Abstract
Background
Ferrosoferric oxide (Fe3O4) nanoparticles are a commonly used magnetic resonance imaging (MRI) reagent. Luteinizing hormone-releasing hormone (LHRH) is highly expressed on the surfaces of tumors, but its expression is low or absent in the corresponding normal tissues, allowing it to be used for targeted imaging and treatment.
Methods
We prepared Fe3O4 nanoparticles using a chemical co-precipitation method, performed coupling with chitosan to prepare LHRH-Fe3O4 nanoparticles, and explored the application value of LHRH-Fe3O4 nanoparticles in targeted imaging and treatment of breast tumors through
Results
The particle size of the LHRH-Fe3O4 nanoparticles was 10 nm, and they could be taken in by human MCF-7 breast cancer cells. The nanomaterial had low cytotoxicity.
Conclusion
LHRH-Fe3O4 nanoparticles serve the purpose of targeting contrast agents to target sites and are expected to be used for targeted imaging and treatment of cancers with high LHRH expression.
Keywords
Introduction
Malignant tumors are a serious threat to human life and health. Their incidence has been increasing on an annual basis and their mortality is surpassing that of cardiovascular and cerebrovascular diseases, making them the leading threat to human health. Molecular imaging based on molecular biology, imaging science, and other subjects is expected to reveal tiny tumor lesions at the molecular level, facilitating early diagnosis of tumors. However, although magnetic resonance imaging (MRI) is a major molecular imaging examination, contrast agents are susceptible to inducing allergic reactions and are phagocytosed by macrophages; additionally, their surfaces are not easily modified, potentially resulting in insufficient targeting. Thus, the application of contrast agents affects the MRI-based diagnosis of tumors. A main focus in the field of MRI is the development of highly specific targeted contrast agents to increase the detection rate of tiny tumor lesions. Ideal targeted contrast agents should have the following characteristics1–3: simple preparation, low cost, a small particle size that allows easy access to target areas, high specificity and affinity for receptors, no toxic adverse effects, and the ability to reach an adequate concentration in the target area to obtain a good imaging effect.
Ferrosoferric oxide (Fe3O4) nanoparticles are positively charged. Their magnetic property is relatively strong, and their preparation is relatively simple. After they enter the body, they can be recognized by immunoglobulins in the blood and then phagocytosed by the reticuloendothelial system. Finally, they are excreted through the kidneys and liver. Compared with other MRI reagents, Fe3O4 nanoparticles are excellent MRI T2 contrast agents5,6 because of their good biocompatibility and magnetic resonance signal sensitivity.
4
As early as 1996, the US Food and Drug Administration had already approved ferumoxsil and ferumoxide, two magnetic iron oxide nanomaterial drugs. These nanoparticles are easily agglomerated within the body and cleared by the reticuloendothelial system because of their surface hydrophobicity and large surface ratio. Therefore, the surfaces of these nanoparticles must be modified to increase their hydrophilicity and prolong their circulation half-life. Polysaccharides such as chitosan and heparin are used as surface modification materials and can increase the entrapment efficiency and load capacity of the drugs; additionally, they can prolong the
Luteinizing hormone-releasing hormone (LHRH), also known as gonadotropin-releasing hormone, is a decapeptide hormone synthesized and secreted by hypothalamic nerve cells. LHRH mainly regulates the generation and release of luteinizing hormone and follicle-stimulating hormone. Current research shows that LHRH is highly expressed on the surfaces of liver, colon, cervical, and ovarian tumors but that its expression is extremely low or absent in the corresponding normal tissues.
