Abstract
Objective
Knee osteoarthritis (KOA) is a chronic inflammatory disease. The monocyte–lymphocyte ratio (MLR) was reported to be a non-invasive, cost-effective marker in various systemic diseases, but it has not yet been investigated in KOA. This cross-sectional study evaluated the diagnostic value of MLR in KOA.
Methods
Two hundred and five KOA patients and 120 healthy control subjects were enrolled. Patient data, including age, sex, blood cell counts, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) levels, red blood cell distribution width, and the Kellgren–Lawrence (KL) score were recorded.
Results
One hundred nineteen patients (55 men, 64 women) were included, with a mean age of 55.47 ± 9.23 years. KOA patients showed a significantly higher MLR, neutrophil–lymphocyte ratio (NLR), and platelet–lymphocyte ratio (PLR) than controls. The MLR area under the curve was 0.81, which was higher than that of NLR and PLR. Multiple logistic regression analysis revealed blood MLR as an independent predictor of KOA. Correlation analysis showed that MLR was positively correlated with ESR and CRP levels. MLR and NLR were significantly higher in KL4 patients than in KL1–3 patients.
Conclusions
MLR has a high diagnostic value for KOA, so could be a reliable disease marker.
Keywords
Introduction
Osteoarthritis (OA), which is associated with age, sex, obesity, metabolic disorders, and genetic and biochemical factors, is a chronic disease characterized by joint destruction, bone hyperplasia, and articular cartilage degeneration.1,2 The known pathogenesis of OA mainly includes immunological, bone metabolism, and biomechanical factors, and knee osteoarthritis (KOA) is the most common form of OA.3,4 OA is typically accepted as an inflammatory or immune disease, although it was considered a ‘wear and tear’ disease in past decades.5,6 Many studies reported that numbers of lymphocytes, neutrophils, monocytes, and platelets are significantly altered during systemic inflammation.7–9
KOA is a chronic, progressive bone joint disease. Typically, once pain and dysfunction are noticed, the stable and complex pressure-resistant frame of articular cartilage has already undergone destruction. 10 Therefore, early diagnosis has become the top priority in the prevention and control of KOA. The most common diagnostic method is imaging, such as radiography. However, early imaging changes in KOA are not obvious. 11 Oak et al reported that radiography can detect joint space width in KOA, but detectable changes only occur in a subset of patients, and changes over time are small. 12 Thus, an objective and non-invasive diagnostic predictor suitable for use at an earlier disease stage is urgently required.
Immune and inflammatory systems are activated in KOA pathology,13,14 and monocytes, neutrophils, lymphocytes, and platelets play important roles. The neutrophil–lymphocyte ratio (NLR) and platelet–lymphocyte ratio (PLR) can reflect the balance of the immune response, and were reported to be independent and inexpensive predictors of many inflammatory and immune diseases.15–17 Indeed, the NLR and PLR were shown to be significantly altered in diseases including hypertension, chronic heart failure, ischemic stroke, peripheral arterial disease, coronary artery disease, and rheumatoid disease.7,18,19
Few studies have reported the diagnostic value of the NLR and PLR in KOA. When predicting severe knee OA, a blood NLR ≥2.1 had 77% specificity and 50% sensitivity according to receiver operating characteristic (ROC) curve analysis. 16 Some rheumatic and cancer studies reported the monocyte–lymphocyte ratio (MLR) as a new predictor of disease severity.20,21 However, no studies have reported the diagnostic value of the MLR. Therefore, in this study, we evaluated the predictive ability of the MLR compared with the NLR and PLR in KOA.
Patients and methods
Patient characteristics
Two hundred and five patients who were hospitalized in the Department of Orthopedics, Shenzhen Hospital of Traditional Chinese Medicine between 1 January 2016 and 31 July 2018 were enrolled in this study. All patients fulfilled the 2015 Guidelines for the Diagnosis and Treatment of Osteoarthritis issued by the Orthopaedics Committee of the Chinese Medical Association. Patients with hypertension (n = 42), diabetes mellitus (n = 34), malignancy (n = 4), renal or liver failure (n = 6), rheumatic disease (n = 5), or active infection (n = 6) were excluded. This left 119 patients with KOA. One hundred twenty healthy individuals who underwent a physical examination at our hospital and had normal test indicators were recruited as controls. The study was approved by the EC office of Guangzhou University of Chinese Medicine (2018-67). Written or verbal consent was obtained from all patients.
