Abstract
Objective
We investigated the risk of acute and late toxicities of concurrent chemoradiotherapy (CCRT) and radiotherapy alone in patients with nasopharynx carcinoma (NPC).
Methods
In this meta-analysis, we searched the PubMed, Embase, Cochrane Library, and Web of Science databases for eligible randomized clinical trials (RCTs). In addition to the incidence of specific toxicities, risk ratios (RRs) or odd ratios (ORs) and 95% confidence intervals (CIs) were obtained using fixed-effect or random-effects models.
Results
In total, 11 RCTs involving 2801 patients with NPC were included in this analysis. For grade ≥3 adverse events, patients who received CCRT treatment had a higher proportion of acute mucositis (39.9% vs. 30.5%, RR=1.30, 95%CI, 1.16–1.46) acute nausea and vomiting (RR=6.26, 95% CI: 2.01–19.45), and neutropenia (RR=30.86, 95% CI: 7.36 to 129.35). For late severe toxicities, CCRT treatment was significantly associated with higher incidence of hearing loss (116.56% vs. 411.43%, RR=1.461, 95%CI, 1.043–21.095). The incidence of acute nausea and vomiting was more frequent in patients receiving CCRT.
Conclusion
Compared with radiotherapy alone, CCRT increases the risk of severe acute toxicities (mucositis, nausea/vomiting, and neutropenia) and severe late toxicity (hearing loss) in patients with NPC. However, larger studies are needed to confirm this finding.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a rare type of malignant carcinoma that has a specific geographical distribution and a high risk of distant metastases. In Southeast Asia, the annual incidence rate ranges from 0.15% to 0.5%. 1 Radiotherapy is the cornerstone of initial treatment due to the radio-sensitivity of NPC and its deep-seated location. However, although radiotherapy alone is an efficacious treatment option for early-stage NPC, the prognosis of patients with locally advanced NPC remains unsatisfactory.2,3 Distant metastasis is the main cause of treatment failure in regionally advanced NPC.4,5 Since the publication of the findings of the 0099 (INT-0099) trial, 6 concurrent chemo-radiotherapy (CCRT) has been the standard therapy for locally advanced NPC,7,8 and further clinical trials have demonstrated that the 5-year overall survival (OS) rate for patients receiving CCRT is 11.7% higher than that for patients receiving radiotherapy alone. 9 Recurrence-free survival and metastasis-free survival have also been shown to be improved in patients with NPC receiving CCRT. 10 In addition, several other studies and meta-analyses have confirmed that CCRT confers additional benefits compared with radiotherapy alone.11,12
Equally important is the frequency and type of adverse events associated with combination treatment of concurrent chemotherapy and radiotherapy. Severe toxicities can impair treatment compliance, diminish patient quality of life, and even be life-threatening. During radiotherapy, up to 95% of patients with NPC showed various levels of dosage-dependent acute dermatitis, contributing to increased risk of infection and delayed treatment. Furthermore, the combined activity of anti-neoplastic drugs and radiographic exposure may induce injuries to the oral mucosa and apoptosis of oral epithelial cells approximately 6–15 days after treatment initiation. 13 Although several studies have investigated the toxicities of CCRT and radiotherapy,3,14–16 no study to date has compared these treatment modalities across almost all categories of acute and late severe (grade 3/4) adverse events in a large patient population.
The objective of the present meta-analysis, which includes almost all eligible randomized controlled trials (RCTs), was to comprehensively compare the incidence and risk of acute and late severe toxicities between CCRT and radiotherapy alone in a large sample size and to provide clinical evidence for the need for increased attention to toxicities in patients with NPC.
Methods and materials
Identification of studies
This meta-analysis was implemented on the basis of the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) statement. 17 Relevant studies were identified by searching the PubMed, Embase, Cochrane Library, and Web of Science databases to a cut-off date of September 30, 2016, regardless of language or publication status. The search was conducted using the keywords “nasopharynx cancer or nasopharyngeal carcinoma or nasopharyngeal neoplasms”, “radiochemotherapy or chemoradiotherapy or chemoradiation or chemo-radiotherapy” and “toxicities or adverse event or side effect or adverse reaction or safety issue,” and was limited to human studies and clinical trials. The reference lists of relevant original and review articles were also manually searched to identify additional studies.