13
Therefore, the LHRH receptor can be used as a tumor-specific target for targeted imaging and treatment. In the present study, we prepared Fe3O4 nanoparticles using a chemical co-precipitation method coupled with chitosan to prepare LHRH-Fe3O4 nanoparticles. The study was performed to explore the application value of LHRH-Fe3O4 nanoparticles in targeted imaging and treatment for breast tumors through
Materials and methods
Preparation of LHRH-Fe3O4 nanoparticles
Fe3O4 nanoparticles were prepared by a chemical co-precipitation method, and LHRH-Fe3O4 nanoparticles were prepared by adding LHRH coupled with chitosan. A 5-mL LHRH-Fe3O4 suspension was centrifuged at low speed for 20 minutes at 12,000 rpm, and the concentration of iron in the supernatant and precipitate was measured by the O-phenanthroline method. The number of LHRH molecules coupled to the surface of chitosan-nanoiron was measured using a bicinchoninic acid protein assay kit. The above LHRH-Fe3O4 suspension was placed in a dialysis bag and dialyzed with 5% dextrose solution for 48 hours. The dialysate was changed every 8 hours. The free LHRH and chitosan were dialyzed, and the dialyzed LHRH-Fe3O4 solution was diluted with distilled water (1:4). One drop was placed on a coated copper mesh, the excess liquid was removed with filter paper, and the mesh was dipped in 2% tungstic acid for 30 s and then dried. The particle size of LHRH-Fe3O4 was then measured by transmission electron microscopy (TEM). After 3 to 5 mL of the solution to be measured was ultrasonically and evenly dispersed and stabilized for 5 minutes, the particle size and size distribution of the prepared LHRH-Fe3O4 solution were measured using a laser particle analyzer (Brookhaven Instruments Corporation, Holtsville, NY, USA). The materials required for preparation of LHRH-Fe3O4 nanoparticles were supplied by Shanghai Jianglai Biotechnology Co., Ltd. (Shanghai, China).
Uptake of LHRH-Fe3O4 nanoparticles by tumor cells
Human MCF-7 breast cancer cells in the logarithmic growth phase were digested by trypsin and inoculated into a six-well plate. When the cells reached 70% to 90% confluence, the supernatant was discarded and LHRH-Fe3O4 or Fe3O4 was added and cultured for 1, 2, 4, and 8 hours. After harvesting and centrifugation, the cells were dissolved in 100 µL of 65% nitric acid at 95°C for 10 minutes. After the addition of 450 µL of water, the iron concentration of the samples was determined by atomic emission spectrometry as previously described. 14 The measured iron concentrations were normalized to the cell number of the samples.
Detection of cytotoxicity by cell counting kit 15
The cells in the logarithmic growth phase were adjusted to a concentration of 1 × 105/mL and then inoculated into a 96-well plate. After the cells were attached, LHRH-Fe3O4 or Fe3O4 was added to the cells. Three replicates were set in each group. The optical density (OD) at 570 nm in each well was measured by the microplate reader. The growth inhibition rate of the cells was calculated with the following formula: growth inhibition rate (%) = mean OD570 in experimental group/mean OD570 in control group × 100%.
Tissue distribution and MRI targeted imaging of LHRH-Fe3O4 in tumor-bearing mice
Approximately 1 × 107 MCF-7 cells were collected in 150 µL of serum-free medium and subcutaneously injected into the right axilla of 6-week-old female BALB/c mice (n = 5). LHRH-Fe3O4 or Fe3O4 was then intravenously administered every 5 days to detect the toxicity
Statistical analysis
IBM SPSS Statistics for Windows, Version 19.0 (IBM Corp., Armonk, NY, USA) was used to analyze the data. The measurement data are expressed as mean ± standard deviation. The independent-samples
Ethics
All animal experiments and procedures were approved by the Institutional Animal Care and Use Committees of Bengbu Medical College (Permit Number: BMC -20160047).
Results
LHRH-Fe3O4 nanoparticle analysis
The particle size of Fe3O4 and LHRH-Fe3O4 was 8 and 10 nm, respectively, as measured by TEM (Figure 1(a)). The effective particle size of the LHRH-Fe3O4 nanoparticles was 10 nm as measured by a particle size analyzer, and the dispersity index was 0.277, indicating that the nanoparticles were uniformly dispersed as shown in Figure 1(b). These findings were basically consistent with the results of TEM.