Laboratory and clinical assessments
We recorded the following patient information: age, sex, cell counts of monocytes, neutrophils, lymphocytes, platelets, white blood cells (WBCs), and red blood cells (RBCs), as well as the RBC distribution width, erythrocyte sedimentation rate (ESR), levels of C-reactive protein (CRP), hemoglobin (HGB), creatinine (CREA), alanine aminotransferase (ALT), and aspartate aminotransferase (AST), and the Kellgren–Lawrence (KL) score. 22 Using these data, we calculated the MLR, NLR, and PLR. We divided the patients into two groups by KL score where grade 0 represents no osteophytes and grade 4 represents large osteophytes: 12 KL1–3, mild/moderate KOA; and KL = 4, severe KOA. All samples were tested by the same instruments at our department. Cell counting was performed using a V253602 automatic five-part hematology analyzer (Mindray Bio-Medical Electronics Company, Shenzhen, China).
Statistical analysis
We conducted data analysis using IBM SPSS Statistics for Windows, Version 19.0 (IBM Corp., Armonk, NY, USA). Continuous data were summarized as means ± standard deviations. Normally distributed data were analyzed as independent samples using the Student’s t test, and data with a skewed distribution were analyzed by Mann–Whitney U tests. Categorical data were summarized as numbers and percentages. Comparisons of categorical variables between two groups were conducted using chi-squared tests. The effects of different variables on KOA were analyzed by univariate and multivariate logistic regression analyses. To test for associations, we used the Pearson correlation, with an ROC curve for sensitivity and specificity. P < 0.05 was considered statistically significant.
Results
Basic characteristics and MLR, NLR, and PLR in patients with KOA
Table 1 shows the main laboratory and clinical characteristics of patients and healthy controls. There was no significant difference in sex or age between the two groups. In the KOA group, the counts of WBCs, neutrophils, and monocytes were higher than in the control group, and the counts of lymphocytes, platelets, HGB, and RBCs were lower. The MLR (0.19 ± 0.11), NLR (2.39 ± 1.73), and PLR (125.85 ± 41.34) were significantly higher in patients with KOA than in healthy controls (0.11 ± 0.03, 1.49 ± 0.40, and 110.61 ± 30.44, respectively; P < 0.001).
Basic characteristics of KOA patients and controls.

males shown as a percentage. KOA, knee osteoarthritis; WBCs, white blood cells; RBCs, red blood cells; RDW-CV, red blood cell distribution width; MLR, monocyte–lymphocyte ratio; NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio.
The MLR has a high diagnostic value for KOA
Using ROC curves, we explored the diagnostic values of the NLR, MLR, and PLR for KOA. The area under the curve (AUC) of the MLR was 0.81 (95% confidence interval [CI]: 0.759–0.865) using the optimal cutoff value of 0.12, with a sensitivity of 84% and a specificity of 66.7%. The AUC of the NLR was 0.77 (95% CI: 0.712–0.831), and that of the PLR was 0.60 (95% CI: 0.528–0.672) (Figure 1). An evaluation of the diagnostic value of MLR combined with NLR (MLR + NLR) gave an AUC of 0.82 (95% CI: 0.770–0.873).