Study inclusion and exclusion criteria
Only RCTs in which CCRT was compared with radiotherapy alone in patients with NPC and in which detailed data on acute or late toxicities were reported were eligible for inclusion in the meta-analysis. Studies meeting the following additional standards were included: (1) endpoints regarding acute toxicities of CCRT and radiotherapy covering at least one of dysphagia, dermatitis, hearing loss, peripheral neuropathy, constipation, mucositis, and nausea and vomiting; (2) endpoints concerning late toxicities including at least one of xerostomia, dysphagia, mucositis, nasopharyngeal mucosal necrosis, otitis, hearing loss, brachial plexopathy, bone necrosis, temporal lobe necrosis, symptomatic brain damage, radiation-induced brain damage, radiation encephalopathy, endocrine dysfunction, visual toxicity, radiation-induced malignancy, secondary tumor, subcutaneous fibrosis, torrential bleeding, trismus, and spinal damage. Additionally, studies were excluded if they (1) were case reports, meeting abstracts, or editorial materials; or (2) were duplicated, incomplete, or outside of the range of the research topic.
Data extraction
Two reviewers independently extracted the data on study characteristics of CCRT and radiotherapy groups (e.g. sample size, ethnicity, and mean age of the study subjects), publication year, treatment arms, number of patients who suffered from acute or late toxicities, duration of follow up, and counts of acute and late toxicities. Where there was disagreement between the two reviewers, the opinion of a third reviewer was obtained.
Statistical analysis
The software R 3.3.1 (www.r-project.org) including the meta package was used for direct meta-analysis. 18 First, a frequency table was generated to describe each toxicity item based on counts of toxic events or number of subjects with reported toxic events. The distinctions of dichotomous variables were statistically evaluated with the use of the risk ratio (RR) according to event frequency or odds ratio (OR) according to subject frequency with a two-tailed 95% confidence interval (CI).One fixed-effect model was established for consistent studies while a random-effects model was fitted for studies with heterogeneous results. Two-tailed P values <0.05 were considered statistically significant. Heterogeneity between the studies was evaluated by means of I2 (if I2>50%) 19 and the Q test of Cochran (if P<0.10). 20 Both indices assess the percentage of variability across studies that is attributable to heterogeneity rather than to chance.
Results
Search results and study characteristics
Eleven eligible RCTs6,10,21–29 involving 2801 patients were included in the meta-analysis after excluding reviews, irrelevant studies, and duplicates. Figure 1 shows the details of the study selection process. Among the included studies, only two used intensity-modulated radiation therapy, while conventional irradiation was used in the remaining studies. The main characteristics of the included studies are presented in Table 1. Patients were recruited between 1989 and 2008, and the median follow-up period was 35 months–7.3 years. The mean age of patients in the CCRT group in all studies was 42–50 years, while patients in the radiotherapy group were aged 44.3–52 years. The CCRT group received treatment including cisplatin, cisplatin complexes, 5-fluorouracil, oxaliplatin, and carboplatin, although the administration duration and dosage varied among the included studies. The prescribed dose among patients in the radiotherapy group was generally in the range of 60–70 Gy. Publication bias was not assessed in this meta-analysis as there were as many as ten toxicity endpoints with varying sample sizes included. The selected studies all featured randomization to treatment arm, indicating that the data were likely to be robust (Table 2).

PRISMA flow chart.
Characteristics of randomized clinical trials included in the meta-analysis.

Risk for bias assessment in selected randomized controlled trials

Incidence and risk of specific acute toxicities
For grade ≥3 toxicities, CCRT was significantly associated with higher incidence of mucositis (36.6% vs. 29%), nausea and vomiting (14.6% vs. 0.2%), and neutropenia (22.8% vs. 0.3%). According to subject frequency, all patients experienced any grade toxicities, and more patients experienced grade ≥3 nausea and vomiting in the CCRT group. In the radiotherapy alone group, no patient experienced grade ≥3 nausea and vomiting (Table 3).
Summary of acute toxicities (grade 3/4 and any grade)

As shown in Figure 2, CCRT significantly increased the risk of acute mucositis, whether assessed by event frequency (RR=1.26, 95% CI: 1.10–1.44, P<0.001) or subject frequency (RR=1.62, 95% CI: 1.05–2.50, P=0.029). Furthermore, CCRT significantly increased the risk of acute nausea/vomiting (RR=31.28, 95% CI: 7.70–127.06, P<0.001) and acute neutropenia (RR=55.85, 95% CI: 11.20–278.58, P<0.001) when compared with radiotherapy alone. No significant differences were observed between the groups in the risk of acute dermatitis and acute hepato-renal dysfunctions.

Forest plot comparing acute toxicities between CCRT and radiotherapy alone according to event frequency of grade 3/4 mucositis (a); subject frequency of grade 3/4 mucositis (b); event frequency of grade 3/4 nausea and vomiting (c); and event frequency of grade 3/4 neutropenia (d). Size of the data square marker indicates the weight of each study in this analysis. The diamond represents the overall estimated effects in the model. CCRT: concurrent chemoradiotherapy; CI: confidence interval; OR: odds ratio; RR: risk ratio.