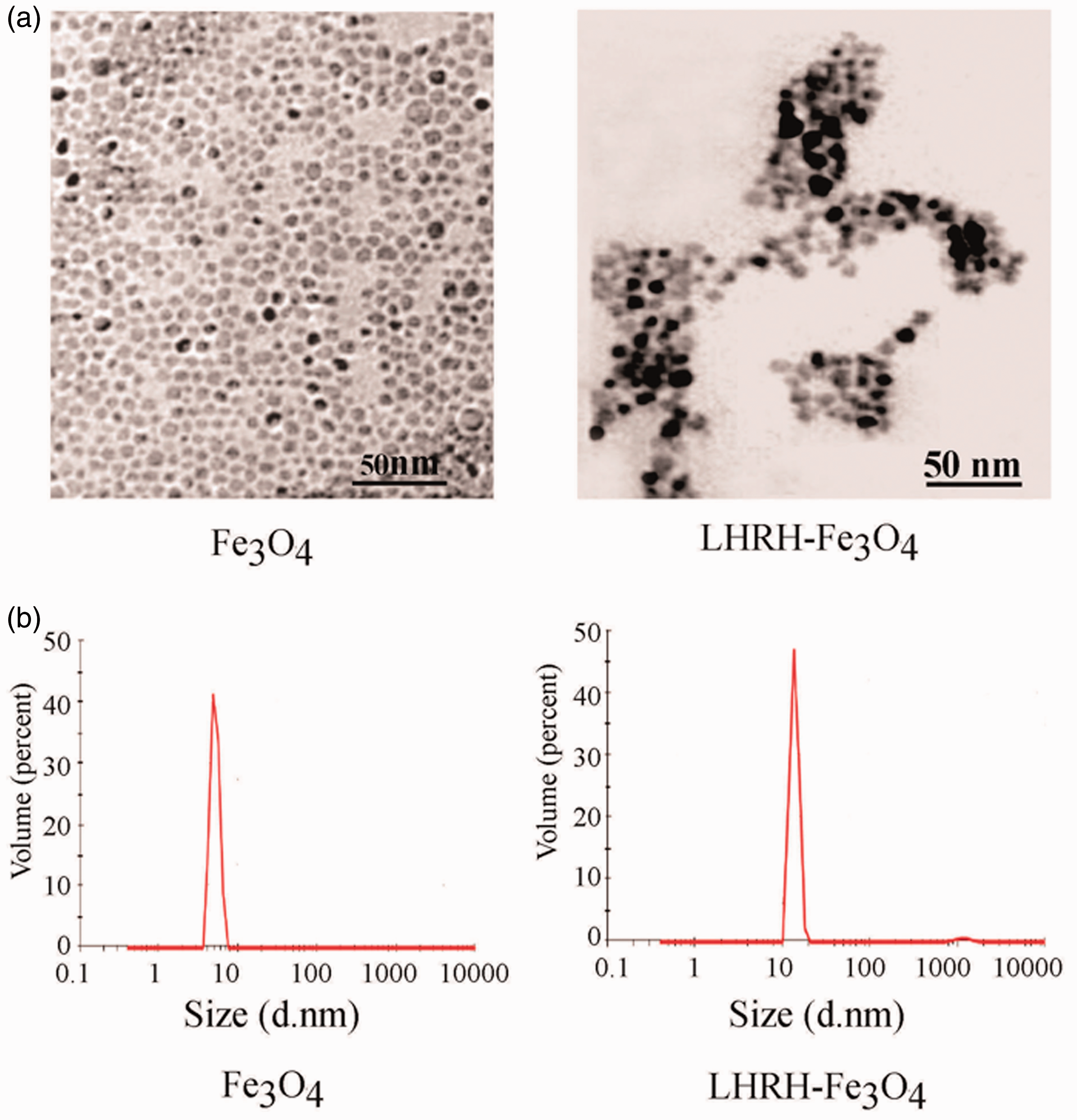
LHRH-Fe3O4 nanoparticle analysis. (a) The nanoparticle was analyzed by transmission electron microscopy. (b) The nanoparticle was analyzed by a particle size analyzer. LHRH-Fe3O4, luteinizing hormone-releasing hormone–ferrosoferric oxide.
Uptake of LHRH-Fe3O4 nanoparticles by tumor cells
LHRH-Fe3O4 or Fe3O4 at a final concentration of 1, 10, or 50 µg/mL was co-incubated with MCF-7 cells for 1, 2, 4, or 8 hours. Different levels of iron particles were present in the cells, and the levels of iron particles increased with the concentration and duration of LHRH-Fe3O4 or Fe3O4 administration. There was significant difference in the levels of iron particles between LHRH-Fe3O4 and Fe3O4 at 10 and 50 µg/mL (p < 0.05) (Figure 2).

