Abstract
Objectives
This study aimed to determine leptin levels in term newborns who were born in the north of Jordan. We also aimed to investigate the relationships of leptin levels with fetal growth parameters, and to assess the difference in leptin levels according to sex and gestational age.
Methods
A cross-sectional descriptive study that involved 170 term newborns was conducted. A working sheet for data collection was created for each newborn and included sex, weight, length, head circumference, gestational age, and Apgar score. Blood samples were obtained from the umbilical cord vein of newborns after delivery to measure serum leptin levels. Data are shown as frequency, percentages, means, and standard deviations.
Results
We found that the mean leptin level was 1.17 ± 0.48 ng/mL. The independent t-test showed that the mean leptin level in boys (0.93 ± 0. 34 ng/mL) was significantly lower than that in girls (1.38 ± 0.47 ng/mL). Pearson’s correlations showed that leptin levels of newborns were positively and significantly correlated with weight, length, and head circumference.
Conclusion
In Jordanian healthy term newborns, leptin levels correlate with sex and intrauterine growth parameters.
Introduction
Leptin is considered an anorexigenic hormone that has an important function in regulating energy by lowering food intake and increasing consumption of energy.1–4 Adipose tissue is the main site of leptin production and to some extent it is produced by the placenta, stomach, and mammary epithelium.5–7 Leptin plays an important role in development of a neonate’s hypothalamic pathways and is involved in controlling energy and appetite. 8 Evidence from animal studies has suggested that leptin can act on the brain to improve formation of the neural circuits regulating future food intake and adiposity. 9 Many studies have focused on adult leptin levels and their correlation with body mass index, but limited studies have investigated newborn leptin levels and their correlation with intrauterine growth. 10
In a cross-sectional study 10 on cord blood samples that were taken from 50 newborns, cord blood leptin levels were correlated with fetal growth. Recently, Ökdemir et al. 11 conducted a study on 84 mothers and their newborns to examine the relationships between leptin levels and maternal weight gain and newborn anthropometric measurements. These authors showed that in newborns, leptin levels significantly varied according to birth weight (P = 0.013). Furthermore, leptin levels were associated with fetal adiposity. Schuster et al. 12 conducted a prospective, longitudinal study to evaluate leptin levels in the blood of 23 healthy lactating mothers and in their breast milk, as well as in their neonates. These authors showed that serum leptin levels were much higher than those in breast milk.
A review of the relevant literature in Jordan showed some studies on leptin among adults, but no studies on leptin in newborns. Abu-Samak et al. 13 conducted a study to analyze the effect of hyperleptinemia on fasting lipid and hematological parameters in healthy young men in Jordan. These authors found that mean serum leptin levels were higher in those with hyperlipidemia than in those without hyperlipidemia.
The current study aimed to determine newborn leptin levels in the north of Jordan, to investigate the relationships between newborn leptin levels and selected growth parameters, such as weight, length, and head circumference, and to assess the differences in leptin levels according to sex and gestational age.
Methods
Study design
A descriptive-cross-sectional design was used to assess the relationship between newborn leptin levels and selected growth parameters, and to assess the differences in leptin levels according to sex and gestational age.
Samples and sampling technique
A convenient sampling technique was used to recruit a convenience sample of 170 term newborns whose mothers agreed to participate in this study. Using a convenience sample may affect the external validity of the results because it is unlikely to be representative of the population under study. The eligibility criteria for the participants were as follows: term newborns of mothers who had no medical illnesses during their pregnancies, and mothers were aged older than 18 years old and agreed to include their newborns in the current study. Newborns of mothers with medical illness or those who were delivered preterm were excluded.
The sample size was calculated on the basis of parameters of G* power software (free software). 14 The sample size required was 138 participants on the basis of the following statistical factors for Pearson’s r: an alpha level of 0.05, a power level 0.95, and an effect size of 0.30. To overcome the problem of attrition or missing data, an extra 32 term newborns were included in the final sample size to have at least 170 participants.
Site and setting of the study
This study was conducted in the University Educational Hospital in Irbid Province, which has the second largest metropolitan population in Jordan after Amman. The total capacity of this hospital is more than 650 beds with an impressive occupancy rate of close to 99%. This hospital includes different specialties, such as internal medicine, cardiology, orthopedics, pediatric neurology, maternity, and dermatology.