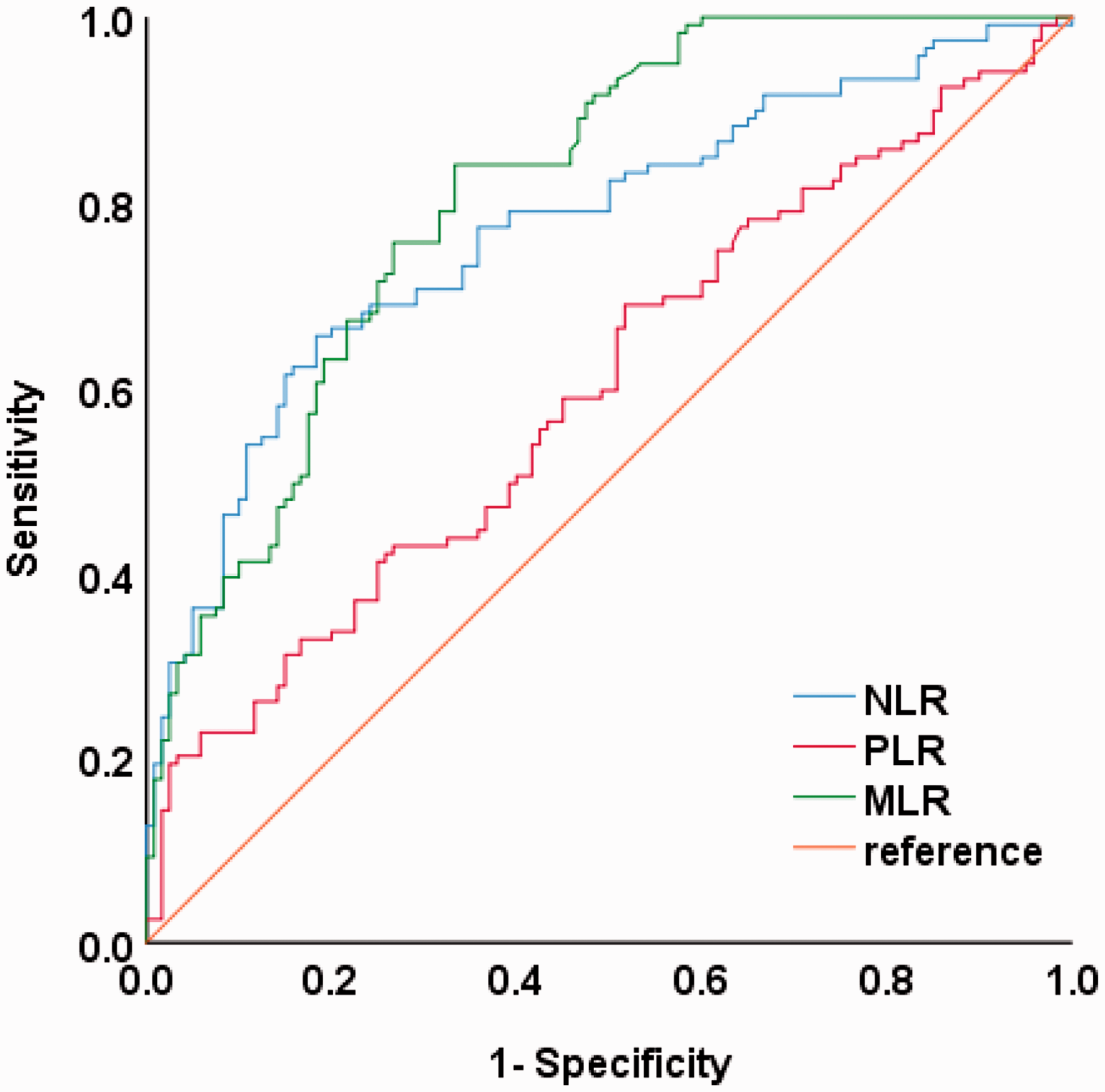
ROC curve of the NLR, MLR, and PLR in KOA. ROC, receiver operating characteristic; NLR, neutrophil–lymphocyte ratio; MLR, monocyte–lymphocyte ratio; PLR, platelet–lymphocyte ratio; KOA, knee osteoarthritis.
Blood MLR as an independent predictor of KOA
Univariate analysis showed that the blood MLR was significantly associated with KOA (P = 0.016). Multiple logistic regression analysis also revealed that the blood MLR was an independent predictor of KOA (P = 0.032; Table 2). Analysis of the correlation between variables showed that MLR was positively correlated with ESR (R = 0.215, P = 0.021) and CRP (R = 0.223, P = 0.017) (Table 3).
Variables associated with OA severity by univariable and multivariable analyses.