Uptake of LHRH-Fe3O4 nanoparticles by tumor cells (n = 5). LHRH-Fe3O4, luteinizing hormone-releasing hormone–ferrosoferric oxide.
Cytotoxicity of LHRH-Fe3O4 nanoparticles
In this experiment, the cytotoxicity of the nanomaterial was detected using a Cell Counting Kit-8. As shown in Figure 3, both groups exhibited significant concentration- and time-dependence (p < 0.05). There were no significant differences in the inhibition rates at all concentrations between the LHRH-Fe3O4 group and Fe3O4 group. When the concentration of LHRH-Fe3O4 was 50 µg/mL for 72 hours, the inhibition rate in the LHRH-Fe3O4 group was 32.26%, while the inhibition rate in the Fe3O4 group was 23.12%. This result showed that the nanomaterial had limited cytotoxicity.

Cytotoxicity of LHRH-Fe3O4 nanoparticles was assayed by a Cell Counting Kit-8 (n = 5). LHRH-Fe3O4, luteinizing hormone-releasing hormone–ferrosoferric oxide.
Tissue distribution and targeted MRI of LHRH-Fe3O4 nanoparticles
Targeted MRI showed that tumor enhancement was not obvious at 5 minutes. After 1 hour, the edge of the tumor began to enhance, the degree of enhancement was low, and the intensification gradually became more obvious as time passed. The intensification diffused from the periphery to the center and peaked at 4 hours. Slight enhancement was observed at 24 hours. The liver tissue of nude mice began to enhance at 5 minutes, peaked at 2 hours, and was not obvious at 4 hours. There was no obvious enhancement in the lung and heart tissues (Figure 4(a)). Moreover, the signal values of each scanning point after injection of LHRH-Fe3O4 were paired with the signal value before the LHRH-Fe3O4 administration. The results showed that the signal value of the tumor tissue was not significantly different from the injection of LHRH-Fe3O4 at 5 minutes but was significantly different at 1, 2, 4, 8, and 24 hours (all p < 0.05). The peak tumor enhancement occurred at 4 hours after injection of LHRH-Fe3O4, and the maximal enhancement rate was 66.88%. These results showed that LHRH-Fe3O4 could effectively concentrate on the tumor under the action of a magnetic field (Figure 4(b)).

Tissue distribution and targeted magnetic resonance imaging of LHRH-Fe3O4 nanoparticles. (a) Magnetic resonance imaging after LHRH-Fe3O4 administration. (b) Enhancement rate analysis after LHRH-Fe3O4 administration (n = 5). (c) Tumor growth to detect the toxicity
In our evaluation of toxicity
Discussion
LHRH receptors are not expressed in normal cervical connective tissues, squamous epithelium, or hepatocytes, but many LHRH receptors with high affinity are present in cervical adenocarcinoma, colon cancer, and liver cancer, and other types of cancer. 13 At present, some laboratories use LHRH targeting for treatment of tumors. Keller et al. 16 reported that AN-207, the analog of LHRH (targeted LHRH receptor), was used to treat renal cell carcinoma. Nechushtan et al. 17 made LHRH and PE66 (the mutant of diphtheria toxin) into a fusion protein that exhibited different levels of inhibition on cell growth of colon cancer, ovarian cancer, breast cancer, and liver cancer. Kakar et al. 18 showed that the conjugate of triptorelin and Fe3O4 had a significant killing effect on MCF-7 and MDA-MB321 cells, suggesting that LHRH plays a very important role in targeted therapy of tumors. Leuschner et al. 19 found that LHRH-superparamagnetic iron oxide nanoparticles (SPIONs) were highly aggregated in human breast cancer. The monosome SPIONs as controls were mainly aggregated in the liver and had very low affinity for tumors, also suggesting that LHRH-SPIONs can enhance the sensitivity of MRI examination of breast tumors.
In the present study, the particle size of LHRH-Fe3O4 nanoparticles was 10 nm, and they could be taken in by human breast cancer cells. The nanomaterial showed limited inhibition of tumor growth
In conclusion, LHRH-Fe3O4 nanoparticles serve the purpose of targeting contrast agents to target sites and are thus expected to be used for targeted imaging and treatment of cancers with high expression of LHRH.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
This work was supported by the Natural Science Foundation for Universities of Anhui Province (General Projects, KJ2015B094by).