Study procedure
The study method and protocol were approved by the Institutional Review Board of the University Hospital. Written informed consent was obtained from the newborns’ mothers, who agreed to include their newborns in the study. The confidentiality of the participants was protected by providing a code number for each participant during the data collection and analysis phases of the study. No physical, psychological, social, or economic harm or risk affected the newborn. There were no complications in the blood sample collection process for the newborns. No blood loss, infections, or complications developed during or after the procedure. A qualified competent staff nurse was responsible for obtaining blood samples from the umbilical cord of the newborns. This procedure was performed under a hospital policy of withdrawing blood under an aseptic technique. A blood sample was taken as a routine check for each newborn in the period immediately after delivery.
Files of the newborns and mothers were carefully checked to ensure their eligibility to participate in the study. The primary researcher collected data regarding sex, weight, length, gestational age, head circumference, and Apgar score. To measure serum leptin levels, blood samples were obtained from the umbilical cord vein of the newborn after delivery.
Serum leptin measurement
Approximately 5 mL of venous blood was collected from each newborn in a serum separator tube. The samples were allowed to clot for 2 hours at room temperature or overnight at 4°C before being centrifuged for 15 minutes at 1000 × g. The serum was then removed and assayed immediately or divided into aliquots, and the samples were stored at −80°C.
Quantitative measurement of serum leptin levels was performed using a leptin enzyme immunoassay or Human Leptin ELISA kit (MBS700713; MyBioSource, San Diego, CA, USA) according to the manufacturer’s instructions. This assay uses the quantitative sandwich enzyme immunoassay technique. Antibody specific for leptin was pre-coated onto a microplate. Standards and samples were then pipetted into the wells and any leptin present was bound by the immobilized antibody. After removing any unbound substances, a biotin-conjugated antibody, specific for leptin, was added to the wells. After washing, avidin-conjugated horseradish peroxidase was added to the wells. Following a wash to remove any unbound avidin–enzyme reagent, a substrate solution was added to the wells and color developed in proportion to the amount of leptin that was bound in the initial step. Color development was stopped and the intensity of the color was then measured.
Statistical analysis
Descriptive statistics, including frequency, percentage, mean, and standard deviation, are used to describe the data. The relationships between leptin levels and newborns’ growth parameters (weight, length, and head circumference) were tested using Pearson’s product-moment correlations. The independent t-test was also used to examine differences in leptin levels according to sex and gestational weeks. Significance was considered at P < 0.05.
Results
General characteristics of newborns
The newborns’ characteristics are shown in Table 1. The mean leptin level was 1.17 ± 0.48 ng/mL, the mean birth weight was 3.11 ± 0.52 kg., and the mean length was 49.71 ± 2.20 cm. The mean gestational age was 38.28 ± 1.36 weeks, and it ranged from 37 to 42 weeks. More than half of the newborns were girls (n = 92).
Sample characteristics of the newborns (n = 170).

SD standard deviation.
Effect of sex and gestational age on newborn leptin levels
The mean leptin level in boys was significantly lower than that in girls (t[168] = 2.62, P = 0.010, Table 2). Furthermore, the difference between the mean leptin level in newborns delivered at 37 to 38 weeks’ gestational age and the second group of newborns delivered at >38 to 42 weeks’ gestational age was not significant (t[168] =−0.39).
Effect of sex and gestational age on newborn leptin levels (n = 170).
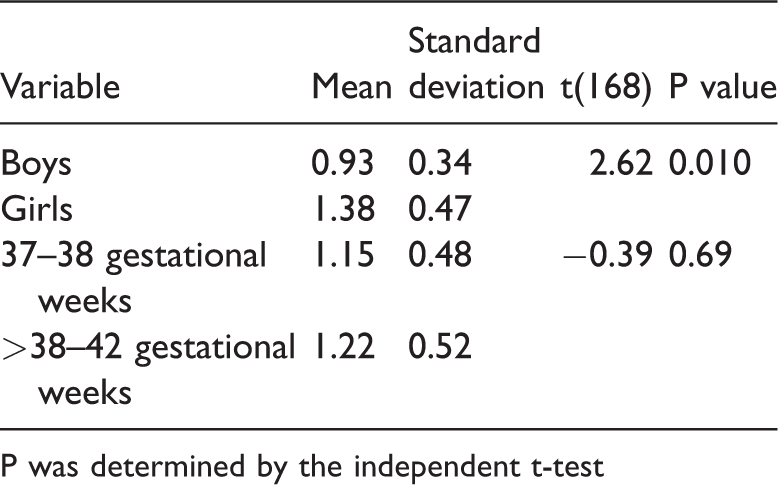
P was determined by the independent t-test
Relationship between newborn leptin levels and growth parameters
There was a significant, but weak, positive correlation between leptin levels and newborn weight (r[168] = 0.16, P = 0.044). Furthermore, Pearson’s correlation also showed that there was a significant, but weak, positive correlation between leptin levels and newborn length (r[168] = 0.15, P = 0.049). Moreover, there was a significant, but weak, positive correlation between leptin levels and newborn head circumference (r[168] = 0.16, P = 0.032). However, there were no significant correlations between the Apgar score at 1 minute (r[168] = −0.02) and the Apgar score at 5 minutes (r[168] = −0.07) and leptin levels (Table 3).