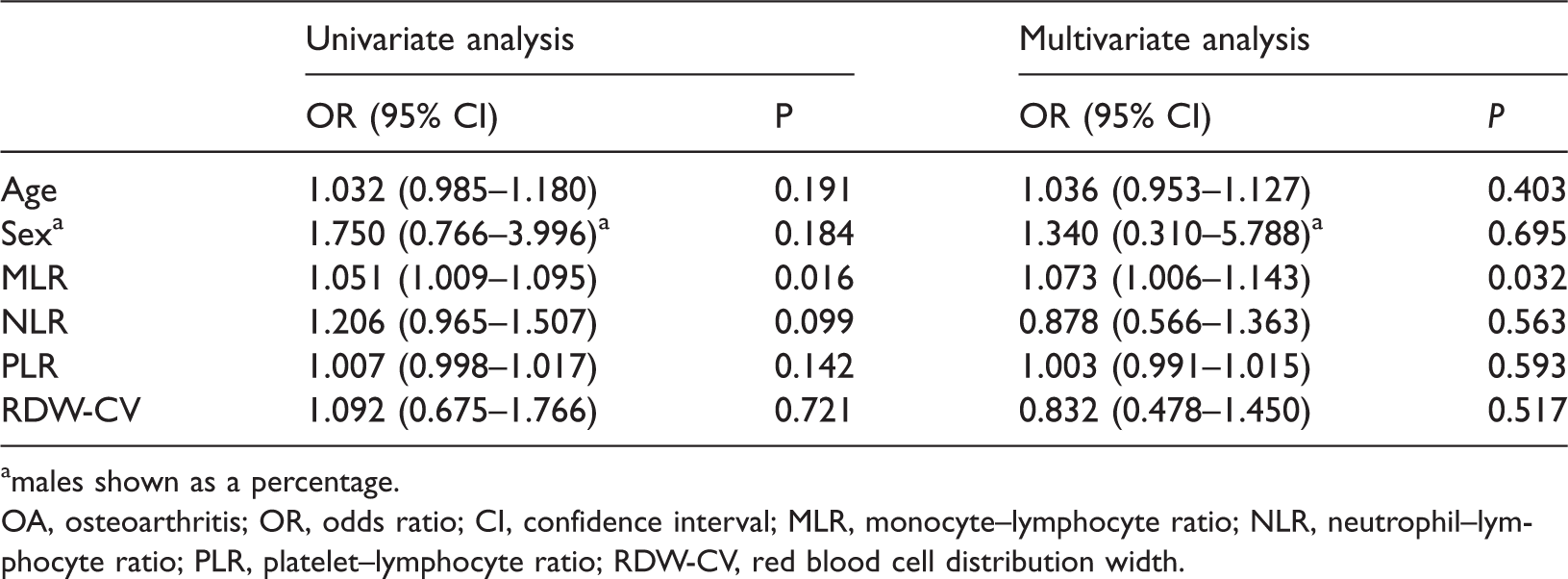
males shown as a percentage.
OA, osteoarthritis; OR, odds ratio; CI, confidence interval; MLR, monocyte–lymphocyte ratio; NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; RDW-CV, red blood cell distribution width.
Analysis of the correlation between variables.

MLR, monocyte–lymphocyte ratio; NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio.
MLR and NLR were higher in KL4 OA patients than KL1–3 OA patients
We graded all patients by KL score, with 33 patients belonging to the KL4 group (severe), and 86 belonging to the KL1–3 group (mild/moderate). The MLR (P < 0.001) and NLR (P = 0.021) were significantly increased in KL4 patients compared with KL1–3 patients. KL4 patients showed the highest monocyte level, while KL1–3 patients had the highest lymphocyte level. However, there were no significant differences in age, sex, RDW, ESR, counts of neutrophils, platelets, or RBCs, or levels of HGB, CRP, ALT, AST, or CREA between the two groups (Table 4).
Characteristic data of KL4 and KL1–3 KOA patients.

males shown as a percentage. KOA, knee osteoarthritis; WBCs, white blood cells; RBCs, red blood cells; RDW-CV, red blood cell distribution width; MLR, monocyte–lymphocyte ratio; NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein hemoglobin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CREA, creatinine.