Incidence and risk of specific late toxicities
For late grade ≥3 toxicities, the event frequency of hearing loss (20.6% vs. 13.5%), mucositis (45.4% vs. 31.0%), and neutropenia (13.5% vs. 0%) was significantly higher in the CCRT group (Table 4). Our meta-analysis showed that CCRT was significantly associated with elevated risk of late severe hearing loss (RR=1.52, 95% CI: 1.03–2.25, P=0.037) compared with radiotherapy (Figure 3). For grade ≥3 xerostomia, the incidence in the CCRT group and the radiotherapy alone group was 5.8% and 2.4%, respectively, and the difference between the groups was not significant.
Summary of late toxicities (grade 3/4 and any grade)

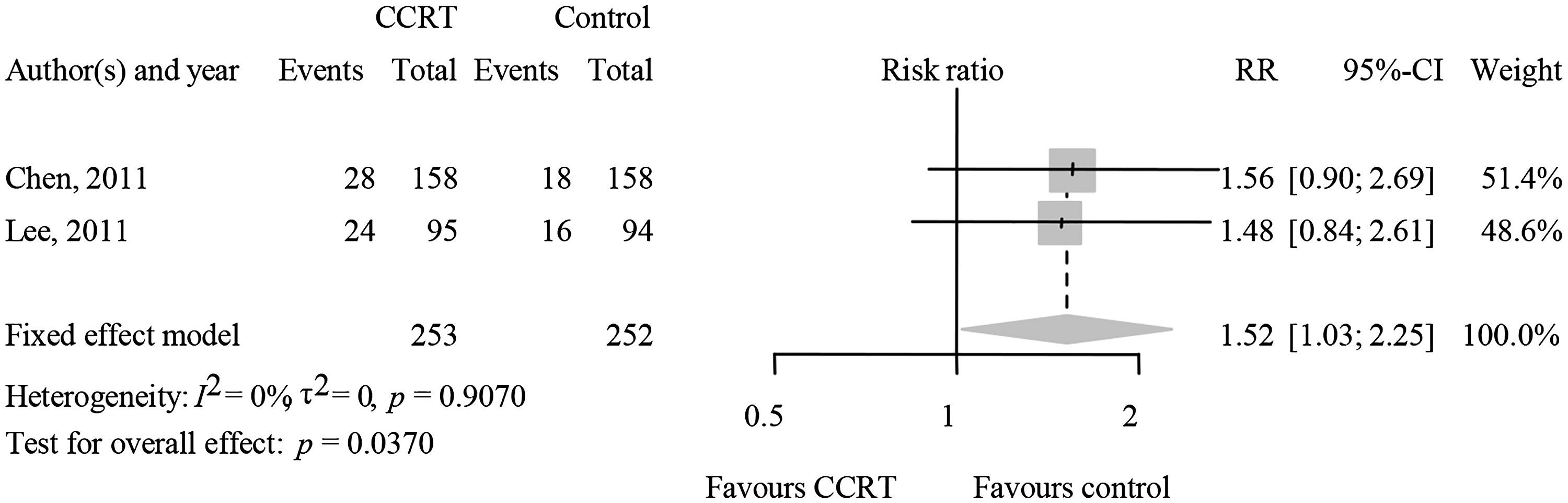
Forest plot comparing late toxicities between CCRT and radiotherapy alone by event frequency of late grade 3/4 hearing loss. Size of the data square marker indicates the weight of each study in this analysis. The diamond represents the overall estimated effects in the model. CCRT: concurrent chemoradiotherapy; CI: confidence interval; RR: risk ratio.
Discussion
To the best of our knowledge, this is the first meta-analysis that comprehensively compares the incidence and risk of severe acute and late toxicities associated with CCRT versus radiotherapy alone in a large population of patients with NPC. This meta-analysis included 11 RCTs, with 2801 patients in total, to compare the toxicities associated with these two treatment modalities. By evaluating the incidence of five subtypes of acute toxicity (mucositis, nausea and vomiting, neutropenia, dermatitis, and hepato-renal function) and four subtypes of late toxicity (cranial neuropathy, hearing loss, peripheral neuropathy, and fibrosis of the neck), we showed that CCRT was associated with more severe acute mucositis, acute nausea/vomiting, acute neutropenia, and late hearing loss than radiotherapy alone. Among the enrolled studies, the median follow-up varied from 35 months to 7.3 years, durations which were appropriate for the analysis of late toxicities. In the present study, toxicities were the primary endpoint, and data on acute, late, and severe toxicities associated with CCRT and radiotherapy were recorded and analyzed to produce a comprehensive analysis of the types and severity of toxicities associated with CCRT and radiotherapy.