Pearson’s correlations between newborn leptin levels and their growth parameters (n = 170).
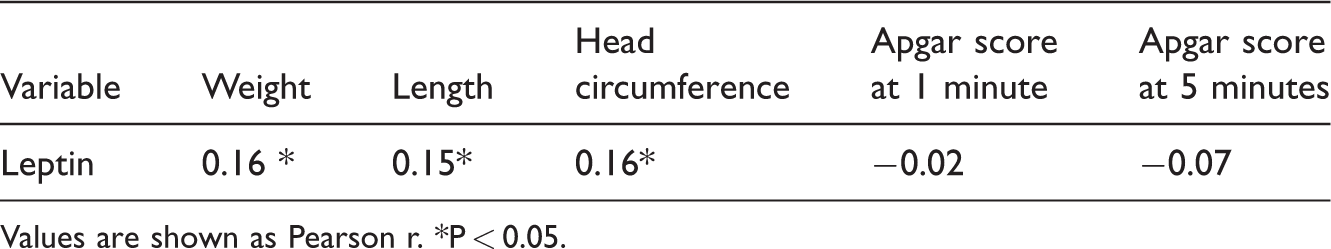
Values are shown as Pearson r. *P < 0.05.
Discussion
The present study showed that the mean leptin level in Jordanian newborns was 1.17 ± 0.48 ng/mL. In Ohkawa et al.’s study, 15 serum leptin levels of newborns were less (approximately 0.8 ng/mL) than those in our study. Our study showed that female newborns had higher leptin levels compared with male newborns. This finding is generally consistent with the results obtained by Torgerson et al., 16 who reported that women had significantly higher levels of leptin than did men (P < 0.05). Some studies reported differences in leptin levels between males and females that were attributed to differences in body composition, to variations in hormones, and to genes responsible for sex specifications. 17 Other studies have reported that sex hormones have different effects on leptin levels; testosterone has an inhibitory effect on leptin, whereas estrogen has a stimulatory effect. 18 However, a prospective cohort study 19 of 100 women who had full term newborns showed no significant difference in leptin levels between male and female newborns.
Our study showed positive correlations between newborn leptin levels and weight, length, and head circumference. These findings are consistent with the results of other studies as follows. Koistinen et al. 10 reported a positive correlation between leptin levels and fetal growth. Mantzoros et al. 20 found that leptin was a predictor of weight gain. Savino et al. 21 reported a relationship between serum leptin levels and body mass index (P = 0.042). However, Karakosta et al. 22 found a negative association of blood leptin levels with growth in early childhood. Additionally, another study conducted by Tung et al. (2009) 23 assessed the relationship between venous umbilical cord leptin levels and anthropometric measurements in term healthy Taiwanese newborns. These authors found that birth weight was the only predictor of leptin levels.
There was no association between newborn leptin levels and their Apgar scores in our study. This finding is not in agreement with that of Taghreed et al. 24 who reported a positive correlation between the Apgar score and leptin levels. However, these authors found growth-restricted fetuses had lower blood leptin levels compared with controls.
Conclusions
We conclude that leptin is present in utero and it correlates with intrauterine growth parameters in newborns in Jordan. This finding confirms the results of other studies about the possible role of leptin in fetal growth.
Study recommendations and implications
A larger, randomly selected, and heterogeneous sample of participants from north, central, and south Jordan is required in the future to make our findings more widespread and applicable. This research can be considered a baseline for conducting other research projects that aim to assess changes in leptin levels in patients with comorbid diseases, such as diabetes and obesity. Furthermore, longitudinal research studies need to be conducted to assess the changes in leptin levels over time and their relationship with different physiological parameters.
This study provides better understanding of fetal growth and different factors that participate in growth. Future studies may implement multiple factors to predict newborns that are growth restricted rather than being small for gestational age.
Study limitations
This study has some limitations. Restriction of the study to the north of Jordan and use of a convenience sample of newborns could pose a problem for its generalizability, and affect the external validity of the results. Therefore, we recommend that further studies be conducted, using a larger more heterogeneous sample, thereby enhancing the generalizability of the findings.
Footnotes
Acknowledgements
The authors would like to thank the study participants and Jordan University of Science and Technology.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