Discussion
This cross-sectional study evaluated the diagnostic value of MLR, NLR, and PLR, which were previously reported as non-invasive and cost-effective markers in various systemic inflammatory diseases, in KOA patients. We found that KOA patients showed higher MLR, NLR, and PLR than control subjects. The diagnostic ability of the MLR was higher than that of the NLR, PLR, and other parameters. Moreover, the MLR was positively correlated with the ESR and CRP levels. In KOA patient subgroups according to KL score, the MLR was significantly higher in KL4 patients than in KL1–3 patients. Additionally, the MLR was significantly associated with KOA, and emerged as an independent predictor of disease. These findings indicate that the MLR has a high diagnostic value for KOA.
Unlike many other expensive inflammatory markers, the MLR and NLR have the advantage of convenient calculation and low cost. Although research into biomarkers is increasing, with reports on cartilage oligomeric matrix proteins, hyaluronic acid, protein polysaccharides, type II collagen carboxy terminal peptides, and matrix metalloproteinases,11,23,24 there are currently no effective, widely accepted, and clinically applicable biomarkers to predict early-stage KOA. Leukocyte subpopulation tests are commonly used to indicate inflammatory disease, but the NLR, MLR, and PLR have recently been shown to be more appropriate inflammation indicators.8,25,26 Although different physiological conditions such as dehydration may affect the absolute value of each subtype of blood cells, the impact on the NLR, MLR, and PLR is relatively small. 15
In this study, we found that the MLR, NLR, and PLR were all significantly higher in KOA patients than controls. Taşoğlu et al. demonstrated that the blood NLR is a promising, novel marker to indicate the severity of KOA. 16 The MLR reflects the balance between effector and host, and was first defined as a biomarker in infectious disease. 27 In our study, it had a higher diagnostic value than the NLR and PLR according to the ROC curve. Moreover, the AUC value for the MLR combined with the NLR was higher than that for MLR or NLR alone. We observed elevated neutrophil and monocyte counts in KOA patients compared with controls but lymphocyte counts decreased, which is consistent with previous studies.28,29
The mechanism of elevated MLR levels in KOA has not been fully clarified. Lymphocytes and monocytes are key cells for innate and acquired immunity, and the MLR represents the balance of immune disease progression. 21 Therefore, the observed increase in the number of monocytes may be caused by the progression of immunity. Monocytes can be activated by inflammasome-mediated pathways, and chronic low-grade inflammasome activation helps drive OA progression. 14 The observed decrease in the number of lymphocytes may be related to their accumulation at the site of inflammation.17,30 Notably, Du et al. found that the lymphocyte–monocyte ratio was higher in rheumatoid arthritis patients than those with OA and healthy controls. 17
We found a high correlation between the MLR, NLR, and PLR and ESR and CRP, but no link with the patient’s age or sex. This indicates that the MLR and NLR combined with ESR and CRP may increase the reliability of diagnosing KOA. Similarly, Sargin et al. detected a significant correlation between the levels of DAS28‐ESR and NLR and between DAS28‐ESR and PLR before and after treatment of rheumatoid arthritis with rituximab.31 We used the KL grading system as the gold standard to classify radiographic OA with respect to disease severity. The MLR and NLR were higher in the KL4 group than the KL1–3 group, KL4 patients showed the highest monocyte level, and KL1–3 patients had the highest lymphocyte level. This suggested that the MLR and NLR are suitable predictors for KOA severity. Moreover, this finding for the NLR diagnostic value is consistent with that of Taşoğlu’s et al. 16
Some limitations exist in this study. First, it was a single center study with a small number of patients, so more patients from different centers should be evaluated in future studies. Second, we did not investigate the pathogenesis of elevated MLR and NLR. Therefore, longitudinal and molecular biology studies are needed to explore the mechanism of increased MLR and NLR.
In conclusion, the MLR was significantly higher in KOA patients compared with healthy controls, and had a higher diagnostic value than the NLR for KOA. To our knowledge, this is the first study to identify the MLR as a novel and reliable potential predictor for KOA.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the NSFC-Guangdong Joint Foundation of China (No. U1601225), the Key Scientific and Technological Program of Guangzhou City (No. 201607020016) and the Sanming Project of Medicine in Shenzhen (No. SZSM201812066).
