussion
BRCA1 S1841Vfs*2 (rs80357721) and BRCA2 Q1886* (rs80358790) are two mutations associated with a high prevalence of familial hereditary breast and ovarian cancer.7,8 BRCA1 S1841Vfs*2 is a mutation that is always associated with ovarian cancer in the Chinese population. 9 However, the occurrence of simultaneous BRCA1 and BRCA2 mutations is very low. In a sporadic breast cancer study, BRCA1 and/or BRCA2 deleterious mutations were detected in 2.5% (20/793) of samples, 10 and double heterozygosity for BRCA1 and BRCA2 mutations were found in 0.3% of an Ashkenazi population of breast cancer patients. 11 According to a study of five Korean breast cancer patients, the age of breast cancer onset in BRCA1/2 double-mutant patients was relatively young (33 years), and the spectrum of disease varied from stage I/II (early), high grade, and almost a triple-negative phenotype. 12 Only one tumor tissue was positive for progesterone receptor detection using immunohistochemistry. 12 Until now, there have been no reports of BRCA1 and BRCA2 double-mutant gastric cancer. Genetic analysis of the patient’s family revealed that one of the patient's sisters also carried the BRCA2 Q1886* mutation, and the patient’s mother was negative for both mutations, which indicated that BRCA2 Q1886* may have arisen from the paternal lineage. However, the BRCA1 S1841Vfs*2 mutation was not identified in any other family member except for the patient; therefore, we cannot rule out the possibility of this being a new mutation site.
Currently, poly ADP-ribose polymerase (PARP) inhibitors such as olaparib, rucaparib, and niraparib have been approved by the Food and Drug Administration (FDA) for advanced ovarian cancer patients with deleterious or suspected deleterious BRCA1/2 mutations. Olaparib monotherapy also showed promising clinical benefits for germline BRCA1/2-mutant triple negative breast cancer. Due to DNA homologous recombination defects, BRCA1- or BRCA2-deficient tumors are highly sensitive to treatment with double-strand DNA break inducers; however, patients with BRCA1- or BRCA2-deficiency also showed differences in clinical response to PARP inhibitors. One study suggested that a lack of the RING domain in BRCA1 resulted in poor responses to PARP inhibitors and tumor progression. 13 The RING domain of the BRCA1 protein may be a marker for double-strand DNA break response in cancer patients. 14 Similarly, although the patient described herein carried heterozygous genetic mutations of BRCA1 S1841Vfs*2 and BRCA2 Q1886*, there were no transcriptional mutations in the BRCA1 and BRCA2 RNA transcripts. In contrast, only the wild-type alleles were expressed, which suggested that the primary resistance to olaparib may be due to allele specific expression of BRCA1 and BRCA2.
Conclusion
This is the first reported case of a gastric cancer patient carrying genetic mutations in both BRCA1 and BRCA2; however, the patient only had specific expression of the wild-type BRCA1/2 alleles, which resulted in no clinical benefit from olaparib therapy. In summary, we suggest that in addition to the genetic mutations of BRCA1 and BRCA2, mRNA expression profiles should be considered in the clinical application of PARP inhibitors.
Footnotes
Declaration of conflicting interest
Junping Shi, Shuo Zhang, Rui Wang, and Ming Yao are employees of OrigiMed; the other authors have no conflicts of interest to declare with this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