For acute toxicities, including grade 3/4 acute toxicities, this meta-analysis showed that CCRT was associated with higher risk of acute mucositis and nausea compared with radiotherapy, consistent with previous reports. 32 The duration and severity of mucositis caused by radiotherapy have been closely correlated with the integrated action of accumulated dose, radioactive source, irradiation volume of mucosa, dose intensity, and xerostomia.31,32 Endothelial tissues and epithelium of connective tissues are damaged during radiotherapy, and the release of inflammatory factors such as prostaglandins, tumor necrosis factor, and interleukin-1 can aggravate tissue injury. 33 Somewhat distinct from radiotherapy, mucositis induced by chemotherapy is mainly attributable to the route of administration, dose intensity, duration of therapy, and chemotherapeutic drug used. Normal DNA synthesis can be affected by chemotherapy, and the repeated and continuous usage of low-dose cytotoxic agents can further exacerbate the risk of mucositis. 34 Therefore, CCRT appeared to promote the development of oral mucositis to a greater extent than radiotherapy alone. In clinical practice, anti-neoplastic drugs are typically administered 1 week after initiation of radiotherapy, leading to further injury to the oral mucous epithelium and associated formation of anabrosis. 35
For late toxicities, our meta-analysis showed that CCRT was significantly associated with late grade 3/4 hearing loss, consistent with previous reports.15,36 The inclusion of studies that used discrepant chemotherapeutic regimens for treating patients with NPC might have affected the credibility of this association. Thus, the meta-analysis considered only the general outcomes of chemotherapy and the findings may therefore be inapplicable to specific medications. Any one of these chemotherapeutic studies may inevitably have over- or underestimated the association between CCRT and risk of late hearing loss.
Two of the included RCTs reported cases of bone necrosis when comparing CCRT with radiotherapy (1:0 4 and 1:1 7 ). No further evaluation of this finding was carried out because of the small number of RCTs included. Furthermore, a study by Lee et al. 37 in 2013 found that concurrent chemotherapy was not associated with a significant increase in the risk of bone necrosis. Future studies in larger populations are needed to further explore these observations.
This study was limited by a lack of heterogeneity as all included studies enrolled only subjects of Chinese Han ethnicity, meaning that it may be difficult to generalize the results to other populations. In addition, there might be potential mutations that could affect the efficacy of CCRT. For instance, it was reported that non-small-cell lung cancer (NSCLC) patients who carried epidermal growth factor receptor (EGFR) mutations (e.g. exon 19 deletions and the L858R point mutation) could achieve better efficacy after treatment of gefitinib than those with normal EGFR genotypes.38,39 As a result, further molecular studies should be explored to discover potential mutations that might influence NPC patients’ responses to different drugs.
There is increasing evidence that, compared with radiotherapy alone, CCRT may improve survival among patients with locally advanced NPC, and CCRT is thus commonly used in clinical practice. For patients at high risk of developing severe toxicities associated with CCRT such as mucositis, nausea and vomiting, and hearing loss should therefore be followed more closely when receiving CCRT.
Conclusions
The current study is the first meta-analysis to comprehensively and intensively analyze and compare acute and late toxicities between CCRT and radiotherapy alone. The findings indicate that CCRT is more likely to induce certain acute severe toxicities (e.g. acute mucositis and acute nausea and vomiting) and late severe toxicity (i.e. hearing loss) than radiotherapy. However, larger studies in more diverse populations are required to stratify patients with NPC by disease grade and specific mutations, and to subsequently compare the incidences of acute and late toxicities after treatment with CCRT or radiotherapy alone.
Footnotes
List of abbreviations
CCRT: concurrent chemoradiotherapy; CI: confidence interval; EGFR: epidermal growth factor receptor; NPC: nasopharynx carcinoma; NSCLC: non-small-cell lung cancer; OR: odd ratio; OS: overall survival; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-analysis; RCT: randomized clinical trial; RR: risk ratio
Acknowledgements
The authors wish to thank Professor Ming Ye from the radiotherapy department of Renji Hospital, affiliated to Shanghai Jiaotong University. We also thank May Dong, Iris Sun, Sam Zhong, and Cuiling Yin for their assistance.
Availability of data and material
The data extracted and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethical approval
This article does not contain data from any studies with human participants or animals performed by any of the authors. As the present study was a meta-analysis of published literatures, the study was exempt from Ethics Committee approval.
Funding
This study was funded by Shanghai Jiading District Science & Technology Commission (KWKYXM, 2014-KW-01) and the Seed Fund (Natural Science Class) of Shanghai University of Medicine & Health Sciences (no. HMSF-17-21-026).
